Germination of sunflower genotypes with modified fatty acid
advertisement

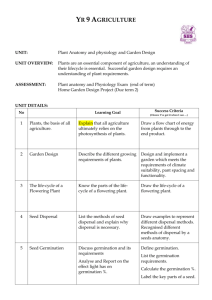
Germination of sunflower genotypes with modified fatty acid composition Raúl González Belo.12, Roberto Benech Arnold, R.34, Jorge Tognetti12, Silvina San Martino5, Natalia Izquierdo14 1 Laboratorio de Fisiología Vegetal. Facultad de Ciencias Agrarias, Universidad Nacional de Mar del Plata, Ruta 226 km 73.5, (7620) Balcarce, Argentina. 2 Comisión de Investigaciones Científicas de la Provincia de Buenos Aires, Calle 526 entre 19 y 20, (1900) La Plata, Argentina. 3 Consejo Nacional de Investigaciones Científicas y Técnicas, CONICET, Av. Rivadavia 1917, (A1033AAJ) Ciudad Autónoma de Buenos Aires, Argentina. 4 Facultad de Agronomía, Universidad de Buenos Aires, Av. San Martín 4453, (C1417DSE) Ciudad Autónoma de Buenos Aires, Argentina. 5 Grupo de Estadística. Facultad de Ciencias Agrarias, Universidad Nacional de Mar del Plata, Ruta 226 km 73.5. (7620) Balcarce, Argentina. e-mail: nizquierdo@balcarce.inta.gov.ar ABSTRACT Soil temperature at planting date determines germination speed and therefore the quality of plant stand. In sunflower crops in Argentina, soil temperature can vary with regions, planting dates and tillage systems. For example, soil temperature in no-tillage systems may be 1-2°C lower than in conventional tillage. Preliminary evidences suggest that the fatty acid composition of sunflower seeds could modify its germination, in a possible interaction with temperature. These effects may be of a high applied importance due to increasing cultivation of sunflower genotypes whose fatty acid composition has been modified for nutritional and health purposes. However, detailed studies are still lacking. The aim of this work was to study the effect of temperature on the germination of sunflower genotypes with modified fatty acid composition. Experiments were performed with traditional, high oleic (HO, oleic acid >80%), high stearic high linoleic (HSHL, stearic acid >15%) and high stearic high oleic (HSHO, stearic acid >15% and oleic acid >60%) sunflower genotypes. In Experiment 1, a traditional hybrid, a hybrid and a line HSHO and a hybrid and two inbred lines HSHL were used. In Experiment 2, a traditional, a HO and a HSHO hybrid were used. In both experiments, seeds were incubated in 8-10 constant temperatures within a range 5°C - 38ºC, in darkness, using growth chambers. Three replications of 20 seeds per genotype were placed in 9 cm diameter plastic Petri dishes. The absorbent paper of the dishes was moistened with distilled water. Germination was recorded as protrusion of the radicle (3mm) and it was monitored each 12-24h over 20 days. At the end of the trial, a tetrazolium test was conducted to the non-germinated seeds to assess their viability. The germination rate was calculated for each temperature and the base temperature (Tb) was estimated for each genotype. In both experiments, the germination rate linearly increased with temperature. In Experiment 1, the maximum germination rate was 27°C for the traditional genotype, whereas for HSHO and HSHL genotypes, the highest germination rate was observed between 31ºC and 38ºC. In the three genotypes in Experiment 2, the highest germination rates were observed around 34ºC. In both experiments, genotype had a significant effect on base temperature for germination (p=1.55-7 and p=4.01-3, for Experiment 1 and 2, respectively). In Exp 1, in general, HSHL genotypes presented lower T b than HSHO genotypes (1.98ºC vs. 2.36ºC, respectively). In both experiments, genotypes with modified fatty acid composition presented higher Tb than the traditional ones (1.75-2.70°C vs. 0.57°C for Exp. 1 and 2.90-3.29ºC vs. 2.33ºC for Exp. 2). For a given fatty acid composition, the inbred lines presented a higher Tb than hybrids. Temperature increased the germination rate in both traditional and modified fatty acid composition genotypes. The latter presented a higher base temperature than traditional ones. This higher base temperature would result in a lower accumulation of thermal time required for germination. The reasons of this higher base temperature of genotypes with modified composition are still unknown. These results suggest that soil temperature at planting time can play an important role in obtaining an adequate stand of plants in sunflower genotypes with modified fatty acid composition. Thus, selecting the growing area and sowing date may be key factors for cultivating these genotypes. Keywords:– stearic – oleic – germination rate - base temperature INTRODUCTION Rapid and uniform seed germination in field are essential for improving crop establishment and growth, and ultimately to obtain high yields. In sunflower, seeds may encounter relatively low soil temperatures depending on cultivation area, planting date and tillage system. For example, average soil temperatures in direct sowing can be 1-2°C lower than