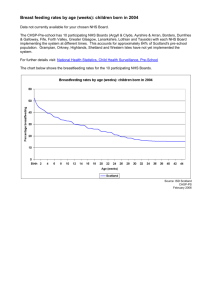
Improving Access to Medicines for NHS Patients and
advertisement

Agenda Item: 2.4 Ref: W08-09/177 NHS Wirral Paper 1: Background and Summary Improving Access to Medicines for NHS Patients and Guidance on NHS Patients Paying for Private Care Background 1. Over the last year a series of cases came to public attention where patients have pursued private treatment for cancer drugs that have not been approved by the National Institute for Clinical Excellence (NICE) on the grounds of cost-effectiveness. 2. This is hitting the headlines now due to the emergence of increasing numbers of drugs that are targeted to treat often relatively small groups of patients for a short period of time near the end of life, prolonging survival without cure. Drug prices are high due to the limited return and exceed the cost-effective parameters used by NICE to determine best use of NHS resources. 3. There is also a considerable time lag between drug licensing and NICE guidance and PCTs are required to make local decisions in the interim. Concerns have been raised nationally regarding the variation in access to medicines across the country via these local decision making processes. 4. A further complication is variable interpretation of previous guidance1,2 on how Trusts should handle cases where patients choose to purchase additional private care. Some patients have had access to NHS care withdrawn whilst other patients have benefited from a more flexible interpretation and maintained NHS care for the same condition. Recommendations from the Review 5. On 4 November 2008, David Nicholson communicated the outcome of the review3 by Professor Mike Richards, National Director for Cancer. The scope of the review was to: understand current law and practice relating to purchasing unfunded drugs investigate the situation in other countries seek the views of as many stakeholders as possible- public, patients, clinicians, NHS managers and the pharmaceutical industry understand the reasons why people have been placed in a position where they feel they need to purchase medicines. 6. The Secretary of State has accepted all fourteen recommendations following the review3, these are detailed in Appendix 1. Key recommendations to increase access to medicines on the NHS and clarify guidance on access to NHS care are summarised below. 7. Reforms to increase access to medicines on the NHS include a series of measures to make more treatments available on the NHS: More timely NICE guidance, to enable NHS availability more quickly. Consistent and robust PCT decisions where NICE guidance is not available or when dealing with applications for exceptional case funding. PCTs are required to review and Improving Access to Medicines for NHS Patients: Board Meeting (9 December 2008) 1/5 NHS Wirral Agenda Item: 2.4 Ref: W08-09/177 improve their local decision-making processes and SHAs will review how PCTs collaborate regarding these decisions by April 2009. This is discussed in Paper 4. Extending access to new drugs, by accepting NICE’s proposal to introduce more flexibility for recommending higher cost-drugs which have been shown to extend the lives of patients with terminal illnesses. This is discussed in Paper 2. More collaboration with the pharmaceutical industry, with the introduction of more flexible pricing structures and patient access schemes, set out in the new PPRS agreement5 8. By making more medicines available on the NHS, these measures will reduce the need for private care. There may still be a small number who wish to buy a drug privately which does not provide sufficient clinical benefit to justify the cost to the NHS. The revised guidance on purchasing additional private care4 is in draft for consultation, discussed in Paper 3. However, with immediate effect, all decisions affecting NHS patients must be made in line with the principles set out in the guidance: There should be no withdrawal of NHS care because a patient chooses to purchase private care over and above their NHS care; and Private care should be separated from NHS care, provided at a separate setting 9. The review3 appraised various options to include in the revised guidance, illustrated below. The options range along a spectrum, from ensuring the complete separation of NHS care and treatment purchased privately, to encouraging complete integration. The review3 sets out further detail describing why separate care was recommended. Recommendations 10. The Board is invited to: note and comment on the Richards Review comment on recommendations regarding End of Life Medicines (Paper 2), specifically questions in Para. 4 and the points in Para 5 comment on the questions in the consultation on paying for additional private care (Paper 3, para 5), including suggestions for an economy-wide clinical consultation note the proposed arrangements for collaboration across PCTs in the North West (Paper 4) Cathy Gritzner, Director of Health Systems Management Dr Shyamal Mukherjee . Medicines Management Lead Dr Shanila Roohi, Health Treatment Panel Lead Julia Simms, Head of Medicines Management Val Attwood, Deputy Head of Commissioning and Contracting Improving Access to Medicines for NHS Patients: Board Meeting (9 December 2008) 2/5 NHS Wirral Agenda Item: 2.4 Ref: W08-09/177 References 1. BMA. A code of practice on private practice. 2006. www.bma.org.uk/ap.nsf/Content/CCSCContractprivMS. 2. Department of Health. A code of conduct for private practice: guidance for NHS medical staff. 2003 www.dh.gov.uk/en/Publicationsandstatistics/Publications/PublicationsPolicyAndGuidance/DH_410 0689. 3. Richards M. Improving access to medicines for NHS patients. 