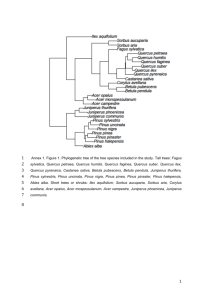
Community Analysis of Duke Forest Vegetation using PCOrd
advertisement

Community Analysis of Duke Forest Vegetation using PCOrd Annemarie Nagle Biology 112 12/5/05 Introduction Computer modeling and statistical analysis have become extremely valuable tools in the modern field of ecology. Whereas ecologists have traditionally characterized forest communities using environmental gradients that dictate species distribution, and have been forced to examine community structure strictly through the lens of obvious causal relationships between these two factors, the advent of statistical analysis software allows more sophisticated and objective comparison of complex data. In these situations where causal relationships between environmental or other factors and species presence and abundance are unknown or assumed to be unknown, analysis programs like S-PLUS and PCord allow ecologists to analyze relationships within their data and allow them to create an accurate picture of complex variable interactions that would be nearly impossible without these tools. In this analysis, PCord was used to examine the community structure of a large section of Duke Forest, in Durham and Orange Counties, North Carolina. Tree species presence and abundance data was collected for 106 plots in the Duke Forest, and corresponding environmental data was measured in each plot as well. Preliminary analysis consisted of obtaining the optimal number of axes through which to examine the data, and looking at possible correlations between species distribution and environmental variables. Next, a grouping system was created, which placed the plots into various categories based on their relationships to one another. Finally, indicator species analysis was used to characterize these groups and obtain an idea of their structure. Methods Stem counts of woody species in Duke Forest plots were used to obtain frequency data for 56 tree species, while environmental sampling methods measured pH, calcium, magnesium, potassium, aluminum, manganese, phosphate, and organic matter content, sand, silt, clay, aspect, slope, distance from water and water content, solar radiation, and elevation. Using PCord, a step-down ordination was carried out to determine the minimal number of axes that would accurately represent trends in the data in an interpretable number of dimensions. Sorenson (Bray-Curtis) analysis was used. Results of this test ordination were examined to ensure minimal stress values and low instability. The optimal number of axes (3) was selected and used to create a focal run for the data. See Table 1 for ordination settings. Setting NMS Step-down Run Distance measure Sorensen (Bray-Curtis) Number of axes 6 Number of runs real data 20 Stability Criterion 0.0005 Max # iterations 400 Iterations to evaluate stability 20 Initial step length 0.20 Table 1. NMS settings for step-down and focal ordinations. NMS Focal Run Sorensen (Bray-Curtis) 3 20 0.0005 400 20 0.20 The data obtained from the NMS focal run was used for preliminary comparison of assigned axes and environmental and species variables. Biplots and r values for individual species correlations with the assigned axes were accessed under the Graph Ordination menu of PCord. Next, cluster analysis was performed to show relationships among plots and attempt to define a number of community types based on species composition represented in the data. Settings for the cluster analysis are shown below in Table 2. Cluster analysis output was examined to ensure minimal values of percent chaining in the data. Setting Distance measure Linkage method Add group membership variable Beta Group membership level Write all higher level groupings Table 2. PCord cluster analysis settings. Cluster Analysis Sorensen (Bray-Curtis) Flexible beta Yes -0.25 6 Yes In order to determine which level of grouping was the most biologically significant