Spectroscopic results - Springer Static Content Server
advertisement

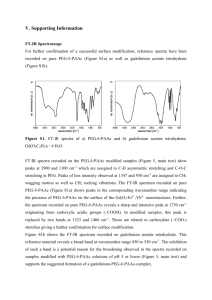
Electronic Supplementary Material Direct electrochemistry of hemoglobin on an ionic liquid carbon electrode modified with zinc tungstate nanorods Chengxiang Ruan1*, Zhaolan Sun2, Jun Liu2, Jin Lou1, Weimin Gao1, Wei Sun2*, Yunsheng Xiao1 1 Jiangxi Key Laboratory of Surface Engineering, Jiangxi Science and Technology Normal University, Nanchang 330013, P. R. China; 2 College of Chemistry and Molecular Engineering, Qingdao University of Science and Technology, Qingdao 266042, P. R. China *Corresponding author, Tel & Fax: +86-532-84023927, E-mail: cxruan@126.com and sunwei@qust.edu.cn Spectroscopic results FT-IR spectroscopy is a sensitive method to probe into the secondary structure of proteins. The profiles of the amide I and II infrared absorbance bands of Hb provide detailed information on the secondary structure of the polypeptide chain. The amide I band at 1700–1600 cm−1 is attributed to the C=O stretching vibration of the peptide linkage in the backbone of protein. The amide II band at 1600–1500 cm−1 is caused by the combination of N–H in plane bending and C–N stretching vibration of the peptide groups [1]. If protein molecule is denatured, the intensity and shape of the amide I and II bands will diminish or disappear [2, 3]. As shown in Fig. S1A, the FT-IR spectra of amide I and II bands of native Hb were located at 1649 and 1533 cm−1 (Fig. S1Aa), respectively. The mixture of Hb, ZnWO4 and CTS together gave the spectra of amide I and II bands at 1643 and 1531 cm−1 (Fig.S1Ab). The similarities of the two spectra suggested that Hb retained the essential features of its native structure after mixed with the CTS-ZnWO4 composite materials, which could be attributed to the biocompatibility of the ZnWO4 nanorods and CTS. Tungstate has some advantages 1 such as high chemical stability and good physical properties. CTS is an abundant natural biopolymer originated from the exoskeleton of crustaceans, which is biocompatible, biodegradable without toxicity. So the Hb molecules in the CTS and ZnWO4 nanorods composite remained its native structure. In UV-Vis absorption spectrum, the position of Soret absorption band of heme may provide information on the conformational structure of heme proteins, especially that of the conformational changes in the heme group region [4]. As shown in Fig. S1B, the Soret band of Hb appeared at 405.0 nm in 0.1 mol L-1 pH 7.0 phosphate buffer solution (curve a), While the Hb molecules in ZnWO4 and CTS mixture gave the Soret band at the same positions without any changes (curves b and c), indicating that Hb molecules remained their native structure in the nanocomposite materials. Figure S1 (A) FT-IR spectra of (a) Hb and (b) CTS-Hb-ZnWO4 film. (B) UV-Vis absorption spectra of (a) Hb, (b) CTS-Hb-ZnWO4 and (c) ZnWO4-Hb in pH 7.0 phosphate buffer solution. 2 References [1] Kauppinen JK, Moffatt DJ, Mantsch HH, Cameron DG (1981) Fourier self-deconvolution: a method for resolving intrinsically overlapped bands. Appl. Spectrosc.35: 271-276. [2] Song YP, Petty MC, Yarwood J, Feast WJ, Tsibouklis J, Mukherjee S (1992) Fourier transform infrared studies of molecular ordering and interactions in Langmuir-Blodgett films containing nitrostilbene and stearic acid. Langmuir 8: 257-261. [3] Nassar AE, Willis WS, Rusling JF (1995) Electron transfer from electrodes to myoglobin: facilitated in surfactant films and blocked by adsorbed biomacromolecules. Anal. Chem. 67: 2386-2392. [4] George P, Hanania GIH (1953) A spectrophotometric study of ionizations in methaemoglobin. J. Biochem. 55: 236-243. 3