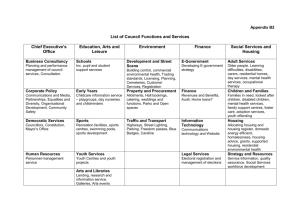
1) Subarachnoid Haemorrhage - Research & Education At Salford
advertisement

North West Clinical Neuroscience Partnership The Management of People with Subarachnoid Haemorrhage A North West Clinical Framework February 2001 Contents Section 1 Section 2 Section 3 Section 4 Introduction The North West Clinical Framework Members of the Working Group page 3 page 3 The Background Subarachnoid haemorrhage The rise of Interventional Radiology (Coiling) Practical issues page 4 page 5 page 6 The management of acute, non-traumatic aneurysmal, subarachnoid haemorrhage Presentation Diagnosis Transfer Investigation Treatment Experience, Outcome and Training Rehabilitation, Transfer and Discharge page 8 page 8 page 9 page 11 page 11 page 13 page 16 Conclusions North West Clinical Framework – Summary Implications for the North West page 18 page 21 Appendix 1 Neurovascular Workshops Summary Report Future management of Subarachnoid Haemorrhage, A Molyneux & RSC Kerr Changes in the management of intracranial aneurysms, RJ Nelson & EA Varian General Discussion ISAT (overhead slides). Dr A Molyneux page 23 page 24 page 25 page 28 page 29 page 31 Appendix 2 Working Group Report Neurovascular Services in the North West Region page 35 Appendix 3 North West Neurovascular Network page 45 Appendix 4 Proposed Minimum Standards for Accreditation page 46 Appendix 5 North West Clinical Neuroscience Partnership page 47 Appendix 6 Role of the Voluntary Agencies Group page 49 References page 50 2 Section 1 Introduction The North West Clinical Framework This guidance has been produced by the North West Clinical Neuroscience Partnership as a Framework for the commissioning and provision of care for people who have sustained a Subarachnoid haemorrhage. The North West Clinical Neuroscience Partnership is a partnership between the 3 Zonal Commissioners of Specialised Services in the North West, working on behalf of the Health Authorities and Primary Care Groups and Trusts in the North West, and the 3 Specialist Neuroscience Provider Trusts. The Partnership works in close cooperation with the North West Regional Specialised Commissioning Group and also includes representation from the Specialised Health Commissioning Service in Wales. The Partners have jointly agreed to develop a series of Clinical Frameworks and to work together to develop a Region-wide commissioning process which will ensure that the 7 million people of the North West and North Wales have access to a high quality Clinical Neuroscience service. The details of the Partners and the Partnership Agreement are included as Appendix 5. A group of Neurosurgeons and Neuroradiologists met between Spring 1999 and Autumn 2000 to produce the Framework and workshops were held in each of the 3 Specialist Provider Trusts at which external advice was a major feature. Following the development of draft guidelines wider consultation will be undertaken including voluntary agencies, staff of Acute General Hospitals, Primary and Community care. The Partnership believes that the implementation of this Framework will improve the care of people with subarachnoid haemorrhage giving better outcome and ensuring that the service remains up-to-date, accessible and appropriate. Members of the Working Group Dr W Gunawardena Neuro-radiologist, Preston Mr N Gurusinghe Neurosurgeon, Preston Mr J Holland Neurosurgeon, Manchester Dr R Laitt Neuro-radiologist, Manchester Dr T Nixon Neuro-radiologist, Liverpool Mr MDM Shaw Neurosurgeon, Liverpool Dr IR Williams Project Director The late Professor R Lye and Mr P Richardson attended some of the meetings as deputies making very helpful contributions. Mr K Lindsay, Dr A Molyneux and Mr R Nelson led the Trust workshops and provided much valuable advice. Comments from Dr CA Young and Dr A Luksza were helpful in drafting parts of the Framework. 3 Section 2 The Background Subarachnoid Haemorrhage Subarachnoid haemorrhage (SAH) is a relatively infrequent event. It is variously estimated to occur in 2–22.5 per 100,000 of the population each year in different countries (Ingall et al1). Excluding the extremes of China (2) and Northern Finland (22.5) most studies give figures between 6 and 11 per 100,000 per year (Bonita et al2,Cesarini et al3, Ingall et al4). There are no good local figures but assuming an incidence of 8-10 would give about 600 events per year in the North West of England and North Wales. In the majority of people with SAH the cause is found to be an aneurysm, a small balloon like swelling arising from an artery at the base of the brain. Possibly as many as 35% of these people have other causes, die immediately or are so badly damaged that no form of active treatment is considered (Cesarini et al3). After initial diagnosis at the local DGH, the remaining 65% are transferred to a Regional or Sub-regional Neurosurgery unit for further investigation and treatment. At the time of transfer less than 50% of patients who survived the initial bleed are free from neurological damage (Cesarini et al3). Since 1931 the treatment has been surgical with open operation leading to the placing of a metal clip across the neck of the aneurysm to prevent further bleeding. Initial figures for outcome were poor but there has been a marked improvement with the development of new surgical techniques and peri-operative care (Cesarini et al3). Even so, depending on the way outcome is measured, 8 to 32 % of people who have no persisting damage after the initial bleed will either die or be severely disabled. For people with persisting damage after the initial bleed the outcome is much worse (Cesarini et al3,Kassell et al5 , Roos6). One of the major causes of death and morbidity following SAH is the failure of normal control of blood flow to parts of the brain resulting in severe and often irreversible damage (ischaemia). The reason for this is not understood and efforts to find an effective prophylactic or therapeutic intervention have not so far been very successful. However trials of treatment with a drug (Nimodipine) which was thought to reduce ischaemic damage showed a convincing reduction in severe morbidity following SAH although the mechanism is far from clear. (Pickard et al 7). Modification of blood pressure, and circulation (Triple H therapy) in the setting of an Intensive Care Unit (ITU) have also been shown to reduce complications (Kassell et al8). The other major cause of mortality and morbidity is re-bleeding of the aneurysm. Such a re-bleed occurs in about 1% of people per day for the first 6 weeks after the initial bleed: for many patients the re-bleed is fatal or severely disabling. Early surgery has been advocated in an attempt to reduce this risk. Unfortunately other risks seem to balance the risk of rebleeding giving no over-all advantage in outcome for early surgery (Kassell et al9). 4 Until recently evidence suggested that the most appropriate management of people with no persisting neurological damage after aneurysmal SAH was to start Nimodipine as soon as the diagnosis had been established and to undertake surgery within 3 days. Post-operative care should be in an ITU where problems will be recognised and treated and Triple H therapy can be instituted as necessary. In spite of this at least 8% (Cesarini et al3) and possibly 32% (Roos6) of people have a poor outcome. More recent work shows that even this might be an under-estimate. Many people who had been classed as having a good outcome using the standard Glasgow Outcome Scale have quite considerable cognitive and psychological impairment which prevent a return to employment or pre-morbid levels of function (Buchanan et al10, Hackett et al11). For people with persisting damage after the initial bleed the outcome is even worse (Kassell et al5). Thus although SAH is not common it is a serious condition leading to death or severe disability in more than half those affected, and disabling many survivors to a greater or lesser extent. In these circumstances the search for alternative forms of treatment, or ways to reduce the mortality and morbidity associated with current treatment, is understandable and laudable. The Rise of Interventional Radiology (Coiling) Over the last 10 years Neuro-radiologists have been trying to find an effective and safe way of occluding aneurysms from within the circulation (Guglielmi et al12). The hope was that by avoiding the need for open operation the morbidity and mortality would be reduced. Techniques have gradually evolved and results have improved. The development of detachable platinum coils introduced through a catheter threaded up the arteries from the groin and positioned precisely in the aneurysm is seen as a real advance (Vinuela et al13). The technique appears to be at least as safe as surgery and to have comparable results in the short and medium term (Brilstra et al14, Byrne et al15, Lempert et al16). Long term results are not yet available. In some centres these coils are now seen as the first line treatment for SAH arising from aneurysms in some anatomical locations at the base of the brain. The final place of coiling in the treatment of intracranial aneurysms is not yet clear. A large MRC funded, international, multi-centre trial (ISAT) comparing open operation with coiling began in 1997 and is due to report in 2002. In the meantime, the studies referred to above have shown the technique to be at least as safe and effective as open operation for aneurysms in the posterior part of the circulation (Basilar aneurysms) and to have good short and medium term results in the treatment of people with anterior circulation aneurysms. This evidence has led to much wider use of the technique in some countries. For example in some centres in France about 80% of people with aneurysmal SAH are treated by coiling: in some UK cities the rate is almost as high (Nelson, personal communication. Management is currently extremely variable across the North West Region and differs considerably from some parts of the UK and Europe. Although the MRC trial will not report until 2002 it is already clear that coiling aneurysms does have a part to play in the management of aneurysmal sub-arachnoid haemorrhage. This is particularly true for older people, those whose neurological or general medical 5 condition is poor and in those situations where surgical access would be difficult or dangerous. Some patients in all parts of the Region will need rapid access to coiling, or at least to have it considered as one of the options available for their acute management. Although no large randomised trial has yet defined the role of coiling and clipping small trials have shown no over-all difference in outcome (Koivisto et al17). The study of Claiborne Johnston18 suggests that whatever the outcome of ISAT there will be a very considerable increase in the demand for coiling and that the time scale is likely to be short. In a review of the management of over 9500 people with ruptured aneurysms admitted to University Hospitals in the United States he concluded that patients treated in hospitals that used coiling were less likely to die in hospital and that the difference was not small. For every 10% of patients treated by coil embolisation there was a 9% reduction in in-hospital mortality. “An institution using coil embolization in 30% of cases would be expected to have 25% fewer in-hospital deaths compared with one never using the technique.” Whatever the reason such figures will be hard to ignore. It was the clear advice of external advisors in the Workshops that we should expect a rapid move to endovascular treatment (coiling) for 35-40% of patients even if the ISAT trial shows that surgery is safer in situations where it is possible. If the trial shows the 2 treatments to be equally effective and to have equal risk then in 2002 at least 70% of patients would be expected to opt for coiling and only 30% would require clipping. A result showing coiling to be as effective or more effective but safer would lead to more than 80% of people with SAH being treated by coiling. Thus we should not plan for less than 35% treated by coiling and could be faced with the need to raise this to 70 or 80%. The detail of the presentations is included as Appendix 1. The implications of such a change would be major and are considered in Appendix 2. In summary the North West does not have sufficient numbers of Radiologists with appropriate training and there is a shortage of trainees. Cooperative working between the Trusts will be necessary even when additional Radiologists have been appointed. This would have a consequential effect on Neurosurgery, with reduced numbers of operations and fewer surgeons involved in treatment of people with SAH, and thus on the provision of the whole service for people with SAH. Practical issues The need to deliver a high quality of care for people afflicted by uncommon conditions produces a tension between easy accessibility and the need to concentrate relatively scarce skills and resources on a few sites. In a Region with densely populated connurbations (Greater Manchester, Merseyside) and more sparsely populated rural areas (Furness, Snowdonia) these tensions could lead to the development of systems of care favouring one group at the expense of the other. Throughout the preparation of this Framework the views of voluntary agencies have been sought and they have been encouraged to seek wider views through their membership and through the regional alliances of neurological charities. In particular they considered the question of the balance between distance from a Centre and level of experience and expertise in defining the quality of a service. The recognition that neurosurgical or neuroradiological treatment for people who had 6 sustained a subarachnoid haemorrhage would involve travel to one or other of the Centres was coupled with 5 very clear statements: 1) Subarachnoid haemorrage has a profound effect on the lives of the individual and the family that is long lasting. Treatment at the Centre, important as it is, is frequently only a small part of the total picture. 