Genetic transfer in bioleaching microorganisms
advertisement

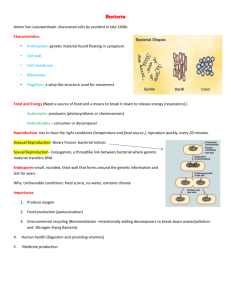
Genetic transfer in bioleaching microorganisms Violaine Bonnefoy Laboratoire de Chimie Bactérienne, Institut de Biologie Moléculaire et de Microbiologie, C.N.R.S., 13402, Marseille Cedex 20, France Genetic transfer techniques allow the introduction of genetic material into cells. The three classical approaches are: transduction - the transfer of genetic information via a bacteriophage (virus) particle (Figure 1) conjugation - the transfer of conjugative or mobilizable plasmids from one bacterium to another by cell-to-cell contact (Figure 2) electrotransformation, by exposing the cells in the presence of free DNA to a pulsed electric field which destabilizes transiently the bacterial membrane and permits the entry of the DNA into the cell (Figure 3). Genetic transfer from one microbe to another can be used to express heterologous genes in the recipient bacteria or to bring back genes that have been modified in more accurate hosts by genetic engineering. The later approach allows the construction of mutants, precisely defined at the molecular level, which can help elucidating the physiology of these microbes or improve some of their specific metabolic properties. Genetic transfer in bioleaching microbes is a real challenge because their life conditions are extreme and quite different from the "classical" bacterium Escherichia coli. Genetic engineering is usually performed in this eubacterium which is consequently often used as donor cells. While the former are strict or moderate acidophiles and obligatory or facultative chemoautolithotrophs, the later is neutrophilic and heterotrophic. The main problem is therefore to find the conditions in which both donor and recipient cells have enough energy to survive. Furthermore, 15/02/2016 bioleaching microbes grow slowly with very low cell yields making them difficult to culture. Nevertheless, genetic transfer was made possible in some bioleaching microbes (Table 1). This has allowed the expression of the phosphofructokinase gene (ptkA) of E. coli in At. thiooxidans (12). More interestingly the transposon Tn5 was shown to be able to transpose into the chromosome of At. ferrooxidans, opening the way to random transposon insertion mutagenesis (9). Finally, conjugation by marker exchange mutagenesis has allowed the construction of the recA mutant in At. ferrooxidans (7), the only "constructed" mutant described so far in bioeaching microbes. Bacterium Genetic transfer technique Acidiphilium sp. electropermeabilization conjugation Acidithiobacillus conjugation thiooxidans Acidithiobacillus electropermeabilization ferrooxidans Acidithiobacillus conjugation ferrooxidans Reference 2; 3 1; 2; 10; 11 4; 12 5 6; 7; 8; 9 References: 1. Bruhn, D. F. and F. F. Roberto (1993). Maintenance and expression of enteric arsenic resistance genes in Acidiphilium. Biohydrometallurgical Technologies. A. E. Torma, Wey, J. E. and Lakshmanan VI. Warrendale, PA, The Minerals, Metals and Materials Society. II: 745-754. 2. Glenn, A. W., F. F. Roberto, and T. E. Ward (1992). Transformation of Acidiphilium by electroporation and conjugation. Can J Microbiol 38: 387-93. 3. Inagaki, K., J. Tomono, N. Kishimoto, T. Tano and H. Tanaka (1993). Transformation of the acidophilic heterotroph Acidiphilium facilis by electroporation. Biosci Biotechnol Biochem 57: 17701. 4. Jin, S. M., W. M. Yan, and Z. N. Wang (1992). Transfer of IncP plasmids to extremely acidophilic Thiobacillus thiooxidans. Appl. Environ. Microbiol. 58: 429-430. 5. Kusano, T., K. Sugawara, C. Inoue, T. Takeshima, M. Numata and T. Shiratori (1992). Electrotransformation of Thiobacillus ferrooxidans with plasmids containing a mer determinant. J Bacteriol 174: 6617-6623. 6. Liu, Z., F. Borne, J. Ratouchniak and V. Bonnefoy (2001). Genetic transfer of IncP, IncQ and IncW plasmids to four Thiobacillus ferrooxidans strain by conjugation. Hydrometallurgy 59: 339-345. 7. Liu, Z., N. Guiliani, C. Appia-Ayme, F. Borne, J. Ratouchniak and V. Bonnefoy (2000). Construction and characterization of a recA mutant of Thiobacillus ferrooxidans by marker exchange mutagenesis. J Bacteriol 182: 2269-2276. 8. Peng, J., Yan, W. and Bao, X. (1994). 15/02/2016 Expression of heterologous arsenic resistance genes in the obligately autotrophic biomining bacterium Thiobacillus ferrooxidans. Appl. Environ. Microbiol. 60: 2653-2656. 9. Peng, J. B., W. M. Yan, and X. Z. Bao (1994). Plasmid and transposon transfer to Thiobacillus ferrooxidans. J Bacteriol 176: 2892-7. 10. Quentmeier, A. and C. G. Friedrich (1994). Transfer and expression of degradative and antibiotic resistance plasmids in acidophilic bacteria. Appl Environ Microbiol 60: 973-8. 11. Roberto, F. F., Glenn, A. W., Bulmer, D. and Ward, T. E. (1991). Genetic transfer in acidophilic bacteria which are potentially applicable in coal beneficiation. Fuel 70: 595-598. 12. Tian, K. L., J. Q. Lin, X. M. Liu, Y. Liu, C. K. Zhang and W. M. Yan (2003). Conversion of an obligate autotrophic bacteria to heterotrophic growth: expression of a heterogeneous phosphofructokinase gene in the chemolithotroph Acidithiobacillus thiooxidans. Biotechnol Lett 25: 749-54. Figure 1. Transduction i. Phage infection ii. DNA hydrolysis iii. DNA packaging in phage capsid iv. transducing phage infection to a new microbial cell v. Recomibination between the microbe chromosome and the DNA transduced by the phage 15/02/2016 Figure 2. Conjugation i. ii. A conjugation tube forms between the donor cell and the recipient cell. A single strand from the plasmid DNA is transferred throught the tube from the donor to the recipient cells. iii. A double-stranded DNA is formed from the single strand in both the donor and the recipient cells iv. 15/02/2016 Figure 3: Electrotransformation i. ii. When the electric fiel is applied, the ions move according to their charge iii. Pathways are formed across the membrane allowing the entrance of DNA iv. When the electric field is stopped, the membrane heals 15/02/2016