Introduction - University of Canterbury
advertisement

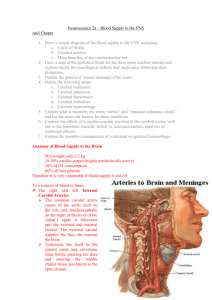
Lumped Parameter and Feedback Control Models of the Auto-Regulatory Response in the Circle of Willis KT Moorhead University of Canterbury, Christchurch, New Zealand CV Doran University of Canterbury, Christchurch, New Zealand JG Chase University of Canterbury, Christchurch, New Zealand T David University of Canterbury, Christchurch, New Zealand Introduction The Circle of Willis (CoW) is a ring-like structure of blood vessels found beneath the hypothalamus at the base of the brain, which distributes blood to the cerebral mass. Simple models of cerebral blood flow dynamics would create a tool capable of diagnosing potential outcomes of surgical or other therapies. A one-dimensional flow model is developed to capture the auto-regulation dynamics by which cerebral blood perfusion is maintained. Figure 1 shows a CoW schematic composed of circulus, afferent (inflow) and efferent (outflow) arteries. Positive flow around the CoW is clockwise but not restricted in direction, while flow in afferent and efferent vessels is restricted to the directions shown. Flow in efferent vessels is regulated by time varying peripheral resistances modelling the effects of autoregulation in the smaller blood vessels they supply. While many individuals have complete and symmetrical CoW geometry, it is not uncommon for some elements to be restricted or omitted (van der Zwan and Hillen 1991) with the communicating vessels having a higher occurrence of omission (Alpers and Berry 1963). Abbreviation RRMCA BA RRPCoA RRACA1 RRACA2 LPCA1 Left proximal Posterior Cerebral Artery LPCoA Left Posterior Communicating Artery LICA RRPCA1 RRICA RBA RACoA +ve RLICA RLACA2 RRPCA2 RLACA1 RLPCoA RLPCA2 RLMCA Left Internal Carotid Artery LACA1 Left proximal Anterior Cerebral Artery ACoA Anterior Communicating Artery RACA1 Right proximal Anterior Cerebral Artery RICA RLPCA1 Vessel Full Name Basilar Artery Right Internal Carotid Artery RPCoA Right Posterior Communicating Artery RPCA1 Right proximal Posterior Cerebral Artery LPCA2 Left distal Posterior Cerebral Artery LMCA Left Middle Cerebral Artery LACA2 Left distal Anterior Cerebral Artery RACA2 Right distal Anterior Cerebral Artery RMCA Right Middle Cerebral Artery RPCA2 Right distal Posterior Cerebral Artery Figure 1: CoW Schematic Resistance Model showing flow directions and the Abbreviations Used 1 Method The CoW is modelled with laminar, viscous and incompressible flow assumptions, with individual arteries being modelled by simple Poiseuille flow in a tube. The relationship can also be represented using an electrical circuit analogy. q P1 P2 , R (1) where P1-P2 is the pressure change across the artery, and R represents the arterial resistance to blood flow, q. Using Poiseuelle flow assumptions, the resistance, R, can be defined as a function of the artery length, l, artery radius, r, and dynamic viscosity of blood, μ. R 8l , r 4 (2) The afferent and circulus arteries are modelled with constant resistances, and the efferent arteries are modelled with variable resistors, as shown in Figure 1. Efferent arteries are limited to changes in radius of up to 40% with the following limit on resistance. (1 0.95) R ref R (1 0.95) R ref (3) The auto-regulation process can be described by a typical Proportional-IntegralDerivative (PID) feedback control system, where the error between desired and actual flowrates drives changes in resistance. u(t ) K p e K i edt K d de dt (4) where e is the error in the flowrate represented by e=(qref – q), and Kp, Ki and Kd are control gains. The peripheral resistance dynamics are therefore described by a first order system, with a driving input, u(t). R ( R Rref ) u(t ) (5) where is the time constant of the auto-regulatory response, and R and Rref are the actual and reference resistances of the efferent artery respectively. Equation (5) can be used with Equations (1) and (4) to create a non-linear system. A( x ) x (t ) b(t ) (6) where A(x) is a matrix containing constant resistances for each arterial element including those obtained from Equation (5), x(t) is a state vector containing timedependent flowrates through those arteries and nodal pressures at the end of the arteries, and b(t) is a vector containing time-dependent arterial and venous pressures. The basic data for length, and radius (Hillen et al. 1988), is used to calculate arterial resistance using Equation (2), which are then normalised to the LICA. Resistance in the ACA2, MCA, and PCA2 is set to approximate the ratio 6:3:4 in the asymptotic flow state (Hillen et al. 1986). The dynamic behaviour of the peripheral resistance enables recalculation of the resistances based on the error in efferent flowrates and the control input, to obtain the new x(t) vector. At each time step, inner iterations are used to find the equilibrium solution for the combined system of equations at that time step. Each time step is 2 