Supplementary Methods - Springer Static Content Server
advertisement

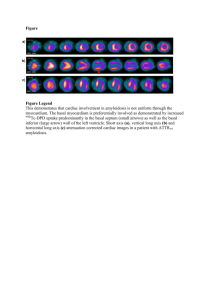
Assessment of myocardial fibrosis by endoventricular electromechanical mapping in experimental nonischemic cardiomyopathy. The International Journal of Cardiovascular Imaging Short title Electromechanical mapping in NICM Peter J Psaltis, MBBS, PhD;1,2 Angelo Carbone, BSc;1 Darryl P Leong, MBBS;1 Dennis H Lau, MBBS;1 Adam J Nelson, BMSc;1 Tim Kuchel, BVSc, MVS;3 Troy Jantzen, BSc, PhD;4 Jim Manavis, BSc;5 Kerry Williams;1 Prashanthan Sanders, MBBS, PhD;1 Stan Gronthos, BSc, MSc, PhD;2 Andrew CW Zannettino, BSc, PhD;2 Stephen G Worthley, MBBS, PhD.1 1 Cardiovascular Research Centre, Royal Adelaide Hospital and Departments of Medicine and Physiology, University of Adelaide, Australia 2 Bone and Cancer Laboratories, Division of Haematology, Institute of Medical and Veterinary Science & Centre for Stem Cell Research, University of Adelaide, Australia 3 Veterinary Services Division, Institute of Medical and Veterinary Science, Adelaide, Australia 4 Biosense-Webster, Johnson & Johnson Medical Pty Ltd, Australia 5 Hanson Institute Centre for Neurological Diseases, Institute of Medical and Veterinary Science, Adelaide, Australia Address for Correspondence 1 Stephen G Worthley Cardiovascular Investigation Unit, Royal Adelaide Hospital North Terrace, Adelaide, South Australia, Australia 5000 Telephone: +61-8-82225608 Fax: +61-8-82222454 E-mail: stephen.worthley@adelaide.edu.au 2 Supplementary Methods General anesthesia and post-procedural care Anesthetic induction was achieved by mask inhalation of isoflurane (4% in 100% oxygen), followed by endotracheal intubation and maintenance of anesthesia by inhalation of a mixture of isoflurane (2-3%) in 100% oxygen. Animals were mechanically ventilated with tidal volume of 10 ml/kg, to maintain end tidal CO2 at approximately 40 mmHg. Additional monitoring under anesthesia included pulse oximetry, limb lead electrocardiography and intra-arterial assessment of blood pressure. Post-operative care after invasive surgeries included subcutaneous administration of 1-2 mg/kg Ketoprofen for analgesia and 0.1 mL/kg Terramycin (oxytetracycline 200 mg/mL) for antibiotic prophylaxis. Cardiac magnetic resonance Prior to CMR, anesthetized sheep had the wool over their left parasternal region shaved and the skin thoroughly cleansed with warm water, detergent and ethanol, to improve electrocardiogram lead adherence. They were then positioned in dorsal recumbence inside the CMR scanner and mechanical ventilation was commenced to ensure adequate breath-holds during image acquisition. The following parameters were used for the cine sequences: repetition time (TR)/echo time (TE) 52.05 ms/1.74 ms; flip angle 70°; matrix 256x150; 25 phases per cardiac cycle; FOV 380 mm with slice thickness 6 mm and inter-slice gap 4 mm through the ventricles. Heart rate was typically 90-100 bpm (R-R’ interval 667-600 msec) during imaging and therefore the breath-hold times were between 8 and 12 sec for all animals. 3 For delayed contrast-enhanced imaging, a segmented T1-weighted inversionrecovery (turboFLASH) sequence was used with the following parameters: TR 2 cardiac cycles; TE 3.4 ms; α 25°; FOV 380 mm; acquisition matrix 256x150; typical voxel size 1.9x1.4x6 mm. The inversion delay (TI) was determined using an inversion recovery scout sequence, and was lengthened during scanning in order to maintain optimal nulling of normal myocardium. Typically, the TI was between 260 and 320 msec. Standard and phase sensitive imaging were performed, although the standard, delayed contrast-enhanced, inversion recovery gradient echo-based (FLASH) sequences were used for analyses. Ventricular ejection fractions and chamber volumes were determined in blinded fashion, with Argus software. End-diastolic and end-systolic images were chosen as the maximal and minimal, mid-ventricular, cross-sectional areas in a cinematic