and useful, a Monte Carlo Test was performed at 1000 runs for each possible level of grouping (3-6 groups). P-values from the Monte Carlo Test were then examined to determine which number of groups was best. A maximum number of significant (p ≤ 0.050) p-values and a minimal average of p-values was used as indicator of the best number of groupings. Indicator species analysis was performed on the species abundance data using the number of groups shown to be optimal in the Monte Carlo Test by examining abundance, frequency, indicator, and p-values of species within the group. Results Three axes were selected for the focal ordination via the step-down run. Stress values at three axes were slightly less than optimal at 15.95997 (values less than or equal to ten are preferred). Instability values were very good, however, at 0.00046. Despite sub-optimal stress values, the slope of the scree plot shown in Figure 1 provides a clear picture as to why three dimensions were selected. The minimum number of dimensions corresponding to minimal increase in stress value is desired, and as one can see with fewer than three dimensions levels of stress being to increase rapidly. Step-Down NMS Scree Plot 40 Real Data Stress 30 20 10 0 1 3 5 Dimensions Figure 1. NMS scree plot showing optimal 3 dimensions for focal run. Examination of cluster analysis results indicated that the analysis was a good representation of groupings in the data, and provided valuable information regarding relationships in groups. Percent chaining was low, at only 1.66%. Number of Groups Average p-value Number Significant p-values 3 0.158786 22 4 0.152929 11 5 0.169357 21 6 0.129429 28 Table 3. Summary of p-values from Monte Carlo Test to determine the appropriate number of groups for use in indicator species analysis. The lowest average p-value and highest number of significant p-values was seen in six groups, so this grouping was selected. Summary: 6 Group Analysis Distance (Objective Function) 1.6E-02 5.1E+00 100 75 1E+01 1.5E+01 2E+01 25 0 Information Remaining (%) 00001 PSP37 00018 00021 00574 00004 00008 00014 00002 00005 00007 00509 00520 00016 00024 00023 00033 00010 00031 00020 00042 00019 00069 00517 PSP36 00555 00581 00598 00589 00571 00579 00009 00012 00011 00015 00017 00022 00067 00618 00513 00514 00620 00537 00619 00501 00504 00524 00502 PSP88 PSP86 00508 PSP87 00617 PSP35 PSP34 00081 00606 00590 00510 00596 00512 00511 00607 00621 00608 00609 00003 00612 00616 00611 00614 00615 00622 00624 00575 00593 00582 00013 00029 PSP44 PSP61 00503 00507 00025 00026 00583 00584 00585 00032 00505 00506 00625 PSP10 00027 00587 00518 00588 00602 00515 PSP43 00028 00516 00030 00610 00613 00623 50 Group6 1 3 12 36 37 87 Figure 2. Cluster dendrogram color-coded to show groupings of sample plots into six categories. TreeLong_NMS Group6 1 3 12 36 37 87 LIST Axis 3 CACR QUPR QUAL Axis 2 Figure 3. PCord biplot of axes 2 and 3 showing plots colorcoded by group membership and corresponding species. TreeLong_NMS Group6 1 3 12 36 37 87 LIST Axis 3 CACR LITU FAGR JUVI QUST QUAL Axis 1 Figure 4. PCord biplot of axes 1 and 3 showing plots colorcoded by group membership and corresponding species. TreeLong_NMS Group6 1 3 12 36 37 87 JUVI QUST Axis 2 LITU FAGR QUPR Axis 1 Figure 5. PCord biplot of axes 1 and 2 showing plots colorcoded by group membership and corresponding species. TreeLong_NMS group6 1 3 12 36 37 87 Axis 3 Mg-A Ca-A pH Elev Dist-H2O Axis 2 Figure 6. PCord biplot of axes 2 and 3 showing plots colorcoded by group membership and corresponding environmental variables. TreeLong_NMS Axis 3 group6 1 3 12 36 37 87 Mg-A Ca-A Dist-H2O Axis 1 Figure 7. PCord biplot of axes 1 and 3 showing plots colorcoded by group membership and corresponding environmental variables. TreeLong_NMS group6 1 3 12 36 37 87 Axis 2 pH Axis 1 Figure 8. PCord biplot of axes 1 and 2 showing plots colorcoded by group membership and corresponding environmental