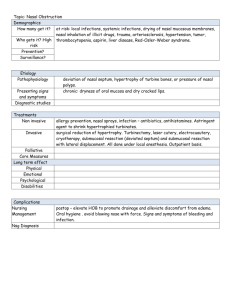
Inhalation powders for nasal and pulmonary delivery
advertisement
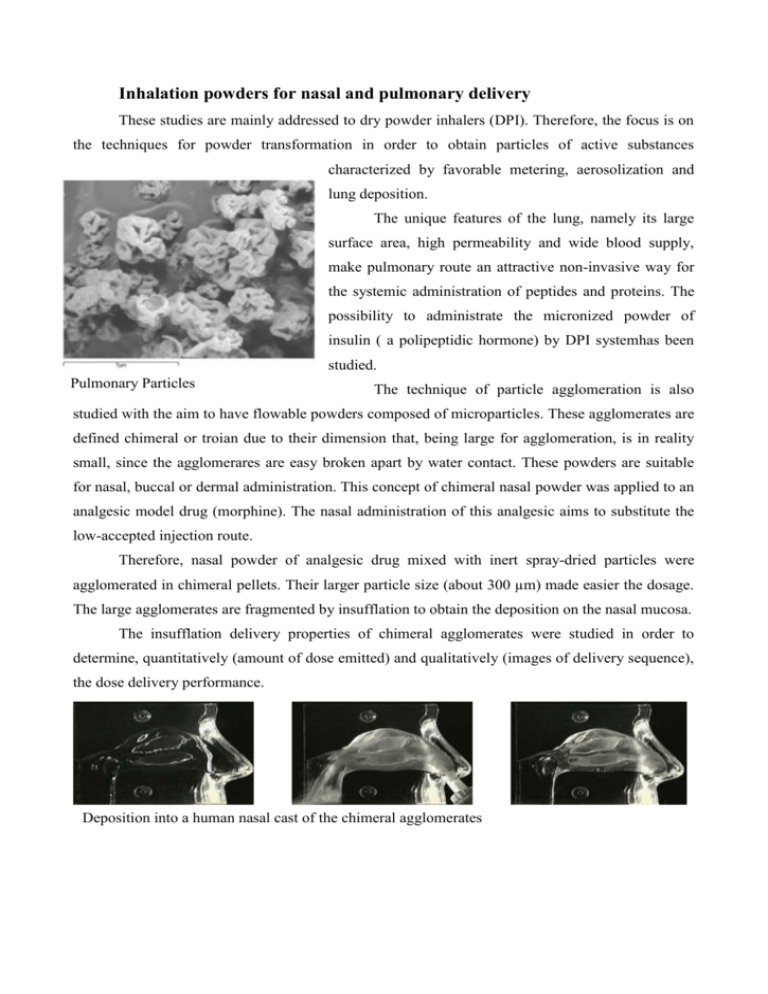
Inhalation powders for nasal and pulmonary delivery These studies are mainly addressed to dry powder inhalers (DPI). Therefore, the focus is on the techniques for powder transformation in order to obtain particles of active substances characterized by favorable metering, aerosolization and lung deposition. The unique features of the lung, namely its large surface area, high permeability and wide blood supply, make pulmonary route an attractive non-invasive way for the systemic administration of peptides and proteins. The possibility to administrate the micronized powder of insulin ( a polipeptidic hormone) by DPI systemhas been studied. Pulmonary Particles The technique of particle agglomeration is also studied with the aim to have flowable powders composed of microparticles. These agglomerates are defined chimeral or troian due to their dimension that, being large for agglomeration, is in reality small, since the agglomerares are easy broken apart by water contact. These powders are suitable for nasal, buccal or dermal administration. This concept of chimeral nasal powder was applied to an analgesic model drug (morphine). The nasal administration of this analgesic aims to substitute the low-accepted injection route. Therefore, nasal powder of analgesic drug mixed with inert spray-dried particles were agglomerated in chimeral pellets. Their larger particle size (about 300 µm) made easier the dosage. The large agglomerates are fragmented by insufflation to obtain the deposition on the nasal mucosa. The insufflation delivery properties of chimeral agglomerates were studied in order to determine, quantitatively (amount of dose emitted) and qualitatively (images of delivery sequence), the dose delivery performance. before insufflation during insufflation Deposition into a human nasal cast of the chimeral agglomerates after