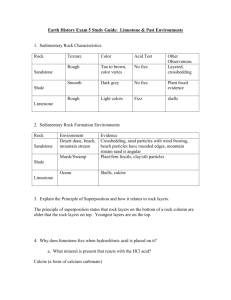
INSTRUCTIONS. - Raymond D. Letterman
advertisement

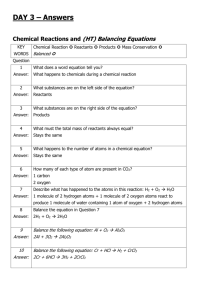
INSTRUCTIONS FOR USING DESCON A Computer Program for the Design of Limestone Contactors by Raymond D. Letterman and Sameera Kothari Department of Civil and Environmental Engineering Syracuse University Prepared as part of a research project sponsored by the United States Environmental Protection Agency Cooperative Agreement No. CR814926 Jeffrey Q. Adams, Project Officer INTRODUCTION A limestone contactor is a treatment device in which water flows through and dissolves calcium carbonate from a packed-bed of crushed limestone. Dissolution of calcium carbonate, with minimal uptake of carbon dioxide from the atmosphere, increases the pH, calcium concentration and alkalinity of the water. Limestone contactor treatment has been shown to reduce the uptake of corrosion by-products, such as lead, copper and zinc, from surfaces in household piping systems (Letterman et al., 1987). Contactors are simple, low-cost devices, that usually require minimal maintenance and are, therefore, especially suitable for corrosion control in small water supplies. The computer program DESCON was prepared to facilitate the design of limestone contactors. The program uses a kinetic model of calcium carbonate dissolution described in a paper by Letterman, Haddad and Driscoll (1991). This model assumes that the rate of dissolution is controlled by two resistances that act in series; a surface reaction that controls the release of calcium from the solid and a mass transfer resistance that controls the rate of calcium transport between the solid surface and the bulk solution. The DESCON program calculates the depth of the limestone bed required to increase the pH of the water to a user specified effluent value (the target effluent pH). Input parameters include the limestone particle size and calcium carbonate content and the influent water chemistry, temperature and superficial velocity. The following material describes the input and output parameters for the DESCON program. The significance of each parameter is briefly discussed and the units required to input the quantity to the program are given. 1 INPUT PARAMETERS Calcium concentration As the influent calcium concentration increases, the calculated equilibrium pH and incremental amount of calcium dissolved at equilibrium decrease and the depth of contactor required to reach the target effluent pH increases. The calcium concentration is entered in the program using units of either mg Ca/L or mg/L as CaCO3. Alkalinity The influent alkalinity and pH are used in the program to calculate the influent, dissolved inorganic carbon concentration and the calcium concentrations at the equilibrium and target effluent pH. The program assumes that all of the acid neutralizing capacity (ANC) of the solution is attributable to the inorganic carbon species, HCO3- and CO3=. If the influent ANC is low (<0.1 meq/L) and ionizable compounds or surfaces (non-inorganic carbon species) such as humic substances are a significant fraction of the ANC, the assumptions used to derive the model may not be met and the calculations may be in error. The alkalinity is entered in the program either in milliequivalents per liter (meq/L) or in mg/L as CaCO3. pH The influent pH is a critical parameter and should be measured very carefully. The amount of carbon dioxide dissolved in the solution should not be allowed to change before the pH is measured. The best way to avoid this is to measure the pH in situ. If this is not feasible, samples should be stored before measurement for as short a time as possible in sealed plastic containers with no headspace and at a temperature equal to or lower than the in situ value. Slight changes in the pH (e.g., 1%) due to, for example, variation in the amount of carbon dioxide in the solution, may have a significant effect on the calculated depth of the contactor. In general, as the influent pH decreases the equilibrium pH decreases and the depth of contactor required to reach the target effluent pH increases. Ionic strength The ionic strength (IS) is used to calculate activity coefficients for correcting the equilibrium constants in the chemistry part of the program. If a reasonably complete chemical analysis of the influent water is available, IS can be calculated and entered directly (in mols/L). Alternatively, the program will estimate IS using input values of either the total dissolved solids concentration (TDS in mg/L) or the conductivity (COND in μmhos/cm) and the following relationships; IS (mols/L) = 2.5x10-5 TDS IS (mols/L) = 1.6x10-5 COND 2 Temperature The temperature of the influent water is used to adjust a number of model parameters including the chemical equilibrium constants, the viscosity of the water, the surface reaction rate constant for calcium dissolution and the molecular diffusivity of the calcium ion. In general, the depth of the limestone contactor required to reach a given target effluent pH increases as the temperature of the water decreases. Therefore, in most cases, the lowest water temperature anticipated for contactor operation should be used in the design calculations. Superficial velocity The superficial velocity is the flowrate to be treated divided by the cross-sectional area of the empty contactor