2) Care at the Centre should be demonstrably of a high standard and of a nature that could not be provided more locally. 3) Adequate facilities, including travel to and accommodation at the Centre, should be available to relatives who should be kept fully informed at every stage. 4) Care should be continuous regardless of the number of locations in which that care is provided and the number of people involved. 5) Communication between professionals in the Centre and the home District should be prompt and sufficiently detailed to allow care and support to be provided whenever and wherever it is needed. In all that follows the working group has tried to keep these points to the fore. It is also important that they remain to the fore as this Framework is used to commission and provide the service. 7 Section 3 The Management of Acute, Non-traumatic Subarachnoid Haemorrhage 1) Presentation Non-traumatic SAH can occur at any age but in most series the average age is around 50. It is more frequent in women. The common presenting features are: Sudden, severe headache (“worst ever”) Photophobia Neck stiffness Reduced level of consciousness Neurological impairment. When all are present there is rarely any serious differential diagnosis, but meningitis, encephalitis, migraine and other forms of stroke can present in a similar way. If meningitis is suspected antibiotic treatment should be started while waiting for the results of the investigations. Sometimes, in spite of a very severe headache nothing abnormal is found. When subarachnoid haemorrhage is suspected the patient should be admitted to hospital immediately. 2) Diagnosis (Fig 1) Diagnosis is made on the basis of the clinical presentation and the presence of blood in the subarachnoid space. This can be demonstrated on a CT scan on the day of the bleed and is still present in 50% of patients 1 week later (Edlow and Caplan 19). The scan might also show blood in the ventricular system or within the brain itself as an intra-cerebral haematoma. If none of these radiological features is present blood or xanthochromia may be seen in the CSF when a lumbar puncture is performed (van der Wee et al 20). Lumbar puncture can be dangerous in the presence of an intracranial haematoma or hydrocephalus. It should not be performed before a CT scan has excluded this risk. Even in the presence of SAH the CSF can be normal for up to 12 hours after a bleed and will return to normal after about 14 days if there is no further bleeding (Edow and Caplan19). Spectrophotometry of the CSF is a more sensitive way of detecting xanthochromia than naked eye inspection and should always be used where there is no obvious xanthochromia in a patient with a story typical of SAH. In the presence of a typical history, a normal CT and LP can occasionally be misleading (Edlow and Caplan19). If the history is typical and both CT and naked eye examination of the CSF are normal spectrophotometry of the CSF specimen should be undertaken. If no alternative diagnosis is positively confirmed the diagnosis of SAH should be kept in mind and further advice sought from the Neuroscience Centre. 8 Because of the need to exclude infection (meningitis) and to establish appropriate treatment for SAH these tests should be completed within 6 hours of admission to the A&E department or hospital. Delays in diagnosis have been associated with a poorer outcome (Hutchinson et al21). However the absence of blood in the CSF less than 12 hours after the onset of symptoms does not exclude a bleed and it might be necessary to re-examine the CSF, after discussion with the Neuroscience Centre, if no alternative diagnosis has been reached. If there is doubt about the diagnosis or the CT scan cannot be interpreted the images should be transmitted to the Neuroscience Centre after discussion with the duty Neurosurgeon. Patients with suspected SAH should only be admitted to hospitals with 24 hour access to CT scanning and laboratory facilities and with image transfer links to the Neuroscience Centre. CT scanning should be performed in every patient suspected of having a SAH. If the CT does not confirm the diagnosis or give an alternative diagnosis CSF should be examined. CT and, if needed, LP should be completed within 6 hours of arrival at the hospital. Spectrophotometry of CSF should be available in the laboratory and should be used when there is no xanthochromia in a patient with a story typical of SAH. When the story strongly suggests SAH the Neuroscience Centre should be contacted if no diagnosis has been reached after CT and LP. 3) Transfer Following diagnosis the Regional Neurosurgical unit should be contacted and advice sought on further management. It is likely that nimodipine treatment will be recommended at this time and transfer to the unit arranged. If the patient is deemed too ill or unsuitable for transfer clear reasons should be given and a management plan discussed and agreed. This should include agreeing time intervals and indications for further contact. The situation should be explained to the patient in as much detail as is possible. Doctors in the DGH must have clear lines of communication with the Neuroscience Centre. On call staff in the Centre must be readily available to give advice to DGH staff and to discuss arrangements for transfer. It is the Centre’s responsibility to ensure that DGH staff are made aware of any need for interventions, investigations or treatment prior to transfer and to define escort requirements. DGH staff then have the responsibility for arranging transport and escorts as agreed and notifying the admitting ward of the expected time of arrival. Relatives should also have the situation clearly explained and be given every assistance in getting to the Centre. 9 Figure 1 Severe Headache Photophobia ? Neck Stiffness ? Neurological Deficit ? ? SAH Admit to Hospital with 24 hr CT scan & Laboratories CT No Blood Blood Mass Effect Contact Neuroscience Centre Contact Neuroscience Centre No Mass Effect CSF (Lumbar Puncture) Abnormal Contact Neuroscience Centre Normal Observe ? Repeat CSF Fails to Improve Or Diagnosis uncertain Contact Neuroscience Centre Improves Discharge 10 Each Neuroscience Centre should agree with each DGH a protocol for contacting the Centre, transferring images and transferring patients. This should identify a named person who has overall responsibility for the arrangements and give a single phone number as a contact point. Relatives should be given every assistance to enable them to travel to the Centre. 4) Investigation When the diagnosis has been confirmed the source of bleeding must be defined. In most patients this will be an aneurysm of one of the arteries around the Circle of Willis at the base of the brain. Arterio-venous malformations account for a small proportion of SAH and in some patients no source is found. Identification of the source depends on intra-arterial contrast angiography of the intra-cranial circulation. This procedure carries a 1.5-3% risk of death or stroke (Kassell et al9) and should only be performed by a Neuro-radiologist in a Neuroscience Centre. It is important that the Neurosurgeon responsible for over-all management of the patient and the Interventional Radiologist are available to discuss the images during the procedure so that all necessary views are obtained. Although many aneurysms can be seen on MR angiograms the detail is not yet good enough to allow technical treatment decisions to be made. In order to allow treatment to proceed angiography should be undertaken within as short a time scale as possible. If no aneurysm is demonstrated other investigations may be required depending on the clinical picture. As endovascular techniques develop it will be helpful to be able to discuss images with colleagues in other centres to seek their advice on the need for further investigation or choice of treatment. Angiography should only be carried out by experienced Neuroradiologists in a Neuro-radiology department. Outcome, complications and incidents should be recorded promptly and analysed every 6 months. All Neuro-radiologists undertaking Angiography should be part of a multi-centre benchmarking and audit programme. All Neuro-radiology departments investigating patients with SAH in the North West should have the ability to transfer images between Centres. 5) Treatment If an aneurysm is demonstrated treatment should be planned jointly by the patient, relatives, the Neurosurgeon, the Interventional Neuro-radiologist and the 11 Neuroanaesthetist. Treatment plans for the management of people with SAH should be developed and used as a basis for individual patient treatment plans generated after assessment and discussion. Nimodipine A large randomised clinical trial has shown that nimodipine reduces the number of people suffering permanent damage following aneurysmal SAH (Pickard et al7). Unless there are medical contra-indications (See BNF for upto-date list) all patients should receive nimodipine intravenously or orally from the time of diagnosis. Trials and Consent Truly informed consent to the performance of a procedure can only be given when information on risk and outcome of that procedure, in that setting are available. However, many questions about the nature and timing of treatment remain unanswered. This should be explained and where appropriate, patients should be informed of clinical trials and should be invited to be included. Patients and/or their representatives should be informed fully about the background to the trial, any known risks, and the nature of the procedures. Consent must always be a pre-requisite to inclusion in a trial. Choice of Treatment Surgical clipping and endovascular coiling should both be available for consideration in every patient. The choice of procedure will be made by the patient or their legally nominated representative and will take into account advice from the neurovascular team. This advice will depend on the site of the aneurysm, its size, the condition of the patient and the availability of facilities and skills. Consent to treatment should follow this process and should be recorded in the case notes, witnessed and signed, with a copy given to the patient or representative. Timing On the basis of large international trials the timing of surgery is not a critical factor in determining the over-all outcome: the advantages of earlier surgery are balanced by the increased risks (Kassell et al9). From the patient’s point of view and in the interest of maximising the use of resources, treatment should be undertaken as soon as the patient is fit enough to withstand the procedure. Anaesthesia Anaesthesia for neurosurgical or interventional neuroradiological procedures is a specialised task. All anaesthetics should be administered by, or closely supervised by an experienced neuro-anaesthetist. Patients should have an opportunity to meet the anaesthetist and ask questions prior to giving consent. The anaesthetist should be satisfied that the risks of anaesthesia are 12 understood and are acceptable in the context of background risks inherent in the natural history of SAH, and the anticipated outcome of treatment. The neurosurgeon and the anaesthetist should hold a joint discussion with the patient to reduce the potential for confusion. Post-operative Care With early surgery much of the delayed ischaemic damage associated with subarachnoid haemorrhage occurs in the post-operative period (Kassell et al9). It is important that patients are observed closely enough to detect changes as early as possible. Monitoring of treatment has to be thorough if further harm is to be avoided. (Cesarini et al3) It is unlikely that this detailed monitoring could occur outside an Intensive Care Unit dedicated to Neurological observation. Management of patients in a Neuro-intensive care unit is a shared responsibility. Nurses, anaesthetists/intensivists and surgeons/radiologists should meet regularly to exchange information and to plan the management of each patient. Throughout this process the presence of relatives is likely to be of support to the patient and is helpful to staff in ensuring that the situation is clearly understood and that decisions are fully informed. They too are often in need of support and information. Convenient facilities, including accommodation must be available for relatives. The treatment of patients with SAH should only take place in Centres where: Individual treatment plans (based on approved treatment plans for SAH) are generated for each patient and a copy given to the patient/relatives. Consent is fully informed and a witnessed and signed copy of the agreement is given to the patient and also filed in the case sheet. Nimodipine is used in at least 95% of patients in whom there is no contra-indication. Both Surgical (clipping) and Radiological (Coiling) treatment can be provided by specialists with adequate training and experience. Skilled Neuro-anaesthesia is available. Neuro-intensive care facilities exist with regular (at least twice daily) multidisciplinary discussions of the patients and constant availability of Neurosurgical and Critical Care consultants. 