Time-resolved SAXS/XRD Studies of 2D
advertisement

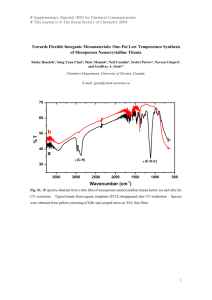
Time-resolved SAXS/XRD Studies of 2D-Rectangular c2mm Mesoporous Silica Nanoparticles Wei-Chia Huang (黃威嘉), Li-Lin Chang (張莉琳), Ching-Yi Lin (林靜儀), and Chia-Min Yang (楊家銘) Department of Chemistry, National Tsing Hua University, Hsinchu, Taiwan Most of the surfactant-templated mesoporous materials [1] display pore topologies closely related to those for the liquid-crystal-like phases typical of surfactant-water systems [1-3]. Among them, the materials with two-dimensional (2D) centered-rectangular (plane group c2mm) lattice, correlated to the ribbon-intermediate phases, may be regarded as deformed structures of the hexagonal mesophase in which the cylindrical micelles are flattened [4,5]. The elliptical channels resulted from the structural deformation are of special interest because of the additional anisotropy of the channels, but it remains a challenge to discover new synthetic strategies to control and extend the degree of the structural deformation. Here we report the synthesis of c2mm mesoporous silica nanoparticles with tunable lattice dimensions and tunable elliptical channel section. In addition, time-resolved small-angle X-ray scattering experiments were performed to study the formation mechanism of the materials. The results showed a fast hexagonal-to-rectangular structural transformation leading to the resulting c2mm mesostructures, based on which a formation mechanism of kinetically driven structural deformation has been proposed. The synthesis is envisioned to bring unprecedented opportunities to explore curvature-dependent chemistry of inclusion and surface functionalization [6,7] in the anisotropic channel space. References: [1] Kresge, C. T.; Leonowicz, M. E.; Roth, W. J.; Vartuli, J. C.; Beck, J. S. Nature 1992, 359, 710. [2] Huo, Q.; Margolese, D.; Ciesla, U.; Feng, P.; Gier, T. E.; Sieger, P.; Leon, R.; Petroff, P.; Schüth, F.; Stucky, G. D. Nature 1994, 368, 317. [3] Firouzi, A.; Kumar, D.; Bull, L. M.; Bessier, T.; Sieger, P.; Huo, Q.; Walker, S. A.; Zasadzinski, J. A.; Glinka, C.; Nicol, J.; Margolese, D.; Stucky, G. D.; Chmelka, B. F. Science 1995, 267, 1138. [4] Husson, F.; Mustacchi, H.; Luzzati, V. ; Acta Crystallogr. 1960, 13, 668. [5] Hagslätt, H.; Söderman, O. ; Jönsson, B. Liq. Cryst. 1992, 12, 667. [6] Kidder, M. K.; Britt, P. F.; Zhang, Z.; Dai, S.; Hagaman, E. W.; Chaffee, A. L.; Buchanan, III, A. C. J. Am. Chem. Soc. 2005, 127, 6353. [7] Petrov, O.; Furó, I. Phys. Rev. E 2006, 73, 011608.