measured perpendicular to the direction of flow. It is also called the approach velocity and is the same as the filtration rate used in describing granular bed filters. The program requires the superficial velocity to be entered in gallons per minute per square foot (gpm/ft2). The superficial velocity determines the residence time of the fluid within the contactor and it has a limited effect on the mass transfer coefficient for calcium transport from the stone surface to the bulk solution. As the superficial velocity increases, the depth of the bed required to reach the target effluent pH increases and the cross-sectional area and volume of the bed decrease. In some cases, if the superficial velocity is high, the limestone particle diameter is small and the limestone bed is deep, the headloss across the contactor bed should be checked using an expression such as the Ergun equation. Under typical conditions, e.g., a limestone particle, volume-mean diameter of 1 cm and a superficial velocity of 4 gpm/ft2, the headloss across a 5 to 10 foot deep contactor is only several inches of water. Limestone bed porosity Experiments with packed-beds of crushed limestone with volume-mean particle diameters of 0.5 to 3.2 cm have shown that the bed porosity is approximately 0.42. A porosity of 0.42 is used in the program (in the subroutine CON). The porosity can be changed by editing and recompiling the program. Limestone particle diameter The volume-mean diameter of the limestone particles can be determined in several ways. For samples with stones large enough to be individually counted, the following is applicable. If the total weight of the sample of stones is W (in grams) and the number of stones in the sample is N, then the volume-mean diameter, d (in cm), is given by d = (6W/πNρ)0.33 where ρ is the mass density of high calcium limestone ( ~2.7g/cm3). At least 50 randomly selected stones should be counted and weighed. If the particles are too small to be effectively counted, then the mass-median diameter (dgm) and geometric standard deviation (σg) can be determined by sieving and the volume-mean diameter calculated using the following equation, 3 log d = log dgm - 3.454 log2 σg. On a cumulative particle size distribution, obtained by plotting sieve analysis results on logprobability graph paper, dgm is the size of the sieve opening at the 50th percentile (50%) and σg is the diameter at 84.1% divided by dgm. As the volume-mean diameter of the stone decreases, the surface area of the stone per unit volume of interstitial fluid increases and the depth of contactor required to reach the target effluent pH decreases. Limestone particle sphericity The effective limestone particle sphericity, ψ, is defined by ψ = 6/(Aρd) where A is the interfacial area of the limestone particles per unit mass of stone and d and ρ are the volume-mean diameter and mass density of the limestone particles. The effective sphericity is used to calculate the total area of limestone per unit volume of interstitial fluid in the bed. The mass density of high calcium limestone is approximately 2.7 g/cm3. Haddad (1986) determined that the effective sphericity of crushed limestone particles is approximately 0.8 and this value is used in the subroutine CON of the program. Changing the effective sphericity requires editing and recompiling the program. Limestone calcium carbonate content The calcium carbonate content of the limestone is used to determine the exposed area of calcium carbonate in the bed. The total area of limestone per unit volume of interstitial fluid is calculated using the bed porosity, the volume-mean particle diameter and the effective particle sphericity. The total area is then multiplied by the mass fraction of calcium carbonate in the stone to determine the exposed area of calcium carbonate. The mass fraction of calcium carbonate is simply the grams of calcium carbonate per gram of stone. If the calcium content of the stone is given as percent CaO (by mass), the CaCO3 mass fraction is approximately 0.018 x %CaO. The above method for correcting the exposed area of calcium carbonate for the purity of the stone assumes that the predominant impurity is an inert material such as quartz. If the stone contains significant amounts of dolomite (CaMg(CO3)2) or other non-calcium carbonate mineral, the effective area of the stone will be greater than what is calculated by this method. The result will be a conservative estimate of the required contactor bed depth. Calcium carbonate solubility product The solubility product of pure calcium carbonate (calcite), expressed as the negative logarithm or pKsp, is approximately 8.34 for an ionic strength (IS) of zero and a temperature (T) than the value for pure calcite. Solubility experiments suggest that pKsp = 8.81 (for IS=0 and T=25ºC) is appropriate for the calcium carbonate in a number of limestones of varying purity and 4 CaCO3 crystal structure. The default value for pKsp in the program is 8.81. Use of this value, in any case, will tend to yield conservative results. It can be replaced, if necessary, in the data input stage. Target pH A user specified, target pH for the contactor effluent is the design objective in the calculation of the required depth of the contactor. When the user is requested to input a target pH during the data input stage, the calculated equilibrium pH