in conventional planting (Aase and Siddoway, 1980; Potter et al., 1985). In recent years, different genotypes with modified fatty acid composition have been developed to meet the growing demand for high quality oils. These oils have better nutritional quality, higher stability towards oxidation, and are more solid at room temperature than traditional ones, which make them best suited to particular uses such as frying or fabrication of margarine (Velasco and Fernandez-Martinez, 2002; Crupkin and Zambelli, 2008). Examples of these are the "high oleic" genotypes that produce >80% of this fatty acid versus <60% in the traditional, or "high stearic" genotypes containing between 25 and 30% stearic acid (Fernandez-Moya et al., 2000) versus <10% in traditional ones. There are also genotypes containing both mutations, such as "high stearic-high oleic" (Pleite et al., 2006), which have 15% and 60% of these fatty acids, respectively (CAA, 2011). In general, genotypes with modified fatty acid composition have less percentage of linoleic acid than traditional ones. Modifications in fatty acid composition might have an effect on seed germination. It is well known that fatty acid composition of cell membranes is closely related to seed germination performance at different temperatures; i.e. increased proportion of unsaturated fatty acids such as linolenic acid enables cells to maintain membrane fluidity at low temperatures (Nishida and Murata, 1996; Murata and Los, 1997; Saeidi and Rowland, 1999). The increase in the unsaturation degree of membrane lipids is also correlated with the sustained activity of membrane-bound enzymes at lower temperature (Raison, 1973, 1980). Thus, the unsaturation of membrane lipids is considered to be one of the most critical parameters for the functioning of biological membranes and, therefore, for the survival of organisms at low temperatures (Cossins, 1994). Much less is known about the possible influence of change in the composition of reserve lipids on germination at different temperatures. Lower germination temperatures may prevent or delay the mobilization of seed reserves in germinating seeds, leading to lower germination (Nykiforuk and Johnson-Flanagan, 1994). The thermal time model has been used to characterize the effect of temperature on the germination rate (Garcia-Huidobro et al., 1982; Gummerson, 1986; Bewley and Black, 1994). This model defines three cardinal temperatures. These are: i) base temperature (Tb), temperature below which the seed cannot germinate, ii) optimal temperature (T o), at which germination occurs with the highest speed and iii) maximum temperature (Tm), for above which germination cannot occur. Thus, germination rate versus temperature has a bilineal relationship with a positive and negative slope between Tb and To and To and Tm, respectively. The cardinal temperatures vary between species (e.g. Covell et al. 1986). For example, To germination of traditional sunflower is 25ºC (Gay et al., 1991), of Cuphea is 21ºC (Berti and Johnson, 2008), of the true seed potato is 20ºC (Alvarado and Bradford, 2002), and of Jatropha curcas is 30ºC (Windauer et al., 2011). Ellis et al. (1986) suggested that Tb is constant within the same seed lot and even within the same species. However, there is evidence that the effect of temperature on the germination rate can vary between genotypes within a species. For example, Hernandez and Paoloni (1998) observed that sunflower genotypes with different linoleic / oleic acid ratio, showed different germination rate at low temperatures. These differences may be due to variations in germination rates, which change cardinal temperatures. There is preliminary evidence suggesting that the fatty acid composition of the stored lipids could modify seed germination, possibly in interaction with temperature, but there is no accurate information about it. The objective of this work was to study the effect of temperature on the germination of sunflower genotypes with modified fatty acid composition. MATERIALS AND METHODS Two experiments were performed. In Experiment 1, we used a traditional hybrid, provided by ACA (Asociación de Cooperativas Argentinas), a hybrid (HSHO_H) and a line (HSHO_L) with high stearic-high oleic composition (HSHO) and a hybrid (HSHL_H) and two lines (HSHL_L1 and HSHLO_L2) with high stearic-high linoleic (HSHL) composition provided by Advanta Semillas SAIC. In Experiment 2, three hybrids were used, one traditional, one high oleic (HO), and one HSHO, all provided by Advanta Semillas SAIC. In both experiments, seeds were incubated in 8-10 constant temperatures within 5°C - 38ºC, in darkness, using growth chambers. Three replications of 20 seeds per genotype were placed in 9 cm diameter plastic Petri dishes. Seeds were placed on absorbent paper saturated with distilled water. During 20 days, the number of seeds germinated was recorded every 12-24h (Windauer et al., 2011). A seed