Protocol for Transfecting and Fixing RBL Cells using BTX
advertisement

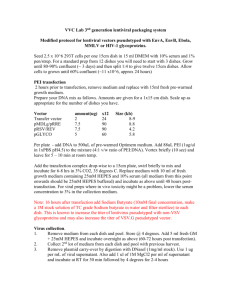
Protocol for Transfecting and Fixing RBL Cells using BTX Electroporator Transfection: 1) Prepare Cytomix buffer as described in AfCS protocol’s PS00000043300 and PS00000040800. ATP and GSH can be added to the stock before freezing with only a modest decrease in transfection efficiency/cell survival. 2) After spinning down cells, resuspend in desired amount of Cytomix. A confluent flask (~2 x 107 cells) will be good for 15-20 transfections, assuming 1 x 106 cells/transfection. More cells can be used, up to 3 x 106. In any case, final volume should be 300ul. 3) Add 10ug of YFP construct and 20ug of CFP construct. DNA amounts are not absolute, and less can be used with a decrease in efficiency if necessary (keep in mind that some constructs transfect poorly, especially CFP versions). 4) Shake cells on orbital shaker (GeneMate OS350) at setting 4 for 10 min to mix DNA/cell suspension. 5) Place each tube a well of a 96-well plate (preferably Fisher cat. # 07-200-94). 6) Clean 8-well electrode well with PBS and ethanol and place in 96-well plate. 7) Pulse at 200 volts for 50ms. 8) Resuspend each transfection in media to desired concentration (assume 50% cell death). 9) Plate in desired format. For 8-well imaging plates, use ~75 000 cells/well, and a total volume of 400-500ul for even cell distribution (higher volumes cause cells to cluster around the edges of the well). 10) Incubate for expression at 37C 6 hours to overnight. Some constructs are not compatible with cell survival, especially dominant negatives: keep this in mind when determining incubation times prior to fixation/imaging. Fixation: 1) If stimulating, sensitize cells for 1 hour to overnight with IgE anti-DNP (1ug/mL), wash 3 times, and then stimulate with 800ng/mL DNP-BSA for 5min at 37C (0.1% BSA, 10mM Glucose, in extracellular buffer). 2) Remove DNP-BSA buffer and add 500ul of 4% para-formaldehyde (cold). 3) Incubate at 4C for 5min, followed by RT 15min. 4) Remove PFA by aspiration, wash once with PBS, and then add 500ul PBS for storage and imaging. 5) Cells will retain fluorescence for ~10 days without mounting at 4C.