13 Appropriately trained nursing and PAM staff are available. Convenient accommodation and any necessary emotional/psychological support are available to relatives. 6) Experience, Outcome and Training There is no good published evidence linking outcome to volume of activity of an individual practitioner for either clipping or coiling aneurysms. It is clear that there is a learning curve for both procedures and that less experienced practitioners seek advice from more experienced colleagues. Over a 13 year period MauriceWilliams22 showed a 35% reduction in his 1 year over-all mortality and an 80% reduction in surgical mortality which he attributed to increasing operative experience. Evidence in some other procedures does show a clear link between volume of activity of an individual, or an institution, and outcome (Hannan et al23, Jollis et al24). Solomon et al25 have shown a convincing link between outcome and institutional volume of subarachnoid haemorrhage procedures at low levels of activity. Intriguingly, as noted earlier Claiborne Johnston18, reporting on the outcome of over 9,000 procedures for ruptured aneurysms has shown a fall of 9% in mortality for every 10% of patients treated by coil embolisation in an institution. An institution using coil embolisation in 30% of cases would expect to have 25% fewer in-hospital deaths compared with one never using the technique. He concludes: “… we found that outcomes of the surgical care improve dramatically as a larger proportion of cases are treated by endovascular techniques ……our results suggest that multidisciplinary, specialised, neurovascular services offering endovascular services are associated with reduced in-hospital mortality." Until this year the Society of British Neurological Surgeons advised that aneurysm surgery should only be undertaken by neurosurgeons who were responsible for at least 25 operations each year26. Although these figures are not repeated in the 2000 edition of “Safe Neurosurgery”27 there is a clear acknowledgement of the link between volume and outcome and of the changes in Neurovascular surgery. It is accepted that for some more senior surgeons their training and experience will stand them in good stead so that their skill will be maintained for some time with a lower volume of activity than that needed for a less experienced or less well-trained colleague. In the light of the rapid changes occurring in neurovascular surgery it seems reasonable to recommend that experience should be concentrated in the hands of as few people as remains consistent with the provision of an accessible, urgent service. Interventional Neuroradiologists have recommended a minimum of 80 cases per year in an institution undertaking training and that a trainee should have performed 40 procedures before accreditation. It would be wise to ensure that these figures are achieved within the Region in order to improve outcome, minimise risk and ensure adequate numbers of trained staff for the future. 14 Unless adequate audit is undertaken it will not be possible to assess outcome. With new procedures it is particularly important to generate information on outcome and risk and to be able to assess performance. It is therefore essential that all clinicians providing this service agree to keep adequate records and to collect and audit data on every patient and procedure. This information should be shared with colleagues in the other North West Centres and with Commissioners. Clinicians and Centres should participate in all relevant national audits. Outcome depends on the initial condition of the patient and on the investigative and therapeutic interventions of many individuals. For each to understand the impact of their contribution a regular review of outcome with Neurosurgeons, Neuroradiologists, Neuro-anaesthetists, Neuro-pathologists and Nursing and Therapy staff will be necessary. As the care of people with SAH becomes concentrated in the hands of fewer Neurosurgeons and Neuro-radiologists it will be necessary to ensure that trainees in both specialties have adequate exposure to the management of SAH. This will require a review of training programmes across the North West to include rotations to the Neurovascular service. During a period of change it is also important that all clinicians keep up to date and maintain their skills. The annual appraisal should focus on the use of continuing education and training, outcome, complaints and adverse incidents with a view to enabling clinicians to practise to the highest standard. All Neurosurgeons and Neuroradiologists providing treatment for people with SAH should be part of a North West Neurovascular Network which will set professional and service standards and monitor outcome. The network will also make recommendations to the North West Clinical Neuroscience Partnership for any necessary change to achieve or maintain these standards. Neurosurgeons would be expected to be responsible for at least 20 operations each year and Neuroradiologists for 40 procedures each year. The figures should be averaged over the previous 3 years. It is expected that the Network will oversee a gradual change towards concentration of the work in the hands of fewer surgeons who will work closely with Interventional Neuro-radiologists as members of a neurovascular team providing a service across the North West. All members of the Network will be required to show through regular audit and appraisal that they remain up to date and competent in their knowledge and practice. Multi-disciplinary reviews of outcome will take place at 3 month intervals. These reviews should include: Neurosurgeons, Neuroradiologists, Neuro-anaesthetists, Neuro-pathologists, Nursing and Therapy staff from all Centres. 15 Centres and individual clinicians should participate in all relevant national audits. Trainees in Neurosurgery and Interventional Neuro-radiology in the North West should only receive their training in the management of Neuro-vascular disease from members of the Network. 7) Rehabilitation, Transfer and Discharge Up to 1/2 of those patients admitted to hospital with a SAH who survive will have significant persisting disability (Hackett et al11). If no intervention is planned management will depend on the condition of the patient but at the outset measures should be taken to minimise subsequent disability through care to skin, bladder, bowels mouth and limbs. Within 72 hours of admission or the completion of any intervention, an assessment of the likely need for formal rehabilitation should be made. Rehabilitation is not just a set of activities but incorporates an approach to all interactions between the patient, staff of all disciplines and relatives. This requires planning followed by action agreed and carried out by all members of the team. (Playford et al28, Hitchcock et al29). Formal therapy interventions should be coordinated as part of this process with clear goal setting. A regular review of progress against these goals should be used to plan continuing treatment. Many patients return to the DGH nearest to their home for continuing rehabilitation after the acute phase of the illness. If the needs are complex it may be necessary to consider a period in a Neuro-Rehabilitation Unit. Transfer must be a smooth process with agreement between Centre and DGH or Neuro-Rehabilitation Unit facilitating the transition. Prior to transfer or discharge from the Centre there should be a formal assessment of the functional ability of the patient and plans for any continuation of therapy should be agreed with those providing treatment and the patient and family. A copy of the assessment and any plan for continuing treatment should be filed in the notes, given to the patient and forwarded to the new team. Copies should also be sent to the GP and the referring physician and neurosurgeon. Some people will have continuing needs when they are to be discharged back into the community. It is particularly important that community services and the family are included in the discharge planning and that arrangements are acceptable to all. As with transfer to the DGH copies of the assessment and agreed plans should be given to the patient and forwarded to the new team. Advice on all aspects of personal, social and family life and employment may be needed and should be considered. Staff working in the community or DGH are likely to have more contact with the family and social services but to have less experience of managing patients with SAH than Centre staff. Advice on a range of issues can improve care and reduce anxiety. A clear statement from the Centre that requests for help and advice would be welcome, accompanied by a reliable telephone contact number, could be extremely helpful. 16 All Centres treating patients with SAH should have a multidisciplinary Neuro-rehabilitation team. Patients should have their rehabilitation needs assessed within 72 hours of admission or completion of a treatment procedure and a treatment plan should be agreed. Patients, family and staff should all have written copies of the agreed treatment plan. Whenever a transfer of care occurs treatment should be discussed and agreed by both teams and the patient and family. Written accounts of treatment undertaken and treatment planned, together with copies of the agreements should accompany the patient at the time of transfer or discharge. All doctors involved in care should be kept informed. The GP and referring consultant should receive letters at the time of admission and discharge. All letters should be dispatched within 72 hours of admission or discharge. The death of a patient should be notified to the GP and the referring clinician within 24 hours. Continuing support should be offered from the Centre to DGH and Community staff. Written documentation from the Centre should give clear details of reliable and rapid access to easily available support and advice. 17 Section 4 Conclusions A North West Clinical Framework - Summary Primary Care When subarachnoid haemorrhage is suspected the patient should be admitted to hospital immediately. Secondary Care/DGH Patients with suspected SAH should only be admitted to hospitals with 24 hour access to CT scanning and laboratory facilities and with image transfer links to the Neuroscience Centre. CT scanning should be performed in every patient suspected of having a SAH. If the CT does not confirm the diagnosis or give an alternative diagnosis CSF should be examined. CT and, if needed, LP should be completed within 6 hours of arrival at the hospital. Spectrophotometry of CSF should be available in the laboratory and should be used when there is no xanthochromia in a patient with a story typical of SAH. When the story strongly suggests SAH the Neuroscience Centre should be contacted if no diagnosis has been reached after CT and LP. Each Neuroscience Centre should agree with each DGH a protocol for contacting the Centre, transferring images and transferring patients. This should identify a named person who has overall responsibility for the arrangements and give a single phone number as a contact point. Relatives should be given every assistance to enable them to travel to the Centre Angiography Angiography should only be carried out by experienced Neuroradiologists in a Neuro-radiology department. Outcome, complications and incidents should be recorded promptly and analysed every 6 months. All Neuro-radiologists undertaking Angiography should be part of a multi-centre benchmarking and audit programme. All Neuro-radiology departments investigating patients with SAH in the North West should have the ability to transfer images between centres. 