Inorganic Chemistry Test I
advertisement

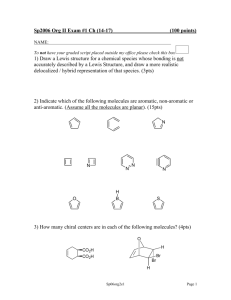
Inorganic Chemistry Homework I 2013. 03.26 1. In the hydrogen atm, the 3s and 3p orbitals have identical energies, but in the chlorine atom the 3s orbital lies at a considerably lower energy than the 3p. Explain. 2. Estimate the effective nuclear charge of F atom. 3. For each of the following pairs, decide which has greater bond angle and defend your answer. (a) NO2+ or NO2(b) PF3 or PCl3. 4. Arrange the following compounds in the increasing order of basicity of oxygen. your logic. cyclobutaneone, cyclopentanone, cyclohexanone, acetone. Explain 5. Iodine forms several anions of the type IOxn-. Write Lewis structures for IO3- and IO4-. Predict the relative I-O bond lengths in these oxyanions. 6. Arrange the following in the increasing C-N bond energy order and explain your logic. CN-, CNO-, NCO7. Predict the geometries of (CH3)2P(CF3)3 and (CH3)3P(CF3)2 8. Give a short answer for each of the following. (a) List the atoms in the increasing order of electron affinity: (b) C=C double bond distance (c) N-N bond distance (d) List the compounds in the order of CO values: CH3C(O)CH3, CH3C(O)Cl, ClC(O)Cl, FC(O)F O, S, Se 9. Describe the structure and assign the following molecules to their point groups. (a) BrF3 (b) TeF510. Deduce the symmetry species and number of normal mode vibrations for the cis- form of glyoxal, HC(=O)-C(=O)H. Indicate which are infrared active and which are Raman active. (Please see the Character Tables) 11. For the six-coordinated Fe(II) carbonyl complex, [Fe(CO)4Cl2], there exist cis- and transisomers. Determine the number of IR-active C-O stretching vibrations. (Please refer the character tables) 12. Identify each of the following and estimate (within 25%) the magnitude of the quantity specified (specify the units). a) The element having the highest ionization potential b) The element having the highest electron affinity c) The neutral diatomic molecule having the shortest interatomic distance d) The neutral diatomic molecule having the highest bond dissociation energy