with a radicle of at least 3mm long was considered germinated. At the end of the experiment, tetrazolium test was performed to non-germinated seeds to assess their viability (ISTA, 1999). For each genotype, the germination rates of the fraction 30% versus temperatures were plotted (Windauer et al., 2011) and the range of sub-optimal germination was identified (between Tb and To) as the range with positive slope in the relationship. When no change point was observed, all temperatures were considered as sub-optimal range. The Tb values were determined by repeated probit regression analysis (Ellis et al., 1986). Tb data were processed by analysis of variance procedures (R, 2011). Differences in Tb among genotypes were evaluated with the Tukey test (P < 0.05). RESULTS AND DISCUSSION In both experiments, germination percentages were above 90%, except for genotype HSHO of the experiment 2 that reached only 78% germination (data not shown). At 5ºC, in both experiments, traditional genotypes presented a faster germination evolution compared to HSHO genotypes (Figure 1). At 22ºC and 30ºC minor differences were observed between genotypes; and in some cases, HSHO genotypes showed a faster germination evolution compared to traditional genotypes. Figure 1. Evolution of germination (%) for traditional and HSHO hybrids of Experiment 1 and 2 at three temperatures. Bars indicate standard error. In both experiments, the germination rate linearly increased with temperature. In Experiment 1, the maximum germination rate was observed at 27ºC for traditional genotype. This is consistent with the findings of Gay et al. (1991), where a traditional cultivar presented an optimum temperature of 25ºC. In Experiment 1, HSHO and HSHL genotypes showed a maximum germination rate between 27ºC and 38ºC. In the three genotypes in Experiment 2, the highest germination rates were observed at 34ºC. In both experiments, genotype effect was found on the base temperature of germination (p=1.55-7 and p=4.013 , for Experiment 1 and 2, respectively). Traditional genotypes presented lower T b than others genotypes (Table 1). In both experiments, no differences in T b among genotypes with modified composition were observed, except in HSHO_L which presented the highest T b. Table 1. Base temperature (Tb, ºC) for the six genotypes of Experiment 1 and the three hybrids of Experiment 2. Tb Traditional 0.57 a† Experiment 1 HSHO_H 2.02 b HSHO_L 2.70 c HSHL_H 1.75 b HSHL_L1 2.01 b HSHL_L2 2.24 bc Experiment 2 † Traditional 2.33 a HO 3.29 b HSHO 2.90 b Means with same letters indicate no significant statistical differences (p<0.05) The Tb recorded for any genotype was lower than that used by the model OilCrop-Sun (Villalobos et al., 1996). These results differ from those found by Ellis et al. (1986), who reported that different genotypes of the same species had similar base temperature. The higher Tb of seeds with modified fatty acid composition compared to traditional ones could be associated to the lower unsaturation degree of the lipids stored in the seeds as observed in other species. According to Dogras et al. (1977), chilling resistance of the seed was positively associated with unsaturation of the fatty acids in the seed of broad beans (Vicia faba L.) and peas (Pisum sativum L.). Unsaturated/saturated fatty acid ratio of seed lipids in Pima cottonseed (Gossypium barbadense L.) showed a low correlation with laboratory germination percentage, but had a strong, positive correlation with emergence under field conditions at low soil temperatures (Bartkowski et al., 1977). Further research is needed to understand the effect of the fatty acid composition of the stored lipids in the seed on the germination performance. Our results suggest that soil temperature at planting time can play an important role in obtaining an adequate stand of plants in sunflower genotypes with modified fatty acid composition. The optimum temperature was between 27ºC and 38 ºC for most of the studied genotypes. The traditional hybrids had lower Tb than genotypes with modified fatty acid composition. A higher Tb delays the emergence in early plantings or increases the exposure of seeds to fungi and insects, reducing plant stand uniformity. The causes of this higher base temperature of genotypes with modified fatty acid composition are still unknown. ACKNOWLEDGEMENT This work was supported by the Agencia Nacional de Promoción Científica y Tecnológica PICT 941 and Universidad Nacional de Mar del Plata AGR340-11. REFERENCES Aase, J.K. and Siddoway, F.H. (1980). Stubble height effects on seasonal microclimate, water balance, and plant development of no-till winter wheat. Agricultural Meteorology. 21(1): 1-20. Alvarado, V. and Bradford, K.J. (2002). A hydrothermal time model explains the cardinal temperatures for seed germination. Plant, Cell & Environment. 