18 The Neuroscience Centre The treatment of patients with SAH should only take place in Centres where: Individual treatment plans (based on approved treatment plans for SAH) are generated for each patient and a copy given to the patient/relatives. Consent is fully informed and a witnessed and signed copy of the agreement is given to the patient and also filed in the case sheet. Nimodipine is used in at least 95% of patients in whom there is no contra-indication. Both Surgical (clipping) and Radiological (Coiling) treatment can be provided by specialists with adequate training and experience. Skilled Neuro-anaesthesia is available. Neuro-intensive care facilities exist with regular (at least twice daily) multidisciplinary discussions of the patients and constant availability of Neurosurgical and Critical Care consultants. Appropriately trained nursing and PAM staff are available. Convenient accommodation and any necessary emotional/psychological support are available to relatives. The Neurovascular Network All Neurosurgeons and Neuroradiologists providing treatment for people with SAH should be part of a North West Neurovascular Network which will set professional and service standards and monitor outcome. The network will also make recommendations to the North West Clinical Neuroscience Partnership for any necessary change to achieve or maintain these standards. Neurosurgeons would be expected to be responsible for at least 20 operations each year and Neuroradiologists for 40 procedures each year. The figures should be averaged over the previous 3 years. It is expected that the Network will oversee a gradual change towards concentration of the work in the hands of fewer surgeons who will work closely with Interventional Neuro-radiologists as members of a neurovascular team providing a service across the North West. 19 All members of the Network will be required to show through regular audit and appraisal that they remain up to date and competent in their knowledge and practice. Multi-disciplinary reviews of outcome will take place at 3 month intervals. These reviews should include: Neurosurgeons, Neuroradiologists, Neuro-anaesthetists, Neuro-pathologists, Nursing and Therapy staff from all Centres. Centres and individual clinicians should participate in all relevant national audits. Trainees in Neurosurgery and Interventional Neuro-radiology in the North West should only receive their training in the management of Neuro-vascular disease from members of the Network. Rehabilitation All Centres treating patients with SAH should have a multidisciplinary Neuro-rehabilitation team. Patients should have their rehabilitation needs assessed within 72 hours of admission or completion of a treatment procedure and a treatment plan should be agreed. Patients, family and staff should all have written copies of the agreed treatment plan. Whenever a transfer of care occurs treatment should be discussed and agreed by both teams and the patient and family. Written accounts of treatment undertaken and treatment planned, together with copies of the agreements should accompany the patient at the time of transfer or discharge. Communication All doctors involved in care should be kept informed. The GP and referring consultant should receive letters at the time of admission and discharge. All letters should be dispatched within 72 hours of admission or discharge. The death of a patient should be notified to the GP and the referring clinician within 24 hours. Continuing support should be offered from the Centre to DGH and Community staff. Written documentation from the Centre should give clear details of reliable and rapid access to easily available support and advice. 20 B Implications for the North West 1) The working group believes that the evidence for the effectiveness of interventional radiology in the management of aneurysmal subarachnoid haemorrhage is persuasive. By the end of 2001 we expect a minimum of 4050% of patients will be treated by neuroradiologists with a real possibility that the figure should be higher. Change is likely to be rapid. 2) To meet this demand there will need to be a minimum of 4 Interventional Neuroradiologists in the North West. The Working Group suggests 2 in Manchester/Salford, and 2 in Liverpool and an additional appointment in Preston by 2002. 3) It will not be possible for the present arrangement of admitting people with subarachnoid haemorrhage to each of the three centres to continue unless working practices change and new appointments are made as soon as possible and not later than June 2002. 4) The working group considers that there is no perfect solution which guarantees ready access to specialist treatment close to people’s homes every day of the year. In making its recommendations the group has taken the view that the outcome of treatment is the most important measure and that while distance travelled is extremely important, it must take second place. 5) In order to plan and implement changes in working practice, recruitment and training of staff and practical arrangements for service provision a Neurovascular Network should be established and a lead clinician appointed by the end of January 2001. 6) Initially the network should include all neurosurgeons and neuroradiologists who treat people with aneurysmal subarachnoid haemorrhage. By 2002 it is anticipated that this work will be undertaken by a maximum of 3 neurosurgeons in Manchester/Salford, 3 in Liverpool and 2 in Preston. Membership of the network will be dependent upon agreement to regular audit and review of practice and outcome. 7) The working group believes that all Neurovascular (neurosurgery and neuroradiology) activity in Manchester/Salford should be united on the Hope Hospital site as soon as is possible and in any event not later than the transfer of services from Central Manchester in May 2001. 8) The working group recommends that serious thought be given to concentration of some of this work as the number of patients undergoing open surgery for clipping decreases. By October 2002 a decision should be taken, in the light of 18 months experience in the Network, on the number of Neurovascular Centres which can be sustained in the North West. 9) In the interim period it may not be possible to cover each site with skilled personnel every day. Agreements will have to be reached between clinicians and with Trusts and Health Authorities to ensure services of the expected 21 quality are available to all patients regardless of their address. The working group recommends that members of the Network should have a contract/appointment in each of the 3/4 Trusts allowing flexibility to work in more than one site should this ever be necessary. 10) By January 2002 it should be clear whether the number of patients undergoing interventional radiology is likely to be greater than 50%. If this figure is likely to be exceeded a further interventional radiologist will be required and an appointment should be made. Given the shortage of trainees the working group recommends that funding for a 1year fellowship in interventional radiology should be sought in time for appointment early in 2001. 11) Implementation of these changes will require much better information than is currently available. Urgent action is required to develop an information strategy which will provide information to and for the network early in 2001. 12) The Network will need to address training issues early. Neurosurgical and neuro-radiological training programmes will require considerable modification. 22 Appendix 1 Neurovascular Workshops August 16th 2000 Hope Hospital, Salford Preston Royal Hospital Walton Centre for Neurology & Neurosurgery Dr Andy Molyneux Neuro-radiologist,Oxford Mr Rick Nelson Neurosurgeon, Bristol Mr Ken Lindsay Neurosurgeon, Glasgow 23 North West Clinical Neuroscience Partnership The Management of People with Subarachnoid Haemorrhage Trust Workshops August 16th, 2000 Summary Report The Project As part of the North West Clinical Neuroscience Project a group of Neurosurgeons and Neuroradiologists had been meeting for about a year to consider the implications of changes in the management of people with subarachnoid haemorrhage (SAH). They had reached a number of tentative conclusions which would have far reaching implications for the provision of a service in the North West and North Wales. In order to encourage further discussion of the options they had constructed a case study to be considered at workshops in each Trust along with the additional perspective of 3 nationally respected specialists from outside the Region. The Workshops The workshops were open to all clinicians, executive and non-executive directors in the 3 Trusts providing In-patient clinical Neuroscience services in the North West. At each of the 3 Trusts there was representation from all the groups and attendance was good, especially for mid August. At each venue the same programme was followed: Local Welcome Trust Chair or Chief executive Coiling and the implications of ISAT Dr AJ Molyneux Implications for Neurosurgery Mr R Nelson Case Study/Discussion Mr K Lindsay A summary of the presentations follows: 24 Future Management of Subarachnoid Haemorrhage. Implications of the International Subarachnoid Aneurysm Trial for patterns of care resources and manpower. AJ Molyneux & RSC Kerr Background Subarachnoid haemorrhage occurs with an incidence of approximately 10 – 15 per 100,000 per year and usually affects a middle-aged population – mean age approximately 53 years. The major cause for subarachnoid haemorrhage is rupture of an intracranial aneurysm. Those patients surviving in good clinical condition have a significant risk of re-bleeding in the first month following the procedure, about 20% at one month declining over subsequent months. Re-bleeding is associated with a 60% mortality. Early treatment of the aneurysm by either surgical clipping or endovascular coil techniques is effective in preventing this re-bleed and thus reducing the mortality associated with it. Up until eight years ago, no alternative, other than surgical clipping was available for the treatment of intracranial aneurysms. However now endovascular coil treatment with detachable platinum coils is used in a proportion of patients, in particular, those patients who present high surgical risk because of poor clinical condition, older patients who tolerate craniotomy less well, and patients with aneurysms of the posterior circulation, particularly those arising from the basilar artery. In the U.K these patients are currently treated in most neurosurgical centres by endovascular coil techniques rather than surgical clipping. This group amounts to approximately 30% of patients. The International Subarachnoid Aneurysm Trial – ISAT, is a multi-centre Medical Research Council, French Health Ministry and Canadian MRC funded trial testing which method, surgery or coiling, results in better outcomes for patients, the primary endpoint is death or disability at one year. This trial commenced in 1997 as a full-scale study funded by the Medical Research Council and has been re-funded to complete recruitment, which is planned to continue until mid 2002. The trial is likely to report at the end of 2002 or early 2003 on the primary endpoints. The trial currently has enrolled almost 1500 patients with a target of 2500. The progress has been reviewed by the Data Monitoring Committee and they judged that if the trial recruited of the order of 2500 patients, then the trial will “affect the management of subarachnoid haemorrhage for many years to come”. The trial is powered to detect a 25% difference in outcomes between the treatments at a 1% significance level. 