is displayed. The target pH selected must be less than the equilibrium pH. The target pH should be a value, e.g., 8.5, that will minimize the uptake of corrosion by-products. OUTPUT Equilibrium pH and calcium concentration The equilibrium pH is the pH when the influent solution has reached chemical equilibrium with the calcium carbonate in the limestone particles. The effluent from an infinitely deep contactor will have a pH and calcium concentration equal to the equilibrium values. The equilibrium pH decreases with decreasing influent pH and increasing influent dissolved inorganic carbon concentration and calcium concentration. For most corrosive waters, the equilibrium pH will be between 8 and 10. Ground waters with relatively low pH and high carbon dioxide concentration tend to have a low equilibrium pH and dilute surface waters acidified by strong acids tend to have a relatively high equilibrium pH. Calcium concentration and alkalinity at target effluent pH The calcium concentration at the target effluent pH is calculated by the program and used in the subroutine CON to determine the required depth of contactor. The alkalinity at the target effluent pH (alkt, in meq/L)) can be calculated using alkt = alk0 + (Cat-Ca0)/20 where alk0 is the influent alkalinity (in meq/L) and Cat and Ca0 are the calcium concentrations (in mgCa/L) at the target effluent pH and in the influent, respectively. Calculated influent dissolved inorganic carbon concentration The influent dissolved inorganic carbon concentration (DIC, in mgC/L) is calculated using the influent pH and alkalinity. The calculated influent DIC is presented in the output so it can be compared with measured values, if available. If a significant discrepancy is observed between the calculated and measured DIC concentrations, the measured influent pH should be checked. pH when effluent is equilibrated with atmospheric CO2 When the effluent from a limestone contactor is opened to contact with atmospheric carbon dioxide the pH will tend to change. In most cases the pH will decrease as carbon dioxide enters the solution. For waters with a high influent concentration of DIC, CO2 may be released 5 causing the pH to increase. The program calculates the theoretical pH of the column effluent after it has equilibrated with atmospheric carbon dioxide. The initial pH in this process is the target effluent pH. The results of this calculation will usually show that for corrosion control the contactor effluent should be kept in a closed system. The calculation assumes the partial pressure of carbon dioxide is 10-3.5 atm. Depth of contactor to reach the target effluent pH The program calculates the depth of the packed-bed of limestone required to reach the user specified target effluent pH. The depth is given in feet. ERROR ANALYSIS The DESCON program can be used to calculate the effect of random, normallydistributed analytical error in the input parameters on the calculated depth of the contactor. An effective way to use the program is to first calculate, as discussed above, the required contactor depth using the mean values of the input parameters. Then, if the designer is concerned about the effect of uncertainty in one or more of the input parameters, the error analysis part of the program can be used. The error analysis will determine how much deeper the contactor must be made to compensate for uncertainty in the magnitudes in the input parameters. The error analysis is accomplished by generating an array of random normal deviates with mean = 0 and standard deviation = 1. This array is used to calculate adjusted values of the input parameters, Xi, with the following equation, Xi = Xmi + tσi Xmi and σi are the mean and standard deviation of the input parameter X and t is one value from the array of random normal deviates. If σi is not known for a given parameter, zero should be entered. A contactor bed depth is calculated using each combination of the adjusted input parameters and the target effluent pH. After the specified number of calculations have been made, all the calculated values of the contactor depth are ranked to determine the depth that corresponds to the specified confidence level. The number of depths to be calculated and the confidence level to be used in the analysis can be changed in the program. The default value for the number of depths calculated in the error analysis is 100. However, as the number and magnitudes of the standard deviations used in this analysis are increased, the number of values in this vector should also be increased, perhaps to over 1000 when all 8 input parameters are assigned standard deviations. The required number of values must be determined by increasing the number until a relatively constant result is obtained. The confidence level used in the error analysis has a default value of 90%. A 90% confidence level means that 10% of the calculated depths are larger than and about 90% are less than the reported value. Increasing the magnitude of the standard deviations used in the error analysis increases the likelihood that one or more of the calculated values of the equilibrium pH will be less than 6 the target effluent pH. Since a contactor depth can not be calculated for this condition, the program tallies the number of these infeasible results and reports it with the output at the end of the analysis. If