solved this way until the final time is reached. Hence, this model can be used, given patient specific CoW geometry from MRI data, to predict the potential outcome of surgical procedures or other therapies – a significant clinical tool. Results Control gains Kp, Ki, and Kd, are determined to match the clinical data from Newell et al. (1994) by matching the percentage change and duration of the time-dependent velocity profiles in the MCA. The complete CoW is modelled with venous pressures of 4mmHg and an arterial pressure drop from 93mmHg to 73mmHg in the RICA using the specific arterial resistances from Ferrandez et al. (2000). The time constant is taken to be 3 seconds, and the proportional, integral, and derivative gains are of the order 0.1, 10, and 0.01, respectively. Simulations are carried out for the balanced configuration and each case where a single circulus vessel is omitted. Where only one vessel in the CoW is omitted, the system is able to obtain the reference efferent flowrates. These results match the 20% drop in flowrate, and 20 second response time of Newell et al. (1994) and the reductions in flowrate for the missing vessel configurations are in good agreement with Ferrandez (2002), as shown in Table 1. Table 1: Comparison of Results with Ferrandez % drop in flow through RMCA after 20% pressure drop in RICA Balanced Configuration Missing LPCA1 Missing LPCoA Missing LACA1 Missing ACoA Missing RACA1 Missing RPCoA Missing RPCA1 (Ferrandez, 2002) 18 Not simulated 18 18 18 20 20 Not simulated (Present Model) 19 19 19 20 20 21 19 19 A realistic high stroke risk case with decreases in radius of the LICA and RICA of 50% and 30% respectively, and a missing LPCA1 is modelled (Lodder et al., 1996). This case represents a situation where the individual would be hypertensive to maintain proper flow to the cerebral mass. When a pressure drop is simulated in the RICA, LICA and BA, the LPCA2 cannot return to its reference flow value representing a potential stroke, as shown in Figure 2. Figure 2: Efferent Response to 20% Afferent Pressure Drop - High Stroke Risk Case 3 Discussion The model created has simulated the omission of any single circulus artery and found that no such omission leads to failure in reaching the reference (required) efferent flowrates for a RICA stenosis. The omitted vessel simulations are in good agreement with known physiological data and prior results, verifying the model dynamics. This result highlights the known robustness of the CoW system in supplying the cerebral mass territories. The high stroke risk case highlights the models’ ability to capture more complex cases, and predict the response to changes in system dynamics. Conclusions The model is a significant improvement over pre-existing models in its incorporation of inner iterations at each solution time-step, recognising the non-linearity of the system, to find the equilibrium state within physiological limitations. Time varying resistance accounts for vaso-dilation and vaso-constriction of the efferent arteries such that a constant flowrate to the cerebral territories can be obtained after occlusion/stenosis in any afferent artery. Results show good correlation with prior results using CFD (Ferrandez et al., 2002) and the physiological data (Newell et al., 1994). The solution for the CoW closed loop lumped parameter system is obtained in a far shorter time period using this time-varying resistance model than with CFD methods, and requires significantly less computational effort while retaining a high level of accuracy. These results highlight the effectiveness of this computationally efficient model as a tool for determining potential outcomes of surgical or other therapies. References Alpers, B. and R. Berry (1963). "Circle of Willis in Cerebral Vascular Disorders, The Anatomical Structure." Archives of Neurology 8: 398-402. Ferrandez, A., T. David, et al. (2000). "Computational models of blood flow in the circle of Willis." Comput Methods Biomech Biomed Engin 4(1): 1-26. Ferrandez, A., T. David, et al. (2002). "Numerical models of auto-regulation and blood flow in the cerebral circulation." Comput Methods Biomech Biomed Engin 5(1): 7-19. Hillen, B., A. Drinkenburg, et al. (1988). "Analysis of Flow and Vascular Resistance in a Model of the Circle of Willis." J Biomech 21(10): 807-814. Hillen, B., H. Hoogstraten, et al. (1986). "A Mathematical Model of the Flow in the Circle of Willis." Journal of Biomechanics 19(3): 187-194. Lodder, J., Hupperts, R., Boreas, A., Kessels, F. (1996). “The size of territorial brain infarction on CT relates to the degree of internal carotid artery obstruction.” Journal of Neurology Apr;243(4):345-9. Newell, D., R. Aaslid, et al. (1994). "Comparison of Flow and Velocity During Dynamic Autoregulation Testing in Humans." Stroke 25: 793-797. van der Zwan, A. and B. Hillen (1991). "Review of the variability of the territories of the major cerebral arteries." Stroke 22(8): 1078-84. 4