25 Currently in the U.K endovascular coil treatment is provided in most Neurosurgical centres although there are some smaller centres where there is no availability for coil treatment of aneurysms. Most centres only have one or at most two neuroradiologists capable of performing the procedure, which is performed in a Digital angiography suite under general anaesthesia. There is a very limited number of endovascular-trained operators in the U.K Interventional Neuroradiologists. Furthermore there are only a few centres that can offer any kind of training in this field and in none of these centres are posts funded specifically for training in this field. Potential Impact of the Trial on Clinical Practice There are three possible outcome scenarios following the reporting of the ISAT Trial. The scenarios are as follows. Scenario 1 Surgical treatment of the patient group enrolled in the trial, namely anterior circulation aneurysms in good clinical condition (grade 1 and 2), is shown to be safer. This outcome would suggest that approximately 70% of patients will be treated by surgery and about 30% by coiling. Currently in the U.K approximately 40% of patients are treated by coiling. This proportion is rising towards 50% (data from ISAT Trial Ascertainment Logs). This scenario will mean that whilst all Neurosurgical Centres should be able to offer coil treatment to patients, the switch to coiling and the volumes will stabilise or reduce slightly from the current proportion (about 40-45% in the UK). Scenario 2 No significant difference between coiling and surgical clipping is shown on the primary endpoint or a modest trend is seen. However secondary endpoints show coil treatment superior, on endpoints such as length of stay, intensive care usage, overall cost of care, duration off work, and neuropsychological effects. In this scenario it would be anticipated that there would a progressive shift to coil treatment bearing in mind that it does not involve craniotomy. The secondary endpoints, such as length of time in Intensive Care and overall hospital usage and costs (based on large scale US surveys 1, 2. ), are more likely to be favourable to coiling. In these circumstances it is likely there will be a progressive trend to coiling – reaching between 70% and 80% of patients. Scenario 3 Coiling is shown to be safer than surgery with significantly reduced death and disability at 1 year. The trial is powered to detect this at a high significance level (5% or 1%) If the trial shows significance level in this range or above for coiling then it will be very difficult to offer surgical treatment to patients whose aneurysms are suitable for 26 endovascular coil occlusion immediately this finding becomes available to the clinical community in the medical literature. This would immediately result in centres, which were unable to offer coil treatment of cerebral aneurysms, for whatever reason, no longer being able to treat patients with aneurysmal subarachnoid haemorrhage, in that they would not be in a position to offer the treatment proven to be significantly safer. Those centres which can offer such treatment will still be limited by the availability of skilled manpower and, in some cases, infra structure resources, such as nursing, anaesthetic time in radiology and suitable digital angiographic room availability. Whilst it is not possible to pre-judge the outcome of the trial and the data is blinded to the Investigators and only available to the Data Monitoring Committee, much of the clinical community believe the scenarios 2 and 3 are much more likely than scenario 1. This is based not only on anecdotal data but and large-scale observational evidence from the US University hospitals database published recently 1,2. However whatever the outcome it is vital that early contingency planning is made to deal particularly with the manpower availability and resource issues. It is probably too late to impact immediately the potential manpower situation, however, the lack of proper training availability and funded posts for interventional neuroradiology training remains a major issue which we believe should be given immediate consideration if we are to be able to offer any kind of level of service to patients of what then may be a technique proven at a high significance level. This is the first time to our knowledge that a new minimally invasive procedure has been subjected to such a rigorous evaluation in comparison with an existing surgical technique, however unlike a proven drug therapy, this technique requires highly skilled manpower and appropriate infrastructure resources to deliver the treatment. The consequences of scenario’s 2 & 3 need urgent consideration. References: 1. Johnston SC Effect of Endovascular services on cerebral aneurysm treatment outcomes. Stroke 2000 Jan 31(1) 111-117 2. Johnston SC Dudley RA Gress DR and Ono L Surgical and endovascular treatment of unruptured cerebral aneurysms at university hospitals. Neurology 1999 Jun 10:52(9) 1799- 1805 27 Changes in the management of intracranial aneurysms: implications for future service and training requirements in the UK RJ Nelson & EA Varian Frenchay Hospital, Bristol A summary of the presentation given by Mr RJ Nelson at workshops in the North West Region on August 16th ,2000. 1) The number of procedures for subarachnoid haemorrhage has increased from 35/million population to 45/million in England and Wales between 1992 and 1998. The rate is 25% higher in Scotland. 2) The number of endovascular coiling procedures has increased from 0 in 1992 to 600 (OPCS figures) or 800 (manufacturer’s figures) in 1998. 3) Expect approximately 50:50 clipping:coiling by 2003 4) With no sub-specialisation in Neurosurgery If 30% coils = 12/13 clips/surgeon/year If 50% coils = 8/9 clips/surgeon/year If 70% coils = 5/6 clips/surgeon/year With sub-specialisation in Neurosurgery If 30% coils = 20/27 clips/surgeon/year If 50% coils = 15/19 clips/surgeon/year If 70% coils = 10/12 clips/surgeon/year 5) Units with combined surgical and endovascular teams will have to consider sub-specialisation 6) Inter-unit cross cover may be required 7) Audit must specify severity 8) In future surgery will be more complex than at present 9) Training will be longer and may be more difficult to arrange 10) Cost is approximately £1million/10% patients coiled 28 General Discussion Although there were differences in emphasis, discussions in the 3 Centres came to broadly similar conclusions: 1) The situation is changing quickly and the service will need to change to reflect this. 2) Coiling (Neuroradiological intervention) is already seen to be the standard form of treatment for aneurysms in the posterior circulation, for people in poorer condition and probably for older people. This is about 30% of people with SAH. 3) The ISAT trial should give guidance on the treatment of younger, fitter people with anterior circulation aneurysms. 4) It is likely that the demand for coiling will increase and it would be unwise to plan for less than 50% within the next 2 years. Many thought 70% would be more realistic. 5) There were not enough Interventional Neuroradiologists in the North West to manage this workload. More should be recruited. 6) Even if more radiologists were to be recruited, cooperation between the Centres would be necessary to ensure cover during holidays and to provide an on-call rota. 7) The number of procedures for SAH was increasing. However, the number of aneurysms treated by surgical clipping was likely to fall. In Bristol this had been very marked. 8) Unless sub-specialisation occurred the number of Clipping procedures per Neurosurgeon would become too small to maintain or gain experience. Each Centre identified the need to reduce the number of neurosurgeons involved in the Neurovascular service. (Preston 2, Manchester and Liverpool 3–4). 9) Cooperation between Centres would be required to ensure the availability of a vascular neurosurgeon every day. 10) The management of people with SAH required a dedicated team. This raised the question of whether the team or the patient should travel when cover was provided for more than one Centre. In general it was agreed that it should be the patient that travelled in order to give continuity of care in the postoperative period. 11) Any restructuring of the service to allow sub-specialisation for SAH would have implications for the rest of Neurosurgery and Neuroradiology. 29 12) There was discussion on the number of Centres to be involved in Neurovascular work. Although there were strong arguments in favour of a 2 Centre solution it was recognised that this was not acceptable in the context of the overall service and the current expectations of future developments. 13) There would also be a profound effect on the training programme. 14) A reduction in the surgical treatment of aneurysms changed the Neurosurgeon’s perception of the specialty and would thus be a very difficult issue for them to address. In assessing trainees the number of aneurysms they had clipped had been a very important indicator of their overall experience. 15) In all 3 Centres neurosurgeons felt it unlikely that they would practise only 1 sub-specialty. They also felt that from time to time it would be appropriate for colleagues to undertake emergency procedures in a sub-specialty other than their own. 16) The population of the North West and North Wales was seen as being large enough to sustain a good service, a good training programme and good research. 17) Coils are expensive; their use has not so far led to reductions in other expenditure. The introduction of coiling will thus have considerable cost implications. Conclusions The need for change was acknowledged and many of the consequences of this were identified. The working group undertook to complete their deliberations and to bring a set of recommendations to commissioners and clinicians. These would be presented at an open meeting to which clinicians from all 3 Trusts and interested managers, executive and non-executive directors of Trusts and Health Authorities would be invited. A date for the meeting would be identified on completion of the report. 30 ISAT Primary Objective To determine whether an Endovascular treatment policy compared with a Neurosurgical treatment policy, reduces the proportion of patients with a moderate or poor outcome (Rankin 3 or worse) by 25% at one year. Tertiary Objectives ISAT - Secondary Objectives. To determine whether Endovascular coil treatment is: •As effective as surgery at preventing re-bleeding •Results in a better quality of life •Is more cost effective •Improves Neuropsychological outcome International Subarachnoid Aneurysm Trial Summary of Ascertainment Log data July 2000 Criteria for entry to Ascertainment log: • To examine the long term outcome over 5 years with specific reference to re-bleed rates. • To determine the long term significance of angiographic results. 1. Proven SAH (C.T. or L.P.) 2. Proven aneurysm on angiography 3. Primary referral to Neurosurgical unit for SAH management July 2000 31 International Subarachnoid Aneurysm Trial Summary of Ascertainment Log data July 2000 International Subarachnoid Aneurysm Trial Summary of Ascertainment Log data Oxford Total SAH (40 months) Number Randomised: Reasons for Non randomisation Anatomy unsuitable: 74 Location of aneurysm Grade or Age of Pt. Refusal of consent: Not Stated Non availability of Radiol 401 145 % (18) 28 60 49 12 20 Total recorded SAH participating centres N = 5372 Number Randomised 1228 (23%) (7) (15) (12) Total treated surgical 2625 579 ISAT Total treated by coiling 2113 628 ISAT (5) No procedure 223 February 1998 International Subarachnoid Aneurysm Trial Summary of Ascertainment Log data Reasons for non randomisation July 2000 International Subarachnoid Aneurysm Trial Summary of Ascertainment Log data Reasons for non randomisation July 2000 Neurosurgical patients Anatomy Location Refusal of consent Grade or Age Not stated/Incomplete Total 2046 1121 (20%) 58 191 156 376 International Subarachnoid Aneurysm Trial Ascertainment Log Oxford Patients Final Treatment Total in Log Randomised Rx Surgically Rx Endovascularly Non Randomised Rx Surgically Rx Endovascularly Conservative Data Missing Total in Logs 5372 Sx 2625 Coiled Endovascular coiled patients N/R 1485 Location 353 Anatomy 411 Grade or age 360 Refusal of consent 200 Summary of Ascertainment Log data Oxford Grade at angio and treatment Grade 1 389 Grade 2 113 Grade 3 35 638 patients 206 (32%) 98 108 432 150 235 20 27 (34%) (54%) ( 3%) ( 4%) 2113 Grade 4, 5 or Ventilated Total 76 Sx Sx Coil Sx Coil Sx Coil 23 Coil 164 207 40 65 11 20 (30) 39 (42) (53) (35) (57) (31) (57) (51) 638 July 2000 Summary of Ascertainment Log data Clinical Grade at angio and Final treatment All centres Grade 1 & 2 3856 Grade 470 3 4,5 & Vent Total 683 Sx Coil Cons Sx Coils Cons Sx 5009 375 Coil Cons Sx Coil 2020 (52) 1522 (39) 126 210 (45) 185 (39) 31 Grade (54) 324 (47) 59 (8) 52% 41% International Subarachnoid Aneurysm Trial Statistical Report # 6 Patient Baseline Assessment Prepared July 2000 WFNS Grade at time of Randomisation Grade Total 1 2 3 4 5 6 (Not assessable) 13 757 (63 %) 293 (24 %) 86 (7 %) 44 (4 %) 9 (1 %) (1 %) 1202 32 International Subarachnoid Aneurysm Trial Statistical Report # 6 Patient Baseline Assessment Prepared July 2000 International Subarachnoid Aneurysm Trial Data Monitoring and Ethics Number of Aneurysms detected at randomisation (N = 1202) No Count Proportion 1 945 79 % 2 201 17 % 3 39 3% 4> 17 2% Size of Aneurysms < 6 mm 630 52 % 6 -10 473 39 % >10 99 All Centres require local ethics (IRB) approval UK requires Multicentre ethics approval and local ethics approval Independent data monitoring and ethics committee appointed by Medical Research Council to report to Trial Steering Committee 8% International Subarachnoid Aneurysm Trial Data Monitoring and Ethics Committee Report to Trial Steering Committee March 2000 International Subarachnoid Aneurysm Trial Data Monitoring and Ethics Committee Report of meeting: March 2000 Reviewed unblinded randomisation data on 1202 patients Reviewed 2 month outcome data on 1056 patients by treatment allocation Reviewed 1 year data on 700 patients Reviewed information on re-bleeding International Subarachnoid Aneurysm Trial Data Monitoring and Ethics Committee Report to Trial Steering Committee March 2000 “In the light of the the evidence we have seen with 2500 patients the trial we believe will usefully influence clinical Practice for many years to come without the endless controversy that we are currently witnessing. We urge all collaborators to hasten efforts to recruit the remaining 1100 or so patients” . “The quicker we recruit, the quicker results will become available and the sooner future patients will be managed in the light of the best and most robust evidence” Future management of subarachnoid haemorrhage Post ISAT scenario’s