Basics of Titration
advertisement

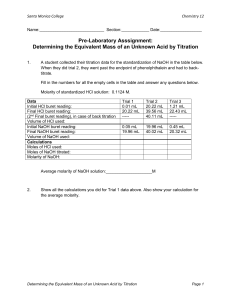
Basics of Titration By Drew Rutherford and David Mork Concordia College Introduction One of the most critically important areas of chemistry focuses on answering questions about the composition of matter. The fundamental question is often “What elements (molecules, ions) are present in the new material and in what quantities?” The first part of the question (What’s in there?) is qualitative analysis. The second part (How much?) is quantitative analysis. Scientists are constantly asking these questions of chemists. The specific branch of chemistry that attempts to answer these questions is called Analytical Chemistry. Analytical chemists have many techniques for identifying and quantifying elements, molecules and ions, but one of the most important is called titration. The technique of titration is often employed to determine the unknown concentration of solutions of acids or bases. This technique relies upon the acid-base reaction known as neutralization. In order to determine the concentration of an unknown acid, the chemist uses a base solution of known molarity. Likewise, if the unknown solution is a base, the chemist uses an acid solution of known molarity. In either case, the solution of known concentration is called the standard solution. Today’s experiment involves standardizing a sodium hydroxide solution with solid potassium hydrogen phthalate (KHP), and then using this standardized solution to determine the molarity of a hydrochloric acid solution. Hydrochloric acid and the base sodium hydroxide are known to react in the balanced acid-base neutralization reaction shown below. The reaction equation shows that 1 mole of sodium hydroxide will neutralize 1 mole of hydrochloric acid. HCl + NaOH NaCl + H2O A buret is volumetric glassware with marks on it at 0.1-mL intervals (like a graduated cylinder) with a stopcock on the bottom. The stopcock allows for slow additions of solutions (drop by drop if necessary). A known amount of solution may be added with a buret by knowing the starting and ending points of the meniscus on the graduated scale. When the number of moles of acid in the Erlenmeyer flask equals the number of moles of base added from the buret, the equivalence point is reached. In a titration, an indicator is used to signal when the equivalence point is reached (complete neutralization). An indicator will change color at the equivalence point. This color change is called the endpoint of the titration. Phenolphthalein, a common indicator, changes from colorless to pink at the endpoint when an acidic solution is titrated with a base. The calculations for a typical titration are given below. Assume 8.10 mL of 0.141 M NaOH solution were used to titrate 10.0 mL of HCl. First, determine the number of moles of NaOH that were consumed by the acid. Next, incorporate the stoichiometry of the reaction into your calculation. Finally, calculate the molarity of the acid. Step 1: Molarity = moles of base Liters of solution 0.141 M = moles of base 0.00810 L 0.00114 moles = moles of base Step 2: 0.00114 moles of NaOH x 1 mole HCl = 0.00114 moles HCl 1 moles NaOH Step 3: Molarity of acid = moles of HCl Liters of solution Molarity of HCl = 0.00114 moles HCl 0.0100 L Molarity of HCl = 0.114 M Procedure Use only deionized (DI) water for this (and any titration) lab. The quality of water used in any chemical analysis is critical to accurate and reproducible results. Tap water has high levels of minerals that interfere with chemical analyses. In titrations, these minerals contribute to pH variations that will seriously affect your results. Use deionized water when diluting stock solutions, dissolving KHP, diluting unknowns, and rinsing glassware. You may wash dirty glassware with soap and tap water, but make the final rinse with DI water. It is common practice to only use base solutions in a buret, rather than alternating between acid and base solutions depending upon which is your unknown. This keeps buret contamination issues at a minimum. Therefore, use only NaOH solutions in the buret. Once a titration is started, it is important that the volume of titrant in the buret is sufficient to determine the endpoint for the trial without refilling the buret. Refilling a buret in the middle of a titration will introduce significant errors in measurement. The most accurate titrations are those where 10-20 mL of NaOH are added to the acid solution in the Erlenmeyer flask. Potassium hydrogen phthalate (KHP) has to be dried overnight in an oven at 110oC, because it absorbs moisture from the air. When you are through measuring out your KHP, be sure to completely close the KHP container to prevent KHP from prolonged contact with the air. 1. Sodium Hydroxide (NaOH) solution – Prepare 800 mL of approximately 0.150 M NaOH from the provided stock solution of NaOH (4 M). Store your solution in a plastic screw-capped bottle. You will titrate this solution with KHP to determine its exact NaOH concentration. 2. Hydrochloric Acid (HCl) solution – Prepare 500 mL of approximately 0.200 M HCl from the provided stock solution of HCl (12 M). Store your solution in a plastic screw-capped bottle. You will titrate this solution with a standardized NaOH solution to determine its exact HCl concentration. 3. Standardize NaOH solution – Fill the buret with your “0.150 M” NaOH solution. It is unnecessary to fill the buret to the 0.00 mL mark. You should note the volume to which you fill the buret in your laboratory notebook. Weigh out 0.450 – 0.550 g of potassium hydrogen phthalate (KHP, MW = 204.23) and transfer completely into a 125-mL Erlenmeyer flask. Although the mass of KHP can be anywhere in this range, you must know the exact mass to four (4) significant digits. Completely dissolve KHP in about 40-50 mL of DI water. Add 2-3 drops of phenolphthalein indicator solution to the flask. Do not splash any solution out of flask, otherwise you will lose some KHP and have to remake the KHP solution. Place a sheet of white paper under the buret and situate the Erlenmeyer flask of KHP solution under the tip of buret (about 1-2” of buret tip should be inside flask). Add titrant (NaOH solution) from the buret until the phenolphthalein indicator reaches the endpoint, a change from colorless to pink (not red) that persists for 30 seconds. Hint: Calculate the volume of 0.150 M NaOH solution it will take to neutralize the KHP to help predict when to slow down the addition of titrant. A single drop under the endpoint and the pink color will fade; a single drop over the endpoint and the color will be too dark. It is wise to stop the addition too soon rather than too late. Swirl the solution every 5 mL early in the addition to promote mixing, and swirl often near the endpoint. Use a squirt-bottle filled with DI water to rinse down the sides of the flask and the buret tip prior to reaching the endpoint to ensure that all of the base has been washed into the acid solution. Once the endpoint has been reached, record the amount of titrant added using the scale on the buret. Repeat this procedure at least three times to ensure accuracy and precision. Sodium hydroxide and hydrogen phthalate (HC8H4O4- ) react according to the following balanced equation: NaOH + HC8H4O4- NaC8H4O4- + H2O From this information, determine the molarity of your NaOH solution. Do not forget to label your standardized NaOH solution. 4. Standardize HCl solution – Fill the buret with standardized NaOH solution. Transfer 10.00 mL of your HCl solution into a 125-mL Erlenmeyer flask using a volumetric pipette. Dilute with 40-50 mL of DI water. Add 2-3 drops of phenolphthalein indicator solution to the flask. Titrate the solution to the endpoint as described above. Repeat this procedure at least three times to ensure accuracy and precision. Using the equation for the reaction of sodium hydroxide with hydrochloric acid, determine the molarity of your hydrochloric acid solution and label your bottle properly. 5. Unknowns – Obtain your assigned unknown from your laboratory instructor or teaching assistant. Carefully transfer 5.00 mL of the unknown to a clean Erlenmeyer flask and add about 20 mL of DI water. Add 2-3 drops of the phenolphthalein indicator. Titrate to the endpoint. Repeat three times. Use the equation for the reaction of sodium hydroxide and hydrochloric acid to calculate the molarity of your unknown solution.