Name
advertisement

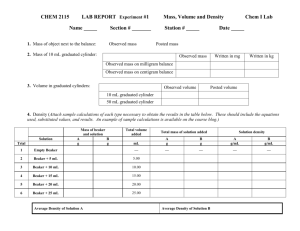
Name: ____________________ Partner: _____________________ Date: ___________________ SNC 1D Physical and Chemical Properties of household substances Introduction One way to identify matter is based on its physical and chemical properties. Physical properties are ___________________________________________________________________________________ ________________________________________. Chemical properties are ______________________ ___________________________________________________________________________________ ____________________________________________________________ . Purpose ______________________________________________________________________________ ______________________________________________________________________________ Materials and Methods Read all instructions first *IMPORTANT- LAB SAFETY* Wear safety goggles at all times! Ensure that all waste goes into the labelled waste beaker for each station. Acids are corrosive!!! Make sure that you tell your instructor if you come into contact with acid. Immediately soak that area with water. Use caution when working around open flame – ensure long hair is tied back and tuck in your tie Materials goggles wood brass water syrup iron graphite aluminum foil dry spaghetti oil sugar sand 125 mL flask rubber stopper baking soda vinegar candle scoopula small beaker waste beakers paper pieces spinach hot plate candle tongs Methods Physical properties STATION 1 – Wood and Brass Make observations about the physical properties of each. See and fill in Table 1. STATION 2 – Water and Syrup Make observations about the physical properties of each. For the viscosity test, you may use the second beaker to pour the water or syrup. Do not mix the substances. See and fill in Table 2. STATION 3 – Conductivity of water, iron, and graphite (THIS WILL BE DEMONSTRATED) See and fill in Table 3. STATION 4 – Aluminum foil and dried pasta Make observations about the physical properties of each. See and fill in Table 4. STATION 5 – Water, oil, sugar, sand. a) Rock the water bottle back and forth and record your observations about the solubility of water and oil. DO NOT OPEN THE LID. See and fill in Table 5. b) Add a small scoop of sand to the flask, close with the rubber stopper. Shake for 1 minute. Record your observations about the solubility of water and sand. Pour the mixture in the waste beaker B) and rinse the flask with tap water. See and fill in Table 5. c) Add a small scoop of sugar to the flask, close with the rubber stopper. Shake for 1 minute. Record your observations about the solubility of water and sugar. Pour the mixture in the waste beaker C). Rinse the flask with tap water. See and fill in Table 5. Chemical properties STATION 6 – Baking soda and vinegar i) ii) iii) iv) v) vi) Using the graduated cylinder measure out 10 mL of vinegar and transfer it to the beaker. Record three physical properties about the vinegar. Record three physical properties about the baking soda. Using the scoopula, add a small scoop of the baking soda to the beaker. Observe and record in Table 6. Describe the physical properties of the resulting mixture. Place the waste in the waste beaker. Rinse out the beaker with tap water. STATION 7 – Combustion of paper i) Obtain a small piece of paper ii) Record three physical properties of the paper in table 7 iii) OVER THE METAL TRAY PROVIDED, hold the piece of paper in the tongs and light it in the flame of the “tea-lite” candle *STAY WELL AWAY FROM THE FLAME KEEPING IT ABOVE THE METAL TRAY AT ALL TIMES iv) Record three physical properties of the substance formed after the paper has been burned (in table 7) STATION 8 – Cooking spinach i) ii) iii) iv) Obtain 1 leaf of spinach Record three physical properties of the spinach in table 8 Place the spinach in the beaker of hot water for 1 minute Using tongs remove the spinach from the beaker of boiling water and place it on the paper towel provided *Be careful it will be hot! v) Record three physical properties of the cooked spinach. Observations STATION 1 Table 1. _________________________________ of_________________________________________. Substance Colour Hardness State Lustre Density Wood Brass STATION 2 Table 2. _________________________________ of_________________________________________. Substance Colour State Viscosity Water Syrup STATION 3 Table 3. _________________________ of_______________________________________________. Property Conductivity Iron Graphite Water STATION 4 Table 4. _________________________________ of_________________________________________. Substance Colour Hardness State Lustre Malleability Aluminum Foil Dried plaster STATION 5 Table 5. _________________________ of_______________________________________________. Property Oil Sand Sugar Solubility in water STATION 6 Table 6. _________________________________________________________________________ Substance Observations (be specific) Before Mixing Vinegar Baking soda After mixing STATION 7 Table 7. _________________________________________________________________________ Observations (be specific) Substance Before burning After burning Paper STATION 8 Table 8.__________________________________________________________________________ Observations (be specific) Substance Before cooking Spinach After cooking Dicussion Questions 1) Which observations were qualitative and which were quantitative? Explain. _______________________________________________________________________________________ _______________________________________________________________________________________ _______________________________________________________________________________________ 2) Based on your observations, how could you identify that stations 6, 7, and 8 tested for chemical properties. (i.e. what observations indicated that they were chemical properties; see your introduction). Consider each experiment. Station 6____________________________________________________________________________ _______________________________________________________________________________________ _______________________________________________________________________________________ Station 7____________________________________________________________________________ _______________________________________________________________________________________ _______________________________________________________________________________________ Station 8_______________________________________________________________________________ _______________________________________________________________________________________ _______________________________________________________________________________________