Trial shows surgery safer Trial shows equivalence on Primary endpoints but coiling better on secondary end points Trial shows coiling significantly safer P 0.05 Trial shows coiling much safer P 0.01 Future management of subarachnoid haemorrhage Post ISAT scenario’s Future management of subarachnoid haemorrhage Post ISAT scenario’s Surgery safer for Grade 1 & 2 patients with anterior circulation aneurysms Posterior circulation aneurysm poor grade patients and older patients will continue to be coiled Probable proportion coiled 35 - 40% Noted that the recruitment rate was holding up but below original target This trial was likely to be the only large scale randomised evidence of the comparison between clipping and coiling Having reviewed the outcome data, it was our unanimous opinion that there was no ethical or any other reason to stop recruitment. The trial procedures and data collection are all exemplary It is vital to maintain and increase recruitment and keep all patients on secure follow up Surgery and coiling equal or trend to safer endovascular but coiling better on secondary end points such as: Higher return to work rates Shorter lengths of stay and ICU time, less overall cost of care and/or societal costs Less rapid but significant shift to coiling of about 70 - 80% of aneurysms 33 Future management of subarachnoid haemorrhage Post ISAT scenario’s Coiling shown to be safer by margin of P0.05 to P0.01 or > No longer reasonable practice to offer surgery if coiling technically possible Immediate shift of management to coiling Centres not able to offer coiling may no longer be able manage SAH 80% + of aneurysms should be coiled Immediate consequences for neurosurgery & neuroradiology Future management of subarachnoid haemorrhage Impact of ISAT Future management of subarachnoid haemorrhage Post ISAT scenario’s Timing of coil treatment does not affect outcome (Recent Oxford data accepted for publication in Neurosurgery) Grade 4 & 5 patients with clot benefit from early coiling and clot evacuation Need weekend and out of hours availability of service for optimum care Need surgeon, interventionist and nursing support Recent grant approval by MRC to extend recruitment and continue follow up for 5 years Recruitment of further 1000 patients likely by June or July 2002 (currently 1419) Report Primary outcome on basis of 2 month data on all patients and 1 year on rest Timing of Primary outcome publication Lancet probably late 2002 Rest of detailed data to follow 2003 - 2004 34 Appendix 2 Neurovascular Services in the North West Region: The effect of Developments in Interventional Neuroradiology Report of the Neurovascular Working Group of the North West Clinical Neuroscience Project August 2000 35 Introduction and Background Interventional Neuroradiology is now more than 20 years old during which time it has expanded enormously. The formation of the World Federation of Interventional and Therapeutic Neuroradiology (WFITN), the Interventional Neuroradiology Committee of The European Society of Neuroradiology (ESNR), the United Kingdom Neurointerventional Group (UKNG) and the publication of The International Journal of Interventional Neuroradiology reflect this expansion and its world-wide impact. Despite this many Neuroscience centres remain unaware of what can be achieved and what should be expected in the practice of interventional neuroradiology. Expansion of workload is particularly evident in the treatment of intracranial aneurysms where Guglielmi Detachable Coils (GDC) are, in some centres, considered the first line treatment for aneurysms in certain anatomical locations (Lempert et al16). Other vascular lesions are increasingly being treated using the endovascular route either as a definitive treatment or in combination with Neurosurgery or stereotactic radiosurgery. These developments are leading to increasing numbers of cases being treated in the North West Region (NWR). However, other Regions in the UK, serving similar populations, have considerably higher levels of activity (Molyneux30). Patients are treated on an inpatient basis with Neurosurgical support. Many of these patients cases would previously have been treated by open Neurosurgical procedures. Some patients would previously have been managed conservatively, particularly older patients with complex lesions. The workload thus includes both new work and work transferred from neurosurgery. The change in practice has brought Interventional Neuroradiologists to the forefront in the management of Neurovascular disease. This management is performed in close collaboration with the admitting Neurosurgeon, Neuroanaesthetists, and where possible with Neurologists who have an interest in Neurovascular disease. For some lesions cooperation with Stereotactic Radiotherapists is also required. The concept of such Neurovascular teams is new but they must be developed to ensure that patients are given the most appropriate therapy and the best outcome (Claiborne Johnston et al18). The organisation of clinical neuroscience services and endovascular intervention services within the Northwest are thus interdependent. There are currently four centres in the Region where there is potential for provision of these services: Central Manchester Healthcare Trust (CMHCT), Hope Hospital Salford, The Walton Centre, Liverpool and Royal Preston Hospital. Currently there is endovascular intervention on the first three sites but not at Preston. The reorganisation of Neurosciences in Greater Manchester will result in single centre on the Hope site with transfer of CMHCT in April 2001. There will therefore be three centres serving the Neuroscience requirements of patients in the NWR and North Wales including endovascular intervention. These centres must have the capacity to accommodate predicted inevitable service expansion. This document outlines the current levels of interventional neuroradiology activity in the NWR and makes predictions of the likely expansion of the workload in the 36 intermediate and long term. This prediction is important in developing a strategic plan for the management of neurovascular disease across the three sites. The importance of developing a sensible strategic plan cannot be over emphasised. Collaborative development across the region will be essential to provide quality, continuity of service and on call cover and the opportunity must not be missed. Current Regional Workload The activity in adult endovascular neurointervention is increasing. Almost all procedures are performed in the Neuroradiology department under GA requiring a Consultant Anaesthetist, ODA and nursing support. Only modern angiography equipment will have the resolution to allow coiling to be performed. CENTRE 1995-6 1996-7 1997-8 1998-9 CMHCT 21 21 48 55 Hope 17 24 26 54 North Manchester 25 26 23 0 25* 25* 30 41* 40** Walton Centre Preston TOTALS 63 96 163 179 TABLE 1 - Interventional activity by centre for the years 1995-99 (no. of cases) *cases referred out of Region **cases referred to CMHCT The figures in Table 1 are not complete but it is clear that endovascular intervention is expanding year on year. Some of this increase is due to the International Subarachnoid Aneurysm Trial (ISAT), which is comparing the outcome of treatment by surgery with that from endovascular treatment, but some represents true change in practice. The possibility that ISAT is constraining the development of endovascular intervention should also be considered, as suitable aneurysms for this technique are randomised to the Neurosurgical limb of the trial. The infrastructure to perform interventional procedures is in place at CMHCT, Salford and The Walton Centre. There is however a manpower problem with only three part time interventionists available to perform the procedures. These 37 Consultants are located one at each site although there is close collaboration between the CMHCT and Salford Consultants and some early collaboration between CMHCT and The Walton Centre. This level of staffing becomes a problem in two main areas. Firstly some procedures are complex and require two operators. Secondly single handed practices do not allow for holiday and sickness cover or on call provision. There is currently no interventional service available on the Preston site and patients are therefore transferred to other centres. Historically this has been to Oxford but in the last eighteen months the service has been provided by CMHCT. Interventional Capacity The capacity to expand the endovascular service to meet increasing requirements is dependent on the infrastructure and on available manpower. Within the region there are currently three Diagnostic Neuroradiologists with an interest in endovascular intervention. These clinicians spend increasing amounts of time involved in intervention without any reduction in their diagnostic workload. Indeed this workload is also increasing. The British Society of Neuroradiology recommends that for each interventional procedure there should be two allocated clinical sessions, one for the procedure itself and one for follow up including clinic visits and check imaging. If each interventionist were to perform three cases per week this would equate to 6 fixed sessions which would put serious pressures on the diagnostic neuroradiology service. This level of workload would allow only 360 procedures to be performed across the NWR. In reality it will be impossible for three people to meet this workload as often procedures need two operators and the destabilising effect on the diagnostic workload would be unacceptable. As interventionists become more experienced procedures become shorter, allowing more than one in each session. On manpower grounds we predict that, with existing staff levels, we will be able to perform 240 cases per year initially but this could rise to 300 if the procedures were performed in the same centre. Although many of the endovascular procedures replace a surgical procedure there is some difficulty in obtaining Anaesthetic sessions. This is in part due to timing of procedures, in part due to transfer of work between centres and in part due to vacated theatre sessions being used for other neurosurgery. Any expansion in endovascular procedures should therefore take into account the need for anaesthetic sessions to be available. This again would be easier if all intervention occurred on one site. Investment is also required in a rolling capital replacement scheme for suitable angiographic equipment. It is now increasingly recognised that intervention is more safely performed on biplane angiographic equipment and that 3D reconstruction can be helpful. We do not have access to this type of machine in the NWR. There are particular problems at Hope where the present angiographic capacity is inadequate for the anticipated workload following the transfer of the CMHCT service in April. 38 Predicted future workload Interventional Neuroradiology has a considerable potential for expansion in the NWR. It is clear from Table 1 that there has been a year on year growth and this is likely to continue. The growth has been due to a combination of improved technology and operator expertise. As a result there are increasing numbers of cases that can be treated successfully via the endovascular route. Data from other more established UK centres supports this but also suggests that activity may increase significantly above its present rate if staffing and infrastructure are established. If ISAT reports equivalence or better for endovascular treatment in the management of aneurysmal subarachnoid haemorrhage there will be further pressures to expand the service. Some of this pressure will come from patients who when given the choice will usually opt for less invasive therapy i.e. endovascular treatment. It is difficult to make accurate predictions about expansion. The total number of patients requiring treatment for aneurysmal subarachnoid haemorrhage in the NWR is approximately 400 cases per year. Of this total currently around 350 undergo some form of intervention. It is expected that this proportion will increase as endovascular techniques improve and become more widely available. Using these figures, and assuming that between 50% and 75% of intracranial aneurysms will be treated via the endovascular route as a minimum and a maximum figure the number of endovascular interventions for SAH can be calculated. To this number of cases must be added predicted figures for other endovascular procedures including AVM treatment. It is likely that actual activity will lie between these two predicted extremes (Table 2). Year 2002 2005 2010 Min 250 325 400 Max 350 450 550 TABLE 2 – Predicted future Regional interventional workload (includes AVMs etc) To meet these predicted levels of activity would require 3 or 4 interventionists to perform an average of 2 or 3 procedures each week. Clearly this change in practice needs to be carefully managed and funded to enable the most efficient and cost effective service to be provided. This is particularly important in view of the close geographical proximity of the three centres and the excellent motorway networks in the NWR. Development should be co-ordinated between these three sites so that any provision is complementary. This will allow 39 sensible planning and provision of services and allow clinicians to work closely together expanding their experience and improving patient outcome. There could also be economic benefits from collaboration. Different service models should therefore be explored in an effort to deliver the best service across the NWR. A number of possible models for regional organisation of the service are outlined below. 