animal handler precuations - University of Southern California
advertisement

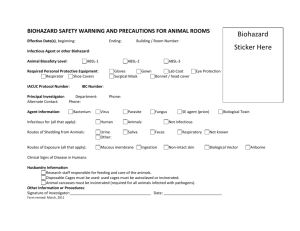
Exposure Control Plan for: Lentiviral Vectors Containment Level: BSL2 I. HAZARDS Lentiviruses are a subset of retroviruses with the ability to integrate into host chromosomes and to infect proliferating and nonproliferating cells. These viruses can cause severe immunologic and neurologic disease in their natural hosts. Most of the lentiviral vectors presently in use are derived from the Human Immunodeficiency Virus (HIV). The major risks to be considered for research with HIV based lentiviral vectors are the potential for generation of replication-competent lentivirus (RCL) and the potential for oncogenesis. These risks can be mitigated by the nature of the vector system and its safety features (i.e., use of multiple plasmids for vector packaging, deletion of overlapping sequences between the packaging plasmids, minimalist design of the vector backbone) or exacerbated by the nature of the transgene insert (i.e., oncogene) encoded by the vector. Although most viral vectors used for gene delivery are not replication competent, virions or infectious particles produced with these agents are capable of infecting lab personnel. Consequently, these materials should be treated and handled with appropriate care – strict adherence to BSL2 containment is a necessity. II. MODES OF TRANSMISSION The most likely modes of transmission within the lab include penetration of the skin via puncture or absorption through scratches, cuts, etc., and direct contact with mucous membranes (eyes, nose and mouth). III. ENGINEERING CONTROLS A Certified Biosafety Cabinet must be used for all manipulations of the viral vector (i.e., pipetting, harvesting, infecting cells, filling tubes/containers, opening sealed centrifuge canisters, shaking, mixing, etc.) and for handling infected cells. Safety Engineered Sharps shall be used for Lentivirus injections. In addition, the use of other sharps (i.e., Pasteur pipettes) must be eliminated wherever possible. For animal injections, the animal must be restrained or anesthetized Biohazard Sharps Containers shall be available to dispose of sharps waste, including broken glass, needles, blades, etc. When centrifuging, use aerosol containment devices such as sealed canisters that fit in the centrifuge bucket, covers for the centrifuge bucket, heat sealed tubes, or sealed centrifuge rotors. Rotors should be removed and opened inside a BSC. Centrifuge tubes should be filled and opened in BSC An in-line HEPA filter must be used for vacuum aspiration of spent media IV. ADIMINISTRATIVE/WORK PRACTICE CONTROLS Access to the lab shall be restricted while work is in progress A sign incorporating the universal biohazard symbol shall be posted at the entrance of the laboratory or tissue culture room where lentivirus is used (see last page) All lab personnel must be informed of the hazards of Lentiviruses All lab personnel must be trained in proper handling, use, and disposal prior to working Lentiviruses All lab personnel are advised to avoid rubbing eyes as a precautionary measure against eye infections All lab personnel will remove lab coat, discard gloves, and wash hands before exiting the lab V. PERSONAL PROTECTIVE EQUIPMENT Lab coat shall be worn while working in the lab Safety glasses or goggles shall be worn when handling Lentivirus Gloves shall be worn while working in the lab Respirators are required for aerosol-producing procedures performed outside of a biosafety cabinet. Contact the EH&S office at (323)442-2200 for fit-testing prior to use of respirators. VI. DISINFECTION General surface disinfection with freshly prepared 10% bleach (1:10 dilution household bleach, such as clorox) allowing a contact time of 15 minutes. For stainless steel surfaces, follow bleach disinfection with 70% ethanol wipedown to avoid corrosion. Liquid waste may be treated by exposing to bleach (final volume 10%) for 15 minutes before disposing into sink. VII. DISPOSAL Solid biohazardous waste, such as culture vials, plates, plastic tubes, etc., are disposed of into biohazard waste bins lined with red bags for pickup by EH&S. Biohazard bins are labeled with biohazard stickers, and lids are to remain secured when not in use. Sharps waste, such as broken glass, pasteur pipets, razor blades, and needles, are disposed of into biohazard sharps containers. When the fill line on the sharps container is reached, the lid will be secured and the container placed next to the biohazard bin for pickup by EH&S. To request a pickup, call the EH&S waste disposal line at (323)442-2225. VIII. ACCIDENTAL SPILL In case of spill: Immediately notify others around you Don appropriate personal protective equipment, such as gloves, labcoat, and safety glasses Use forceps to remove any broken glass or other sharp items, which should be placed into biohazard sharps containers Cover the spill with paper towels or other absorbent materials Apply 10% bleach directly onto and around the paper towels covering the spill Allow 15 minute contact time before cleaning, starting at perimeter and working inwards towards center