2008. London: Department of Health. www.dh.gov.uk/en/Publicationsandstatistics/Publications/PublicationsPolicyAndGuidance/DH_089 927. 4. Department of Health. Draft guidance on NHS patients who wish to pay for additional private care. 2008. www.dh.gov.uk/en/Consultations/Liveconsultations/DH_089926. 5. Pharmaceutical Price Regulation Scheme (PPRS) http://www.dh.gov.uk/en/Healthcare/Medicinespharmacyandindustry/Pharmaceuticalpriceregulatio nscheme/index.htm Improving Access to Medicines for NHS Patients: Board Meeting (9 December 2008) 3/5 NHS Wirral Agenda Item: 2.4 Ref: W08-09/177 Appendix 1 Recommendations of the Review3 Recommendation 1: The measures the government is already taking to improve the timeliness of the NICE decision making process are extremely welcome and should be strongly supported. The Department of Health and NICE should publish an update on the timelines for delivering these important commitments. Recommendation 2: The Department of Health should urgently consider how PCTs can be encouraged to work together to make proactive commissioning decisions. Consideration should be given to whether collaborative processes already developed, such as in the North East for cancer drugs, could be used as a model. Recommendation 3: The commitment made in the draft NHS Constitution to ensure transparency in PCT decision making, and the resulting work being undertaken by the National Prescribing Centre to support PCTs in delivering this, is extremely welcome. The government should set out as soon as possible more detailed plans for how it will achieve the commitment in the NHS Constitution, including the timescale for this work. Recommendation 4: In developing collaborative arrangements for decision making, the government should also consider how PCTs can be better supported to make decisions on funding off-label drugs, whether as a matter of policy or on an exceptions basis. Recommendation 5: The Department of Health should work: with NICE to assess urgently what affordable measures could be taken to make available drugs used near the end of life that do not meet the cost-effectiveness criteria currently applied to all drugs; and with the pharmaceutical industry in the context of the current Pharmaceutical Price Regulation Scheme (PPRS) negotiations to promote more flexible approaches to the pricing and availability of new drugs. This will require partnership working with the pharmaceutical industry and greater flexibility in approach from all parties. Recommendation 6: The Department of Health should urgently undertake further work to investigate the extent and causes of international variations in drug usage. Recommendation 7: The Department of Health should clarify the policy on how the NHS should handle situations where a patient wishes to purchase additional treatment. The objective should be to ensure consistency in practice across the NHS. Improving Access to Medicines for NHS Patients: Board Meeting (9 December 2008) 4/5 NHS Wirral Agenda Item: 2.4 Ref: W08-09/177 Recommendation 8: The Department of Health should make clear that no patient should lose their entitlement to NHS care they would have otherwise received, simply because they opt to purchase additional treatment for their condition. Recommendation 9: The government should make clear that: clinicians should exhaust all reasonable avenues for securing NHS funding before a patient considers whether to purchase additional drugs; patients should be able to receive additional private drugs as long as these are delivered separately from the NHS elements of their care; and providers should establish clear clinical governance arrangements to ensure that patients who do elect to purchase additional private treatment receive good continuity of care. Recommendation 10: SHAs, working where appropriate through cancer networks, should ensure that local policies are developed to ensure that any revised guidance issued by the government is implemented properly. This might include using a designated hospital with private facilities for all patients wishing to purchase additional drugs, making use of homecare provision or designating an area of an NHS hospital or the delivery of privately funded treatments. Recommendation 11: The Department of Health should take a lead on commissioning a national audit of demand for unfunded drugs and on the outcome of treatments, working closely with professional organisations and NHS managers. Recommendation 12: Doctors who are likely to have conversations with patients about treatments that are not routinely funded on the NHS should ensure that they have the necessary knowledge and skills to communicate complex information effectively and in a balanced way. This will help patients to make informed assessments about the balance of risk, cost and benefit involved in any potential treatment. The Department of Health should commission a training programme for clinicians to enhance the quality of discussion about these difficult issues. Relevant Royal Colleges should consider how assessment of communication skills could best be incorporated into re-certification processes. Recommendation 13: The Department of Health should consider how patients could best be given access to balanced written information on the benefits, toxicities and, where appropriate, costs of novel treatments, especially those given to patients near the end of life. Recommendation 14: In responding to this Review, the government should confirm how situations where patients wish to purchase additional non-drug interventions should be handled. Improving Access to Medicines for NHS Patients: Board Meeting (9 December 2008) 5/5