No 11: Rhizopus - Johns Hopkins Medicine

THE JOHNS HOPKINS MICROBIOLOGY NEWSLETTER Vol. 26, No. 11
Tuesday, July 17, 2007
A. Provided by Emily Luckman, Division of Outbreak Investigation, Maryland Department of Health and Mental Hygiene.
There is no information available at this time.
B. The Johns Hopkins Hospital, Department of Pathology, Information provided by,
Aatur Singhi, M.D.
Case presentation: A 29 year-old male with history of acute myelogenous leukemia, status post double cord blood transplantation with post-transplant course complicated by graft-versus-host disease and a episode of fungal pneumonia 1 year ago resulting in RLL pneumonectomy. He presents with hemoptysis and nonproductive cough for the last 3 days. He denies associated fevers, chills, rigors or shortness of breath. He has persistant lower chest wall pain that is exacerbated by coughing. He denies weakness or lightheadedness.
He continues to take immunosuppressants, however has been off his antifungal therapy (800mg posaconazole and 500 mg caspofungin daily) for several weeks with previously stable to improving CT scans. CT chest was ordered and remarkable for new infiltrate surrounding the previously stable LUL. Fungal culture of sputum was positive after 3 days for moderate wooly, gray to black colonies that quickly reached the lid of the Petri dish. Microscopically, nonseptate, root-like hyphae were visualized consistent with Rhizopus , most likely arrhizus .
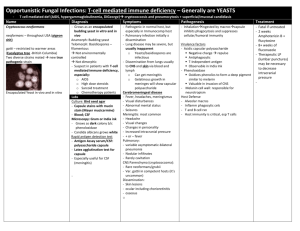
Epidemiology: Rhizopus spp. belong to the Zygomycetes class of fungi and are among the fungi causing the group of infections referred to as zygomycosis, an angio-invasive disease. Zygomycosis includes mucocutaneous, rhinocerebral, genitourinary, gastrointestinal, pulmonary and disseminated infections.
Rhizopus spp. are the most common organisms isolated from patients with zygomycosis with Rhizopus
(oryzae) arrhizus and Rhizopus microspores var. rhizopodiformis the species causing a majority of cases
(Ribes, 2000). Risk factors for development of zygomycosis include poorly controlled diabetes mellitus, hematologic malignancies (especially with neutropenia), receipt of a solid-organ or hematopoietic stem cell transplant (HSCT), deferoxamine therapy for iron or aluminum overload states, burn wounds, and corticosteroid therapy. The most dramatic presentations are in patients with diabetes who have ketoacidosis and who tend to have rhinocerebral (rather than pulmonary) zygomycosis.
More recently, an increasing number of cases involving zygomycosis have been found in patients receiving HSCTs. Siwek et al. document an increased rate at the University of Iowa with 4 cases, out of 45 transplants performed (8.9%) within a 1 year time period. These patients had all received voriconazole for prophylaxis prior to the time of transplantation and all were markedly immunosuppressed. All 4 received allogenic transplants and had aggressive therapy for biopsy-proven or presumed GVHD. Siwek et al. note that there were no cases among HSCT recipients in the preceding 3 years. In Boston, during a 10 month period, 4 of 124 HSCT recipients developed zygomycosis (3.2%) (Marty, 2004). The prior rate at their institution during the preceding 32 month period was 2 cases among 370 patients (0.5%). All patients who developed zygomycosis had received an allogenic transplant and all had GVHD. Further, in Seattle, 6 of 139 patients from 1998 to 2003 were identified to have zygomycosis (4.3%) (Imhof, 2004).
Laboratory diagnosis and fungal identification : Direct examination of specimen with Calcofluor white and culture are available as well as histopath. Zygomycetes are often considered common contaminations within the mycology laboratory, but the potential clinical implications of isolates should be investigated before they are dismissed as saprophytes. Zygomycetes grow within 48-72 hours and produce nonseptate or sparsely septate, broad hyphae that quickly reach the lid of the petri dish (“lid lifters”). The colonies are characterized as wooly, initially white, turning gray to black as spores are developed. Rhizopus spp. produce unbranched sporangiophores that support sporangia (40-350 um) with an ellipsoidal columella and containing sporangiospores. The sporangiophores are frequently brown in color and usually unbranched. They can be solitary or form clusters. The important diagnostic feature of Rhizopus is the presence of hyaline to brown root like structures called rhizoids. Rhizoids are located at the point where the stolons and sporangiophores meet.