Dispose of materials into biohazard bins For large spills, you may contact the EH&S office for assistance at (323)442-2200 IX. EXPSOURE RESPONSE In the event of an exposure, take the following precautions: Remove any contaminated clothing Wash all affected areas; for eye exposures, rinse for 15 minutes in eyewash or flush area with water, for needle-stick or other sharps exposure, wash wound area with soap and water for 15 minutes Report the exposure to your supervisor immediately For students, seek treatment at the Eric Cohen Student Health Center: Healthcare Consultation Center (HCC) 1510 San Pablo St., Suite 104 (323)442-5631 For employees, seek treatment at Internal Medicine: Healthcare Consultation Center II (HCC II) 1520 San Pablo St. (323)442-5100 For any emergency, you may also contact the Department of Public Safety at (323) 442-1000 Healthcare personnel treating exposed patients should be informed of the nature of the agent (i.e., lentiviral vector derived from HIV-1) Information on workers’ compensation can be found at this website: http://capsnet.usc.edu/WC/, or you may call: (213)7406205 Incidents should be reported to the Institutional Biosafety Committee as soon as possible: IBC@caps.usc.edu or (323)4422200 Revised 6/2/09 X. SPECIAL PRACTICES FOR ANIMAL INJECTIONS Laboratory staff must make arrangements for housing these animals and for disposing of the contaminated bedding by contacting the appropriate animal caretaker supervisor before dosing begins Facility: When animals are infected with lentiviral vectors, the Animal Biosafety Level of the project will generally be assigned to ABSL-2. This requires Biosafety Level-2 practice and facilities for procedures involving lentiviral vectors. Signage: Attached door sign must be posted on the door leading into the housing or procedure room. Cages must be labeled with the biohazard cage card label with agent identification and injection date upon injection of agent. Signage/labels must remain in place for a minimum of 1 week after the date of injection/exposure. Animal Excretion: Infected animals my excrete lentivirus. Precaution must be taken not to create aerosols when emptying animal waste material and when washing down cages, or cleaning the room with pressure hoses. Surfaces that may be contaminated will be decontaminated ASAP with bleach solution. This practice must be followed for one full week after infection. Cage Change: Use a certified Class II biosafety cabinet when moving animal from dirty to clean cages. If multiple cages are being changed in the same biosafety cabinet, cages with animals infected with the lentiviral vectors should be changed last. Spray the inside surfaces of each cage with bleach solution and leave the cages in the biosafety cabinet for 15 minutes before returning them to the cage washing facility. Decontaminate the biosafety cabinet immediate after removing the contaminated cages with bleach solution. Bedding: A biosafety cabinet or negative airflow cage changing station should be used when disposing bedding into biohazard red bag. The red biohazard bag must be sealed and placed into the biohazard container. Animal Carcass: Infected carcasses should be placed in red biohazard bag and stored in designated freezer for EH&S pick-up. EH&S must be informed when carcasses are put into freezer. Revised 6/2/09 University of Southern California Laboratory Safety (323) 442-2200 ANIMAL HANDLER PRECUATIONS Building: Room Number: The animals in this program are part of an approved experiment involving radioactive materials, biohazardous organisms, and/or chemical carcinogens. The requirements below apply to this experiment. Principal Investigator: Department: Phone: Emergency Contact Person: Phone: IACUC Protocol: Identification of Hazard(s): LENTIVIRAL VECTOR Animal Biosafety Level: 2 Required PPE 1. Lab coat/coveralls 2. Head cover 3. Shoe covers 4. Surgical Mask 5. Safety Glasses (if animals handled outside of a biosafety cabinet or ventilated cage-changing station) Special Instructions 1. All animals must be housed in micro-isolation cages. 2. Individual cages must be labeled with biohazard stickers upon injection of agent. 3. All animal handling must occur inside certified biological safety cabinet. 4. All animal injections must occur in certified biosafety cabinet in a procedure room. 5. Biosafety cabinet must be decontaminated with 5% bleach after use or cage change. 6. Contaminated bedding must be disposed into Biohazard Waste inside biosafety cabinet or negative airflow cage changing station. 7. Contaminated animal carcasses must be disposed in a red biohazard bag and place in the designated freezer. Inform EH&S (HSC: x22225, UPC: x07215) immediately for pick-up. 8. Hands must be washed with soap and water upon leaving room. Revised 6/2/09 AUTHORIZED PERSONNEL ONLY WHEN LENTIVIRUS WORK IN PROGRESS University of Southern California Laboratory Safety (323) 442-2200 BIOSAFETY PRECUATIONS Building: Room Number: Identification of Hazard(s): LENTIVIRAL VECTOR Biosafety Level: 2 Principal Investigator: Department: Phone: Emergency Contact Person: Phone: IBC Protocol#: Date: Authorized Personnel: Revised 6/2/09