a significant fraction of the depth calculations are "infeasible", it is likely that the specified target effluent pH can not be achieved with a limestone contactor. EXAMPLE DESIGN CALCULATION In this example design problem, the influent water chemistry parameters and the calcium carbonate content and volume-mean diameter of the limestone particles are given. The target effluent pH (pH = 8.5) has been established by corrosion control objectives and the flowrate to be treated is 20 gpm. An effective combination of the superficial velocity and the depth of the packed-bed of limestone must be determined. In contactor design, the cross-sectional area of the contactor is the design flowrate divided by the superficial velocity. To minimize the effect of short-circuiting of fluid along the walls of the container the diameter of a columnar-type contactor should be greater than 20 times the volume-mean diameter of the limestone particles . The influent water and limestone characteristics in this example are: Influent water flowrate = 20 gpm calcium concentration = 5 mg Ca/L ionic strength = 0.01 mols/L alkalinity = 0.1 meq/L pH = 6.5 temperature = 10ºC Limestone characteristics calcium carbonate content = 90% (mass %) volume-mean particle diameter = 1 cm Using the target effluent pH of 8.5 and an assumed superficial velocity of 10 gpm/ft2, the DESCON program calculates a required bed-depth of 11.2 ft. Given the design flowrate of 20 gpm and using the assumed superficial velocity of 10 gpm/ft2, the cross-sectional area of the contactor is 20 gpm/10 gpm/ft2 = 2 ft2. For a cylindrical vessel, the diameter is, diameter = ((4x2 ft2)/π)0.5 = 1.6 feet. Since the volume-mean diameter of the limestone particles is 1 cm, a column diameter of 1.6 ft (48 cm) or greater should be sufficient to minimize the effect of short-circuiting at the walls of the column. A rule of thumb is to use a column diameter that is roughly 15 to 20 times greater than the volume-mean diameter of the stones. Maintenance of a limestone contactor can be facilitated and utilization of the calcium carbonate content of the stone can be enhanced by installing the limestone in two vessels connected in series. Since the maximum rate of calcium carbonate dissolution is at the influent 7 end of the contactor, the rate of stone shrinkage will be greatest at this location. When shrinkage of the stone begins to influence the performance of the bed, the stone in the first vessel can be replaced with fresh stone. This vessel is then re-installed as the second vessel and the other receives the influent until it is time to replace its stone. In this example, it is decided to put the packed-bed in two vessels connected in series. Each bed will be 2 foot in diameter and 5.6 feet deep. Since the cross-sectional area of a 2 foot diameter column is 3.1 ft2, the superficial velocity at the design flowrate of 20 gpm will be 20 gpm/3.1 ft2 = 6.4 gpm/ft2. With this superficial velocity and fresh limestone, DESCON predicts the effluent pH will be about 8.8. The weight of stone, M, required to construct a contactor is given by the following expression, M = A x L x (1-ε) x r where A, L and ε are the cross-sectional area, depth and porosity of the bed and r is the specific weight of the limestone. For the design example, M = 3.1 ft2 x 11.2 ft x (1-0.42) x 169 lb/ft3 = 3400 lb. PROGRAM LIMITATIONS Computer simulations by Haddad(1986) have shown that the effect of shrinkage of the limestone particles on the performance of a contactor is minimal even after several years of operation. However, when dissolution of calcium carbonate from the limestone matrix forms a "residue layer", the rate of dissolution and the performance of the contactor will decrease with time. Recent dissolution rate experiments with a rotating disk apparatus (Letterman, 1992) have shown that the formation of a residue layer is negligible when the combined amount of aluminum and iron in the stone is less than about 10 mg (mgAl+0.3 mgFe) per gram of stone. It is recommended that high purity, high calcium limestone be used for contactor applications whenever possible. OTHER ASSUMPTIONS The program uses the diffusivity of the calcium ion in calculating the overall dissolution rate constant. A review of the literature and rotating disk experiments (Letterman, 1992) suggest that a calcium ion diffusivity of 0.8x10-5 cm2/s (at 25ºC) is an appropriate assumption. This value is used in DESCON. REFERENCES Letterman, R.D., C.T. Driscoll, M. Haddad and A. Hsu (1987) "Limestone Contactors for Corrosion Control at Small Water Utilities," Final Research Report submitted to the U.S. Environmental Protection Agency, Water Engineering Research Laboratory, Cincinnati, OH 8 (EPA/600/S2-86/099). Letterman, R.D., M. Haddad, and C.T. Driscoll, "Limestone Contactors: Steady-State Design Relationships," Journal of Environmental Engineering, Am. Soc. of Civil Eng., 117:3:339-358. Letterman, R.D. (1992) "Design Tools for Limestone Contactors," Final Research Report submitted to the U.S. Environmental Protection Agency, Risk Reduction Engineering Laboratory, Cincinnati, OH (Cooperative Agreement CR814926). Haddad, M. (1986) "Modeling of Limestone Dissolution in Packed-Bed Contactors Treating Dilute Acidic Water", PhD Dissertation, Department of Civil and Environmental Engineering, Syracuse University, Syracuse, NY. 9