variables. Species Abbreviation QUPR PITA FRAX Species Name Quercus prinus Pinus taeda Fraxinus sp. Abundance Frequency 0 84 1 0 100 100 Indicator Value 98.6 83.6 73.6 p-value 0.001 0.001 0.001 Max Group 36 87 37 FAGR PIVI ULAL JUVI ILDE LIST QUST CECA LITU ULRU CACR OSVI QUAL MORU CAOV PIEC QURU QUCO OXAR QUVE QUMI COFL CAGL CATO ACRU PRSE QUPH CAOL NYSY ACNE QUSH PLOC PRAM CRMA CACO CEOC DIVI ILOP CAPA ACSA BENI ULAM QUMA QUFA JUNI MATR CACA SAAL Fagus grandifolia Pinus virginiana Ulmus alata Juniperus virginiana Ilex decidua Ligustrum sinense Quercus stellata Cercis canadensis Liriodendron tulipifera Ulmus rubra Carpinus carolina Ostrya virginiana Quercus alba Morus rubra Carya ovata Pinus echinata Quercus rubra Quercus coccinea Oxydendrum arboreum Quercus velutina Quercus michauxii Cornus florida Carya glabra Carya tomentosa Acer rubrum Prusus serotina Quercus phellos Carya ovalis Nyssa sylvatica Acer negundo Quercus shumardii Platanus occidentalis Prunus americana Crataegus marshallii Carya cordiformis Celtis occidentalis Diospyrus virginianus Ilex opaca Carya pallida Acer saccharum Betula nigra Ulmus americana Quercus marilandica Quercus falcata pagodaefolia Juglans nigra Magnolia tripetala Carya carolinaeseptenrionalis Sassafras albidum 0 69 7 63 0 3 59 0 0 0 0 0 1 0 0 39 0 0 0 0 0 0 0 1 4 12 6 0 0 0 0 0 0 0 0 0 19 0 0 0 0 0 19 3 0 0 0 0 100 67 100 0 100 100 0 0 0 0 0 67 0 100 100 0 0 0 33 0 67 67 100 100 33 100 0 67 0 0 0 0 0 0 0 100 0 0 0 0 0 67 33 0 0 0 73.1 69.2 66.4 63 61.8 59.7 59.1 58.9 58.3 48.7 48.6 45.2 44.4 42.9 39.4 38.8 37.7 37.4 37.2 35.5 35.3 34.5 34.4 34 33.3 32.8 29 28 27.7 27.3 27.3 26.1 25 23.2 21.8 21.6 18.8 18.4 18.1 15.2 14.8 14.5 13.7 13.2 12.6 10.3 9.1 0.001 0.003 0.004 0.001 0.002 0.001 0.001 0.007 0.001 0.011 0.016 0.013 0.001 0.031 0.056 0.029 0.051 0.027 0.03 0.029 0.008 0.033 0.065 0.096 0.059 0.209 0.029 0.15 0.236 0.028 0.038 0.047 0.054 0.042 0.052 0.123 0.291 0.2 0.058 0.35 0.145 0.24 0.281 0.443 0.259 0.159 0.22 12 87 3 37 3 3 87 37 12 3 3 37 1 37 87 87 37 36 36 1 3 1 37 87 1 37 87 37 36 3 3 3 37 3 3 37 87 12 36 12 3 3 87 1 3 12 3 0 0 9.1 0.729 1 QUNI CRUN AMAR ILAM CRAT Quercus nigra Crataegus uniflora Amelanchier arboreum Ilex ambigua Crataegus sp. 0 0 0 0 0 0 0 0 0 0 8.4 7.7 7.5 6.3 5.9 0.272 0.315 0.578 0.585 0.536 Table 4. Summary statistics of Monte Carlo Test and indicator species analysis for six groups sorted by IV. Discussion Cluster analysis allowed the beginnings of characterization of possible tree communities in the Duke forest. By examining a series of biplots of species and environmental variables overlaid on plot arrays color-coded to reflect group membership, possible defining environments and compositions were identified for the groups. We were also able to better assess outliers or misleading trends in the data. Groups were not constructed by taking into account environmental variable relationships between the plots, but were solely based upon species composition and distribution. However, it is widely recognized that environmental variables often influence individual species distributions, so it is valuable to compare group relationships with these variables. The groups labeled 37(blue) and 36(pink) seemed to lie along a pH gradient, with 37 on the positive end and 36 on the negative. Group 12(light blue) may have a weaker positive correlation with pH. Group 1(red) is one of the largest groups, and seems to be correlated with Dist-H20 and Mg-A, as it seems Group 3(green) is as well. Group 1 has a positive correlation with Dist-H20 and a negative correlation with Mg-A, while group 3 has the opposite. Groups 36 and 37 are also located in the region of group 1, and appear to have similar but less strong correlations with the two variables. Aluminum also seems to occur most frequently in group 1. Acer rubrum was highly correlated with Axis 2. When viewed with its groupings, however, Acer rubrum seems to occur