display. Short axis endocardial and epicardial borders were traced manually for each slice in enddiastole and end-systole. These areas were multiplied by the slice thickness (10 mm) and added together to obtain the EDV and ESV, respectively. Papillary muscles were included for the volume measurements. Care was taken not to include atrial slices at endsystole secondary to apical movement of the base of the heart during LV contraction. Ejection fraction was calculated by the formula: EF (%) = (EDV-ESV)/EDV x100. Transthoracic echocardiography Transthoracic echocardiography (Acuson XP-128, 4 MHz probe, Siemens Medical Systems, PA, USA) was performed serially during animal studies to monitor changes in LV dimensions and contractile function, especially during and after doxorubicin dosing. Images were obtained once animals had been fully anesthetized and 4 mechanically ventilated for 30 min and placed in the right lateral decubitus position. Two dimensional-guided M-mode measurements of LV end-diastolic dimension (EDD) and end-systolic dimension (ESD) were taken from right parasternal short axis views, just basal to the insertion of the papillary muscles. For each parameter, measurements from three separate consecutive cardiac cycles were averaged. Left ventricular fractional shortening (FS) was derived from the equation: FS (%) = (EDD-ESD)/EDD x100. Cardiac catheterization and hemodynamic measurements An 8 Fr sheath was used to access the left external jugular vein and a 7.5 Fr thermodilution catheter was passed for obtaining right atrial, right ventricular, pulmonary arterial and pulmonary capillary wedge pressures which were recorded in end-expiration. Cardiac outputs were obtained at end-tidal CO2 40 mmHg, from triplicate thermodilution measurements, using iced 5% dextrose boluses (10 mL). Left ventricular systolic and end-diastolic pressure measurements were obtained using a 5 Fr pigtail catheter passed retrogradely into the LV from the femoral arterial access site. Masson’s trichrome staining Sections were initially dewaxed, taken to distilled water and then stained with Celestin Blue for 5 min. After rinsing with distilled water, they were stained with haematoxylin for 5 min and then washed with running tap water for 10 min. They were then placed in Biebrich scarlet-acid fuchsin for 8 min and again rinsed in distilled water. Following this, the sections were treated with Phosphomolybdic-phosphotungstic acid 5 solution for 15 min, placed into Aniline blue solution for a further 5 min and then rinsed in distilled water. Finally, they were differentiated in 1% acetic acid for 3-5 min, then dehydrated, cleared and mounted. 6 Supplementary Figure Legends Supplementary Figure 1. Left ventricular segmentation for DE-CMR For the purpose of DE-CMR analysis, horizontal slices were used from the basal (a), mid (b) and distal LV levels. As was the case for NOGA® XP and histology, segmentation was performed by dividing the basal and mid-ventricular levels into four segments each (septum (S) 120°, anterior (A) 80°, lateral (L) 80° and posterior (P) 80°), while the distal level was analyzed in its entirety. The margins of the interventricular septum were used to guide segmental orientation. Supplementary Figure 2. Left ventricular segmentation for histology Formalin-fixed sheep hearts were sliced transversely through the left ventricle, at 1 cm intervals commencing at the apex. Bold line markings show the surfaces used for processing basal, mid and distal ventricular levels (a). Further segmentation of the myocardium was performed as per the nine-segment strategy used by NOGA® XP. The basal (b) and mid (c) ventricular levels were divided into four regions each, corresponding to the septum (S: 120°), anterior (A: 80°), lateral (L: 80°) and posterior (P: 80°) segments. The distal level was analyzed in its entirety. As for DE-CMR, the margins of the interventricular septum were used to assist with segmental orientation. 7