1. Completely independent development of services on the three sites with three separate units. 2. A single Regional centre for all endovascular intervention and vascular neurosurgery. 3. Two centres at Hope and the Walton Centre with transfer of patients form Preston to these units. 4. Regional Neurovascular team or teams with treatment at all three sites. i) Staff at all three sites with a co-ordinated service to provide cross cover when needed. ii) Staff providing a service across three centres when required. Whatever service model is decided upon there will be a shortage of staff in the short term. With growing realisation of the role of endovascular procedures there will be a requirement for on call services. This can only be sensibly provided with a minimum of 5 consultants. It is unlikely that the North West could recruit an additional 12 interventionists to allow an on call service on each site. In addition to maintain skill levels and embrace constantly changing technology interventionists should ideally perform at least 50 cases per year and have access to centres performing more complex procedures. Even the optimistic projections for the region do not expect a workload which could support more than 6 or 7 and could be met by 4 or 5 consultants. The larger catchment areas covered by Hope and The Walton Centre will probably generate a sufficient number of cases for at least two interventionists on each site. The workload in Preston is less and it may only be possible to justify a single interventionist on this site in the short and intermediate term. In view of this, if interventional neuroradiology were to be available on site at all three centres, it would be essential for operators across these sites to work closely together. In addition there is the potential for the development of at least one and possibly two regional training posts in intervention. This would be enhanced by close collaboration between units. The potential for research and development is also increased considerably with regional co-operation. Neurosurgical Provision in the Future The developments in endovascular techniques will have a profound effect upon neurovascular surgery and in particular on intracranial aneurysm surgery. There are approximately 45 procedures performed per million population per year for aneurysmal subarachnoid haemorrhage in the NWR. If 66% of these patients are treated via the endovascular route (as is the case in one UK unit currently, and which 40 is below the rate in France where it has become 80-85%) only 15 aneurysms per million will need to be treated surgically. It has been suggested in the past by the Society of British Neurological Surgeons that a Neurosurgeon should manage 25 cases per year to maintain competence. If this is accepted, a vascular Neurosurgeon would need to serve a population of almost 2 million. If only 15 operations a year were sufficient to maintain expertise each surgeon would need to serve 1 million assuming a 66% coiling rate. Higher coiling rates would increase the population base needed. Because it is necessary to provide a 24-hour, 7-day per week, 365 day per year service, a minimum of 2 surgeons would be required. This would however result in there being only one surgeon available, for 4 months of the year because of annual, study and professional leave. A more likely estimate of the number of surgeons required to provide full cover throughout the year, taking into account the working time directive and the construction of an acceptable on-call rota, is 4 – 6. Hence, the population base needed to ensure the quality of the Neurovascular Surgical Service would be a minimum of about 6 million. As subspecialisation increases in Neurosurgery, the NWR will need to move to this position which will only be sustainable with co-operation between the Centres in Salford, Preston and Liverpool. As the Neurovascular surgeons will continue to be involved in the management of patients undergoing coiling, for example in the management of delayed ischaemia, it is essential that Neurovascular teams consisting of Interventional Neuroradiologists and Vascular Neurosurgeons are formed. Later it would be ideal to add a Neurologist with a vascular interest and a Radiotherapist to this team. Rapid, high quality image transfer will be essential to allow such teams to work together effectively across the sites and make appropriate management decisions for the benefit of the patients in the NWR. Though most Neurosurgeons to date have been trained in aneurysm surgery and undertake it, rapidly many will become less skilled as the number of operations for aneurysms falls. Unfortunately, those patients coming to surgery will have the more technically difficult aneurysms. Inevitably this will result in further subspecialisation and in trainees gaining less experience in aneurysm surgery. It is likely that in time only a few trainees will be fully trained in vascular neurosurgery in order to deal with the residual, technically difficult aneurysms. Even to gain basic principles of the management of aneurysms, it will be necessary for Neurosurgical trainees to rotate to the Neurovascular service. Possible Service Models 1. Independent development of Intervention and surgery on the three sites. This will result in dilution of expertise and make on call provision of the service impossible. Preston may only be able to justify one interventionist working in isolation and 1 or 2 vascular neurosurgeons who will have no cover for holiday and study leave periods. It is unlikely that either Liverpool or Manchester will employ more than two interventionists in the intermediate term, which also makes local on call provision impossible. Numbers of operations are also likely to be too low for expertise to be maintained by the number of surgeons required for the on call rota 41 and for training to be undertaken. Audit and clinical governance will be of relatively little value with such small numbers. 2. A single regional centre for all endovascular intervention and vascular neurosurgery This would require a large redistribution of beds and nursing staff to the selected site. It would also involve relocation of existing staff and would in certain situations require patients to be transferred over relatively long distances. Concentration of staff would, however, increase experience and would probably benefit patient outcome. It would also simplify the provision of an out of hours service. However, Neurosurgeons and Neuroradiologists usually have more than one subspecialty interest and take part in general Neurosurgical and Neuroradiological emergency cover. There are strong links between some Neurosurgical sub-specialties, for example skull base surgery and vascular surgery. Unless there were to be only one Neuroscience Centre in the North West, not all of these other interests and responsibilities would be on the same site. The infrastructure to provide Neurovascular services for the whole of the NWR is not currently available on any one site. Movement of existing equipment or investment in new equipment would be costly. This, together with the inevitable impact on other aspects of Neurosurgery and Neuroradiology, makes it a difficult model to implement in the short to medium term. 3. Two centres at Hope and Walton with transfer of patients from Preston to these sites This model is closest to current practice for patients requiring endovascular procedures but does not give the people of the North of the Region a “local” service. Patients from Preston are transferred to CMHCT after investigation. This works well for relatively small numbers when the time frame is not critical, although there is pressure on access to beds and radiologist time in Manchester which does result in delays. Inevitably delay would lead to a poorer outcome for some patients. Increased collaboration with Liverpool could help to overcome these problems and could also ensure that the service is more available. It has the disadvantage of not delivering a service on the Preston site which would lead to the transfer of some patients for a second time. If surgery remained on the Preston site but no endovascular work was undertaken team working between radiologist and neurosurgeon could be difficult. Unless surgeons from Preston were to take part in the neurovascular service in one or both of the other centres there would be a de facto differentiation of the centres and Preston might well provide a particular focus for other forms of neurosurgery. Either outcome would necessitate the inclusion of all 3 centres in the planning and provision of neurosurgical services. Thus it does not seem possible to construct a 2 site service without 3 site cooperation. 42 4. Regional team or teams with cases performed on all three sites In this model Neurovascular disease would be managed on all three Neuroscience sites with local access to endovascular intervention and vascular neurosurgery. In this model staff could either (i) be based at each of the three sites with a team approach to covering the sites and sharing experience and expertise, or (ii) have a commitment to provide the service on a regional basis. The first of these models would probably be the best for continuity of care but will require careful selection of staff and also capital investment in equipment on both the Hope and Preston sites. It would be important for staff to share experience and take part in common audit. Close collaboration would also make a regional on call service possible. The second model involves staff working across three centres as required. This could necessitate clinicians working in different centres and might be less attractive. A combination of these two models may also be possible with regional staff supporting other staff based at each centre. Movement of patients would be minimised in all variants of this model as they depend upon movement of the doctor rather than the patient. Clinics could be held on each site in collaboration with the designated vascular Neurosurgeon. Proposal It is the recommendation of this group that collaboration across the three Neuroscience centres in the NWR is essential in establishing an Interventional Neuroradiology service and a Vascular Neurosurgical service to provide care for people with aneurysmal subarachnoid haemorrhage. Independent development on 3 sites would be to the detriment of patients and should be actively discouraged. The ideal solution would be to have state of the art, high quality services available to all three sites. These aims preclude service models 1,2 and 3. Service model 4 with the development of a regional specialist neurovascular team responsible for the service at each site would seem the most sensible way forward. Members of the team would have to collaborate closely sharing experience, providing cross-cover when required and taking part in common audit and governance arrangements. It is our view that the management of patients with neurovascular disease should only be undertaken by members of the neurovascular team. The number of people in the team will be governed by activity and outcome. Some members may have, or develop, experience in different areas providing super-specialist services, for example in the treatment of arteriovenous malformations. The growth of superspecialisation might take some time and in the short and intermediate term the team will agree mechanisms which will provide the best service for patients. There is an immediate need to provide a system for rapid transfer of angiographic, MR and CT images between centres to enable clinicians to discuss cases and make prompt and appropriate decisions. This will require teleradiology links between the three centres and this should therefore be actively pursued at an early stage. This will also be of value in other subspecialty areas. 43 Recommendations 1) Interventional Neuroradiology and Vascular Neurosurgery must be available to the population of the NWR 2) Development of NWR endovascular intervention and vascular neurosurgery across the three Neuroscience centres should be integrated. This requires close collaboration between the three centres and between Neuroradiologists and Neurosurgeons who should form a North West Neurovascular team. 3) The number of Neurosurgeons clipping aneurysms should reduce with local lead clinicians in this subspecialty becoming members of the regional team or network. 4) The Regional Clinical Neuroscience Project and commissioners should drive this development with advice from local and national experts. 