Battery / Fuel Cell Vehicle Range - Michigan Technological University
advertisement

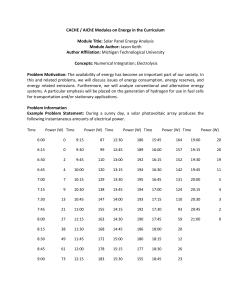
CACHE / AIChE Modules on Energy in the Curriculum Module Title: Battery / Fuel Cell Vehicle Range Module Author: Jason Keith Author Affiliation: Michigan Technological University Concepts: Energy usage for transportation Problem Motivation: The availability of energy has become an important part of our society. In this and related problems, we will discuss issues of energy consumption, energy reserves, and energy related emissions. Furthermore, we will analyze conventional and alternative energy systems. A particular emphasis will be placed on the generation of hydrogen for use in fuel cells for transportation and/or stationary applications. Problem Information Example Problem Statement: Consider a fuel cell / battery hybrid system. The fuel cell has 400 cells and the electric motor operates at 300 V. a) The vehicle consumes 2.1 kW power from the battery alone at 20 miles per hour. If the battery has 1.1 kW-hr of energy storage, determine the driving range in miles. b) The vehicle consumes 15 kW power from the fuel cell alone at 60 miles per hour. If the vehicle carries 4 kg hydrogen, determine the driving range in miles. Example Problem Solution: a. Battery energy (E, in kW-hr) is equal to the product of operation time (t, hr) and power consumption rate (P, kW): E Pt (1) As such, the time of operation is given as: t 1.1 kW-hr 0.52 hr 2.1 kW The driving range (R, mi) is equal to the product of the speed (v, mph) and the operation time (t, hr): R vt From which the driving range can be calculated as: R (20 mph)(0.52 hr) = 10.5 mi (2) b. For a fuel cell, we first need to relate the power consumption and the voltage to the current flow. The current flow will then be related to the hydrogen consumption rate. Once this is known, the vehicle range can be determined. The current (I, A) is equal to the power (P, W) divided by the voltage (V, V): I P V (3) Thus, I 15 kW 1000 W 50 A 1 kW 300 V For a hydrogen fuel cell, the current can be used to determine the hydrogen consumption rate (H2, mol/s), the number of cells (N), and Faraday’s constant (F = 96485 C/mol e–): H2 IN 2F (4) Note that in this equation, 2 represents that for every mole of hydrogen fuel (H2) there are 2 moles of electrons produced (from the anode reaction H2 → 2 H+ + 2e–). Thus, H2 mol H 2 50 A 400 cells C/s 0.104 2 mol e 96485C A s mol e mol H 2 The molecular weight (M = 2 g/mol) can be used to give the hydrogen consumption rate as: mH2 M H2 0.207 g H2 s Next, the total hydrogen supply (Mtank, g) can be used to determine the total operation time for the fuel cell (tfuelcell, s). tfuelcell M tank mH2 (5) Since the problem statement gives the hydrogen supply as 4 kg, we must first convert to 4000 g to have consistent units. Thus, tfuelcell 4000 g 19300 s 5.36 hr g 0.207 s Finally, we can use equation 2 to determine the range: R (60 mph)(5.36 hr) = 320 mi Home Problem Statement: Consider a fuel cell / battery hybrid system. The fuel cell has 375 cells and the electric motor operates at 300 V. a) The vehicle consumes 3 kW power from the battery alone at 35 miles per hour, traveling 50 miles. If the total energy in the battery is 16 kW-hr (as it is in the Chevy Volt), determine the fraction of the available energy in the battery that is consumed. Note that it is typical that only a fraction of the battery energy is used to allow for thousands of charge/discharge cycles. b) The vehicle consumes 10 kW power from the fuel cell alone for 4 hours at 40 miles per hour. Determine the fuel economy of the vehicle in miles per kg hydrogen.