25(8): 1061-1069. Bartkowski, E.J., Buxton, D.R., Katterman, R.H. and Kircher, H.W. (1977). Dry Seed Fatty Acid Composition and Seedling Emergence of Pima Cotton at Low Soil Temperatures. Agronomy Journal. 69(1): 37-40. Berti, M.T. and Johnson, B.L. (2008). Seed germination response of cuphea to temperature. Industrial Crops and Products. 27(1): 17-21. Bewley, J.D. and Black, M. (1994). Seeds: Physiology of Development and Germination. Plenum Press. New York, USA. p. CAA, Codigo Alimentario Argentino. Capitulo VII - Alimentos grasos aceites alimenticios. 2011. [On line]. http://www.anmat.gov.ar/alimentos/normativas_alimentos_caa.asp. [Consult: 12/2011] Cossins, A.R. (1994). Temperature adaptation of biological membranes. London: Portland. 227p. Covell, S., Ellis, R.H., Roberts, E.H. and Summerfield, R.J. (1986). The influence of temperature on seed germination rate in grain legumes. I. A comparison of chickpea, lentil, soybean and cowpea at constant temperatures. Journal of Experimental Botany. 37: 705-715. Crupkin, M. and Zambelli, A. (2008). Detrimental Impact of Trans Fats on Human Health: Stearic Acid-Rich Fats as Possible Substitutes. Comprehensive Reviews in Food Science and Food Safety. 7(3): 271279. Dogras, C.C., Dilley, D.R. and Herner, R.C. (1977). Phospholipid Biosynthesis and Fatty Acid Content in Relation to Chilling Injury during Germination of Seeds. Plant Physiology. 60(6): 897-902. Ellis, R.H., Covell, S., Roberts, E.H. and Summerfield, R.J. (1986). The influence of temperature on seed germination rate in grain legumes. II. Intraspecific variation in chickpea (Cicer arietinum L.) at constant temperatures. Journal of Experimental Botany. 37(183): 1503-1515. Fernandez-Moya, V., Martinez-Force, E. and Garces, R. (2000). Metabolism of Triacylglycerol Species during Seed Germination in Fatty Acid Sunflower (Helianthus annuus) Mutants. Journal of Agricultural and Food Chemistry. 48(3): 770-774. Garcia-Huidobro, J., Monteith, J.L. and Squire, G.R. (1982). Time, temperature and germination of pearl millet (Pennisetum typhoides S. & H.). I. Constant temperature. Journal of Experimental Botany. 33: 288-296. Gay, C., Corbineau, F. and Côme, D. (1991). Effects of temperature and oxygen on seed germination and seedling growth in sunflower (Helianthus annuus L.). Environmental and Experimental Botany. 31(2): 193-200. Gummerson, R.J. (1986). The effect of constant temperatures and osmotic potential on the germination of sugar beet. Journal of Experimental Botany. 41: 1431-1439. Hernandez, L.F. and Paoloni, P.J. (1998). Germinación y emergencia de cuatro híbridos de girasol (Helianthus annuus) con diferente contenido lipídico y en relación con la temperatura. Investigación agraria. Producción y Protección Vegetales. 13: 345-358. ISTA (1999). International rules for seed testing. International Seed Testing Association. Zürich. 303p. Murata, N. and Los, D.A. (1997). Membrane Fluidity and Temperature Perception. Plant Physiology. 115(3): 875-879. Nishida, I. and Murata, N. (1996). Chilling sensitivity in plants and cyanobacteria: The Crucial Contribution of Membrane Lipids. Annual Review of Plant Physiology and Plant Molecular Biology. 47(1): 541568. Nykiforuk, C.L. and Johnson-Flanagan, A.M. (1994). Germination and Early Seedling Development under Low Temperature in Canola. Crop Science. 34(4): 1047-1054. Pleite, R., Martínez-Force, E. and Garcés, R. (2006). Increase of the stearic acid content in high-oleic sunflower (Helianthus annuus) seeds. Journal of Agricultural and Food Chemistry. 54(25): 93839388. Potter, K.N., Cruse, R.M. and Horton, R. (1985). Tillage Effects on Soil Thermal Properties. Soil Science Society of America Journal. 49(4): 968-973. R (2011).R: A Language and Environment for Statistical Computing. In R Development Core Team: R Foundation for Statistical Computing. Raison, J.K. (1973). The influence of temperature-induced phase changes on the kinetics of respiratory and other membrane-associated enzyme systems. Journal of Bioenergetics and Biomembranes. 4(1): 285309. Raison, J.K. (1980). Membrane lipids: structure and function. In The biochemistry of plants, (Ed P.K. Stumpf). New York: Academic:57-83. Saeidi, G. and Rowland, G.G. (1999). The effect of temperature, seed colour and linolenic acid concentration on germination and seed vigour in flax. Canadian journal of plant science. 79(3): 315-319. Velasco, L. and Fernandez-Martinez, J.M. (2002). Breeding Oilseed Crops for Improved Oil Quality. Journal of Crop Production. 5: 309-344. Villalobos, F.J., Hall, A.J., Ritchie, J.T. and Orgaz, F. (1996). OILCROP-SUN: A Development, Growth, and Yield Model of the Sunflower Crop. Agronomy Journal. 88(3): 403-415. Windauer, L.B., Martinez, J., Rapoport, D., Wassner, D. and Benech-Arnold, R. (2011). Germination responses to temperature and water potential in Jatropha curcas seeds: a hydrotime model explains the difference between dormancy expression and dormancy induction at different incubation temperatures. Annals of Botany. 108(8): 1-9.