in many of the groups (most abundantly in 12 and 36), and so possibly may be viewed as abundant everywhere and less weight given to its correlation with the axis. Carpinus carolina occurs most abundantly in groups 12 and 3, so may be characterized by higher pH and longer distance from water and higher values of Mg-A. Fraxinus sp. is found almost exclusively in group 37, which may be present where pH values are higher. Ligrustrum sinense is found in groups 3 and 12. Liriodendron tulipifera, although not corresponding strongly to any axis seems to fall primarily in group 12. A number of oak species, Quercus alba, Quercus velutina, and Quercus stellata seem to occur at the highest frequency in group 1. Quercus prinus is located almost exclusively in group 36. Ulmus alata appears to be nearly exclusively in group 3. Group 87 (yellow) was difficult to assign qualities due to lack of trends in environmental or species content, and due to it being the smallest group in the analysis. Visual examination of the data discussed above provided insight as to what may drive community structure and what characterizes the six types of communities defined by the analysis. However, the statistics provided by the Monte Carlo and Indicator Species analyses are able to give us a more objective picture of species composition in the created groupings. Pvalues are powerful indicators of the likelihood that a pattern observed has occurred by chance. Low p-values (here, for p ≤ 0.050) tell us that our results are significant and have real meaning. 3 36 1 3 37 Abundance (fidelity) and frequency (constancy) can be combined to give indicator values, which discuss the degree to which a particular species is diagnostic for the grouping that it is placed in. Abundance is the percentage of the total occurrence of a species that is found within the group of interest. Frequency refers to the percentage of the plots within the group of interest that contain a species. A perfect indicator species for a group would only be present in the group of interest (100% of its abundance) and would be present in every plot contained within the group (frequency would also be 100%). Our analyses returned no perfect indicator species. The species with the maximum IV reported (98.6), Quercus prinus, did not occur in any of the plots and so can be omitted. However, Pinus taeda, with an IV of 83.6, seems to be a nearly perfect indicator for the group 87. Pinus virginiana is also a good indicator (IV= 69.2) species of group 87. The existence of two strong indicators from the Pinus genera may indicate that this group represents communities characterized by dominance of pines. For group 37, Juniperus virginiana is the best indicator (IV= 63). These were the closest indicators to “perfect” reported by the data. Indicator species for the other groups were farther from “perfect”. The best indicator reported for Group 3 was Ulmus alata (IV=66.4). The highest IV in Group 1 was only 44.4, which belonged to Quercus alba. There did not seem to be any meaningful indicator species for the other two groups, 36 and 12. From this data, it appears that Pinus taeda and Pinus virginiana may be used with relative confidence as indicator species for the community type contained in grouping 87. Juniperus virginiana could also be a valuable indicator. It is possible that this number of groupings was still too small to explain the patterns seen on the landscape of Duke Forest, or that the communities may be too similar or the scale of sampling too small to separate the data into distinct groups with unique indicator species. Groups that do not appear to have any correlation to any measured environmental variables may be characterized by variables that were not tested. Further analysis is needed to determine the characteristics of the individual groups and possible biotic or abiotic explanatory factors contributing to observed species distributions.