Reportable Events - HCA Ethics & Compliance
advertisement

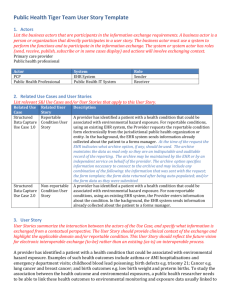
DEPARTMENT: Ethics and Compliance PAGE: 1 of 2 APPROVED: November 13, 2001 EFFECTIVE DATE: January 1, 2002 POLICY DESCRIPTION: Reportable Events REPLACES POLICY DATED: RETIRED: REFERENCE NUMBER: EC.020 SCOPE: All Company-affiliated facilities including, but not limited to, hospitals, ambulatory surgery centers, home health agencies, physician practices, patient account service centers, and all Corporate departments. PURPOSE: To establish a process for identifying potential reportable events and reporting actual reportable events to the appropriate legal and Federal health care program authorities. POLICY: The Company will report all Reportable Events related to compliance with laws and regulations relating to participation in Federal healthcare programs. Additional guidance is provided in the Correction of Errors Related to Government Reimbursement Policy, EC.012, and the Confirming and Processing Overpayments Policy, GOS.BILL.005. Definition: “Reportable Event” means any situation that involves: A substantial overpayment (an occurrence or series of occurrences exceeding $100,000); or A matter that a reasonable person would consider a potential violation of any criminal, civil, or administrative statue or regulation applicable to any Federal health care program for which criminal penalties, civil monetary penalties, or exclusion may be authorized. A Reportable Event may be the result of an isolated event or a series of occurrences. PROCEDURE: 1. Within three business days of discovering a potential Reportable Event, or as soon as possible thereafter, the following information must be gathered: a. A complete description of the relevant facts, persons involved and legal and Federal health care program programs implicated; b. A complete description of actions taken to correct the potential Reportable Event; and c. A complete description of any further action planned to address the potential Reportable Event and steps to prevent it from recurring. 2. If the potential Reportable Event is a substantial overpayment, the following action must be obtained: a. Obtain payer’s name, address, and contact person to whom the overpayment was sent; b. For Medicare overpayments identified through HCA’s Ethics and Compliance Program and/or the processes required under the CIA (including internal and IRO audits, Ethics Line cases, or other corporate-level monitoring or review), complete an Overpayment Refund Form, which is Appendix B of the Corporate Integrity Agreement (CIA) and attached hereto as Attachment A; 11/2001 DEPARTMENT: Ethics and Compliance PAGE: 2 of 2 APPROVED: November 13, 2001 EFFECTIVE DATE: January 1, 2002 POLICY DESCRIPTION: Reportable Events REPLACES POLICY DATED: RETIRED: REFERENCE NUMBER: EC.020 c. Notify the AVP, Corporate Integrity, or the Director, CIA Implementation, within three business days of discovery of a potential Reportable Event and follow up with a written report including a completed Overpayment Refund Form; and d. If the potential Reportable Event is determined to be a Reportable Event, the Director, CIA Implementation, will file a CIA Reportable Event Form with the Office of the Inspector General (OIG), U.S. Department of Health and Human Services. 3. If the potential Reportable Event is NOT a substantial overpayment (e.g., it may be a potential violation of law) the following steps must be taken: a. Notify the appropriate Operations Counsel within three business days of discovery; b. Operations Counsel will review and determine if the situation is a possible Reportable Event; c. If a potential Reportable Event is identified as a possible Reportable Event, Operations Counsel will provide a completed CIA Potential Reportable Event Form to the Director, CIA Implementation; d. The Legal and the Ethics & Compliance Departments will determine if the possible Reportable Event is an actual Reportable Event; and e. If such determination is made, the Director, CIA Implementation, will file the Reportable Event with the OIG. The Chief Executive Officer or Administrator and the Ethics and Compliance Officer of each Company-affiliated facility are responsible for the implementation of this policy. REFERENCES: Corporate Integrity Agreement, effective January 2001 Correction of Errors Related to Government Reimbursement Policy, EC.012 Processing Overpayments Policy, GOS.BILL.005 11/2001