5) Plans should be put into place to recruit further Interventionists as the workload increases. This may require in house training across the region. 6) Protected beds and anaesthetic sessions will be required. 7) Teleradiology linkage of the three sites should be installed as a matter of urgency. 8) Regional training in Neurovascular surgery and interventional neuroradiology should be organised. 44 Appendix 3 North West Neuro-vascular Network The North West Neuro-vascular Network will be the mechanism for the provision of care for people with non-traumatic sub-arachnoid haemorrhage, arterio-venous malformations and other intracranial vascular lesions. Neurosurgeons and Neuroradiologists involved in the provision of such a service will have accepted the working practice and standards of the Network and will work in accredited Centres. The Network will consist of : A Neuro-radiologists who: 1) 2) 7) Have been adequately trained in interventional techniques Will collect and supply all activity and outcome information to the Network Audit Will participate in regular quarterly multidisciplinary audit of outcome and complications Will report all clinical incidents and near misses Will perform on average more than 40 procedures each year, at least 20 of which should be aneurysms Will keep up to date by attendance at specialty society and post graduate meetings Will contribute to Neurosurgical and Neuroradiological training programmes B Neuro-surgeons who: 1) 2) Have been adequately trained in Neurovascular techniques Will collect and supply all activity and outcome information to the Network Audit Will participate in regular quarterly multidisciplinary audit of outcome and complications Will report all clinical incidents and near misses Will keep up to date by attendance at specialty society and post graduate meetings Will be responsible for/perform on average 20 procedures each year (averaged over 3 years) Will contribute to Neurosurgical and Neuroradiogical training programmes 3) 4) 5) 6) 3) 4) 5) 6) 7) The Network will be led by a clinician who will represent the interests of the team/network in the Neuroscience Partnership and will in turn be responsible for ensuring that the agreed goals and standards are met and that there is an agreed programme of audit and appraisal. Members of the network will have contracts which allow them to work in any one of the 3 Centres should this be necessary. 45 Appendix 4 Proposed Minimum Standards for Accreditation District General Hospitals 1) A & E Department with 24 hour consultant staffing 2) Observation ward or Acute admissions unit capable of 24 hour investigation and neurological observation 3) 24 hour CT scan availability within 1 hour 4) Image transfer facility to Neuroscience Centres 5) 24 hour access to biochemistry/pathology/microbiology laboratory (including CSF spectrophotometry) 6) Accredited junior staff posts with adequate supervision 7) Affiliation to the Neuroscience partnership Neuroscience Centres 1) 24 hour resident Neurosurgery and Neurology staff 2) 24 hour easy availability of consultant “Vascular” Neurosurgeon who is a member of the Neurovascular Team/Network, Neuroradiologist (including Interventional Neuroradiologist who is a member of the Neurovascular Team) and Neurologist. 3) Image transfer facilities (MR,CT,Angio) from DGHs and between Centres 4) Neuro-ITU 5) 24 hour availability of fully staffed Theatre and Neuroradiology department 6) Angiography and imaging equipment and staff training to an approved level 7) Participation in audit and benchmarking scheme 8) Participation in Regional Neuroscience training scheme 9) Membership of the Neuroscience Partnership. 46 Appendix 5 North West Clinical Neuroscience Partnership The Partnership came into being on September 1st 2000. Goal To minimise the impact of damage to the nervous system on the life of individuals and society through: 1) Seeking and using opportunities for prevention 2) Ensuring prompt access to appropriate, expert care following acute illness or injury 3) Developing paths of care which guarantee appropriate, accessible and sensitive care for people with chronic conditions 4) Supporting all providers of care whether lay or professional 5) Increasing understanding, in the NHS and wider society, of the impact of damage to the nervous system on the lives of affected individuals 6) Involving patients, carers and voluntary organisations in decision making, planning and evaluation of services. Working Together Trusts and Commissioners will work together to develop strategies to achieve the above goals. They will: 1) Together agree a North West Service Framework for the provision of Clinical Neuroscience services 2) Together encourage the development of North West Commissioning for the Clinical Neurosciences. In order to achieve this they agree to: 1) Share activity and outcome information 2) Develop a joint programme of benchmarking and audit 47 3) Develop a common understanding of manpower and training requirements 4) Ensure that proposals and job plans for new senior clinicians (Consultants and Nurse Consultants) are shared at an early stage and in all cases prior to appointment 5) Share development plans and capital bids above £100,000 at an early stage and at latest, prior to submission of a business case or service plan 6) Explore mechanisms for joint evaluation and introduction of new drugs and technologies 7) Define the information requirements to support co-operative working and develop the information system to deliver them. Monitoring This is a co-operative venture and it is not anticipated that individual groups or organisations will be less than fully committed. In the event of a dispute, the group will determine an acceptable procedure, possibly involving the RSCG, to resolve the issue. Conclusion This partnership represents an agreement between the three Tertiary Centre Trusts and the three Zonal Neuroscience Commissioners to work together to ensure that the population of the North West and North Wales has equitable access to a Clinical Neuroscience service which will meet their needs and will be both effective and efficient in its use of resources. Members (As at Sept 1st, 2000) Chief Executive, Preston Acute Hospitals NHS Trust Chief Executive, Salford Royal Hospitals Trust Chief Executive, Walton Centre for Neurology and Neurosurgery Specialised Commissioning Lead, Neurosciences, Greater Manchester Zone Specialised Commissioning Lead, Neurosciences, Lancashire and South Cumbria Specialised Commissioning Lead, Neurosciences, Merseyside and Cheshire Member, Specialist Health Services Commisioners for Wales Regional Specialised Commissioning Manager, NW Regional Office, NHS Clinical Director, Neurosciences, Preston Director of Neurosciences, Greater Manchester Medical Director, Walton Centre Chief Executive of Health Authority as Chair, (Sefton nominated from 2000) 48 Appendix 6 North West Clinical Neuroscience Project Role of Voluntary Agencies Group The North West Clinical Neuroscience Project was initiated by a group of Providers and Commissioners across the North West of England and North Wales. It seeks to improve the quality, accessibility and equity of care for people with neurological conditions. The Voluntary Agencies Group was convened to be a source of information and advice for the Project. 1) They will provide background information on the experience of people affected by damage to the nervous system (as patients, relatives or carers) drawing attention to successes and failures in the present provision of care. 2) They will also advise on unmet need and potential improvements 3) All proposals for changes in the organisation or provision of services will be brought to the Group for comment and discussion. 4) It is expected that all members of the group will encourage wider discussion of the issues as they are raised and will feed comments and suggestions into the Group. 5) The Project concludes in March 2001. The role and standing of the Voluntary Agencies Group should be reviewed at that time. 6) The Group should develop proposals for a continuation of a mechanism for the inclusion of a user voice in the commissioning and provision of Clinical Neuroscience Services. 49 References 1 Ingall T, Asplund K, Mahonen M et al: A multinational comparison of subarachnoid haemorrhage in the WHO MONICA stroke study. Stroke 31: 1054-1061, 2000 2 Bonita R, Beaglehole R, North JDK et al: Subarachnoid haemorrhage in New Zealand: an epidemiological study. Stroke 14: 342-347, 1983 3 Cesarini KG, Hardemark H-G & Persson L: Improved survival after aneurysmal subarachnoid hemorrhage: review of case management during a 12-year period. J.Neurosurg 90: 664-672, 1999 4 Ingall TJ, Whisnant JP, Wiebers DO et al: Has there been a decline in subarachnoid haemorrhage mortality? Stroke 20: 718-724, 1989 5 Kassell NF, Torner JC, Haley EC et al: The International Cooperative Study on the Timing of Aneurysm Surgery. Part 1: Overall management results. J.Neurosurg 73:18-36 1990 6 Roos Y: Antifibrinolytic treatment in subarachnoid hemorrhage. A randomized placebo-controlled trial. Neurology 54: 77-82, 2000 7 Pickard JD, Murray GD, Illingworth R et al : Effect of oral Nimodipine on cerebral infarction and outcome after subarachnoid haemorrhage: British aneurysm nimodipine trial. Br Med J 298: 636-642,1989 8 Kassell NF, Peerless SJ, Durward QJ et al: Treatment of ischemic deficits from vasospasm with intravascular volume expansion and induced arterial hypertension. Neurosurgery 11: 337-343, 1982 9 Kassell NF, Torner JC, Jane JA et al: The International Cooperative Study on the Timing of Aneurysm Surgery. Part 2: Surgical results. J Neurosurg 73: 37-47, 1990 10 Buchanan KM, Elias LJ and Goplen GB: Differing perspectives on Outcome after Subarachnoid Hemorrhage: The Patient, the Relative, the Neurosurgeon. Neurosurgery 46: 831-840, 2000 11 Hackett ML and Anderson CS: Health outcomes 1 year after subarachnoid hemorrhage: An international population-based study. The Australian Cooperative Research on Subarachnoid Hemorrhage Study Group. Neurology 55: 658-662, 2000 12 Guglielmi G, Vinuela F, Dion J, et al: Electrothrombosis of saccular aneurysms via the endovascular approach. Part 2: Preliminary clinical experience. J.Neurosurg 75: 8-14, 1991 50 13 Vinuela F, Duckwiler G and Mawad M: Guglielmi detachable coil embolisation of acute intracranial aneurysm: perioperative anatomical and clinical outcome in 403 patients. J Neurosurg 86: 475-482, 1997 14 Brilstra EH, Rinkel GJ, van der Graaf Y et al: Treatment of intracranial aneurysms by embolisation with coils: a systematic review. Stroke 30: 470-476, 2000 15 Byrne JV, Sohn MJ, Molyneux AJ et al: Five year experience using coil embolisation for ruptured intracranial aneurysms: outcomes and incidence of late rebleeding. J. Neurosurg 90: 656-663, 1999 16 Lempert TE, Malek AM, Halbach VV et al: Endovascular Treatment of Ruptured Posterior Circulation Cerebral Aneurysms. Stroke 31: 100-110, 2000 17 Koivisto T, Vanninen R, Hurskainen H et al: Outcomes of Early Endovascular Versus Surgical Treatment of Ruptured Cerebral Aneurysms: A Prospective Randomised Study. Stroke 31: 2369-2377, 2000 18 Claiborne Johnston: Effect of Endovascular Services and Hospital Volume on Cerebral Aneurysm Treatment Outcomes. Stroke 31: 111-117, 2000 19 Edlow JA and Caplan LR: Avoiding pitfalls in the diagnosis of subarachnoid hemorrhage. New Eng J Med 342: 29-36, 2000 20 van der Wee N, Rinkel GJ, Hasan D et al: Detection of subarachnoid haemorrhage on early CT: is lumbar puncture still needed after a negative scan? J Neurol Neurosurg Psychiatry 58: 357-359, 1995 21 Hutchinson PJ, Seeley HM and Kirkpatrick PJ: Factors implicated in deaths from subarachnoid haemorrhage. Are they avoidable? Br J Neurosurg 12: 37-40, 1998 22 Maurice-Williams RS and Kitchen D: Ruptured intracranial aneurysms—learning from experience. Br J Neurosurg 8: 519-527, 1994 23 Hannan EL, O’Donnell JF, Kilburn H et al: Investigation of the relationship between volume and mortality for surgical procedures performed in New York State hospitals. JAMA 262: 503-510, 1989 24 Jollis JG, Peterson ED, DeLong ER et al: The relation between the volume of coronary angioplasty procedures at hospitals treating medicare benificiaries and short-term mortality. N Engl J Med 331: 1625-1629, 1994 25 Solomon RA Mayer SA and Tarmey JJ: Relationship Between the Volume of Craniotomies for Cerebral Aneurysm Performed at New York State Hospitals and InHospital Mortality. Stroke 27: 13-17, 1996 51 26 Safe Neurosurgery. Society of British Neurological Surgeons. 1993 27 Safe Neurosurgery 2000. Society of British Neurological Surgeons, London. 2000 28 Playford ED, Lawson L, Limbert V et al: Goal-setting in rehabilitation: report of a workshop to explore professionals’ perceptions of goal-setting. Clin Rehabil 14: 491496, 2000 29 Hitchcock R, Hutchings CJ, Stephenson S et al: Neurological rehabilitation in Indonesia and the UK: differences and similarities. J Allied Health 27: 45-49, 1998 30 Molyneux AJ, Personal communication 52