Sporangia are located at the tip of the sporangiophores. They are round with flattened bases. Sporangiospores are unicellular, round to ovoid in shape, hyaline to brown in color and smooth or striated in structure.
Clinical aspects: The manifestations of zygomycosis depend on the location of involvement. One of the most common clinical presentations is rhinocerebral disease, which can manifest as unilateral, retro-orbital headache, facial pain, fevers and nasal sinusitis that progresses to black discharge. Late symptoms that indicate invasion of the optic nerve and vessels include diplopia and visual loss. These late symptoms indicate a poor prognosis and are usually followed by reduced consciousness. Pulmonary disease presents as fever, dyspnea and cough. Hemoptysis may occur if necrosis is present. Cutaneous disease manifests as cellulitis which progresses to dermal necrosis. Patients with skin disease may have had prior trauma or exposed to contaminated medical equipment. GI zygomycosis is observed in patients who are severely malnourished. This may occur throughout the GI tract but most commonly affects the stomach, ileum, and colon. Again, the presentation is nonspecific, with abdominal pain, distension, nausea, and vomiting. Hematochezia may occur.
Other disseminated forms may involve the kidneys, bones, and heart, with symptoms attributed to these organ systems. Treatment involves correcting the underlying abnormality and prompt institution of amphotericin B with surgical resection.
Pathogenesis: Species of Rhizopus are often associated with soil, plants and fruit. May species grow well at high temperatures; some are used in food fermentations, whereas others are pathogenic to humans and animals.
However, disease is unusual because of the low virulence of these organisms, which mainly affect individuals with immunocompromised conditions. The major route of infection is by inhalation of conida; other routes include ingestion and traumatic inoculation. When spores are deposited in the nasal turbinates, rhinocerebral disease develops; when spores are inhaled into the lungs, pulmonary disease develops. When the agents are introduced through the skin, cutaneous disease develops. Ingestion leads to GI disease, primarily among malnourished patients. Rhizopus spp. are molds in the environment and become hyphae in tissues. Once the spores begin to grow, fungal hyphae invade blood vessels, producing tissue infarction, necrosis, and thrombosis. Neutrophils are the key host defense against these fungi; thus, individuals with neutropenia or neutrophil dysfunction (diabetes, receiving immunosuppressants) are at highest risk. Cases among patients with AIDS are few, suggesting that the host defense against this infection is not primarily mediated by cellular immunity.
References:
1.
Imhof A, Balajee SA, Fredricks DN, Englund JA, Marr KA. Breakthrough fungal infections in stem cell transplant patients receiving voriconazole. Clin Infect Dis 2004; 39:743-6.
2.
Lee FY, Mossad SB, Adal KA. Pulmonary mucormycosis: the last 30 years. Arch Intern Med 1999;
159: 1301-9.
3.
Marty FM, Cosimi LA, Baden LR. Breakthrough zygomycosis after voriconazole treatment in recipients of hematopoietic stem-cell transplants. N Engl J Med 2004; 350:950-2.
4.
McPherson RA and Pincus, MR. Henry’s Clinical Diagnosis and Management by Laboratory
Methods, 25 th edition. Elsevier, Philadelphia, 2007.
5.
Ribes JA, Vanover-Sams CL, Baker DJ. Zygomycetes in human disease. Clin Microbiol Rev 2000;
13:236-301.
6.
Siwek GT, Dodgson KJ, de Magalhaes-Silverman M, et al. Invasive zygomycosis in hematopoietic stem-cell transplant patients receiving voriconazole prophylaxis. Clin Infect Dis 2004; 39:584-7.