205 Ex. I Mat. Harris 4th ed
advertisement

CHEMISTRY 205 LECTURE
EXAM I Material
Chapter 1
Part I.
FUNDAMENTAL CONCEPTS - Review!
I. Units
A. Mass of one mole of a substance
1. MOLAR MASS
2. FORMULA WEIGHTS is the mass of one mole of a substance.
6.02 x 1023 molecules, atoms, formula units, or ions.
*Note: Chemists frequently use molar masses and formula weights interchangeably
B. MILLIMOLES
Problems:
1. How many millimoles of chloride ions in 13.4 g of BaCl2 . 2H2O?
2. How many millimoles of CO3 are in 355 mg of Sn(CO3)2?
II. Solution Chemistry
A. Concentration
1. Molar concentration, CA
moles of solute
a. Molarity = 1 liter of solution
b. Units of Molarity
Molar concentration = 1.55 M HNO3
(1). M
mol
(2). L or mol-L-1
millimoles
(3).
mL
c. Calculation:
How many millimoles of Bromide ion in 55 ml of a 0.50 M AlBr3 solution?
Page 1
2. Analytical molarity/Formality
Gives the total number of moles of solute per 1 liter solution.(Regardless of whether or not the
species ionizes into other components". Used as a "recipe" for solution preparation.
Formality =
moles of solute before dissociation/ionization
1 liter of solution
3. Equlibrium molarity
Gives the concentration of the species actually present in solution. Takes into account ionization.
B. Percent concentration (Parts per hundred)
1. Weight-weight%, w/w%
2. Volume-volume%, v/v%
3. Weight-volume%, w/v%
C. Parts per million, ppm
D. Parts per thousand, ppt
E. Parts per billion, ppb
F. Density and Specific Gravity
Page 2
G. Preparation of solutions
1. A solid is the solute
Need: 500.0 mls of a 0.35 F K2Cr2O7 solution.
2. Dilution of a stock solution ( A more concentrated solution)
Use: M1V1 = M2V2
3. Serial Dilutions
a. Consider: A solution to be analyzed for PO43- is too concentrated.
b. A 500 ml sample is too dilute to analyze for arsenic.
Page 3
H. p-System of concentrations : Log scale
pX - -log [X]
Where X can be any species in solution:
For example:
Problems:
What is the p-value for each species in a 0.40 M CaCl2 solution
III. Stoichiometry
Problems
1. The reaction of I2O5 with BrF3 is as follows:
6 I2O5 + 20 BrF3
>
12 IF5 + 15 O2 + 10 Br2
a. How many grams of bromine can be produced from 44.0 g of BrF3?
2. The reaction of 113.4 g of I2O5 with 132.2 g of BrF3 was found to produce 97.0 og IF5.
What is the percent yield?
6 I2O5 + 20 BrF3
>
12 IF5 + 15 O20 + 10 Br2
Page 4
3. 25.60 mL of 0.01260 M Na@CrO$ is mixed with 50.00 mL of 0.150 M AgNO #.
a. How many grams of silver chromate (s) are produced?
b. Calculate the (equilibrium) molarity of all species left in solution (ie dissolved !!).
The balanced eqn.:
The total
ionic eqn
Calculation:
Page 5
4. 5.0 g of zinc are reacted with 1855 mls of 0.250 F hydrochloric acid. How many grams of hydrogen gas are
produced? Calculate the (equilibrium) molarity of all species left in solution.
The balanced eqn.:
The total
ionic eqn
Calculation:
Page 6
Chapter 3
Part II. EXPERIMENTAL ERROR
I. Background
Measurements will contain experimental error. Data must be presented so that it can be critically judged. In
quantitative analysis, the analyst must:
1. Accurately record and correctly calculate the results.
2. Calculate a single value to report, the average. Also, determine if this value is reasonable.
3. Estimate how good the results are in respect to scatter (precision).
Statistics are the mathematical tools a chemist will use to determine "how good" is the data .
PART II CLASSES OF ERROR
A. Determinate Errors {Systematic Errors}
Those errors having a definite value and an assignable cause
1. Instrument errors
2. Method errors
3. Personal errors
B. Indeterminate errors {Random errors}
These errors produce a random scatter of results.
C. Gross Errors { Careless, uncontrollable, unexpected events}
D. Significant Figures, Review
*NOTE: Keep all numbers until the end, then adjust the answer for sig. fig.
1. Addition-Subtraction
Page 7
D. Significant Figures, continued
2. Multiplication-Division
3. Logs
4. Graphs
E. Rounding, Review
1. Rounding down
2. Rounding up
3. Elimination of "5"
a. If the adjacent number is odd --> Round-up
b. If the adjacent number is even --> Round-down
Page 8
F Equipment uncertainties
1. Equipment - Every experiment has some uncertainty caused by limitations in the equipment used. This is
unavoidable error and does not reflect on your lab technique. Below are uncertainties for equipment used in the
laboratory. To reproduce measurements with only these small errors will require careful work.
Equipment
Uncertainty
“top Loading” balance
± 0.05 g
Analytical balance
± 0.0002 g
1000 mL graduate cylinder
± 2 mL
500 mL graduate cylinder
± 1 mL
100 mL graduate cylinder
± 0.4 mL
50 mL graduate cylinder
± 0.08 mL
50 mL Buret
± 0.02 mL
Barometer
± 0.1 torr
a. Absolute uncertainty
b. Relative uncertainty =
absolute uncertainty
measurement
c. Percent relative uncertainty =
absolute uncertainty
X 100
measurement
Page 9
2. Propagation of uncertainties
a. Addition and Subtraction
(1) Uncertainty
e=
e12 + e22 + e32 ...+...en2
(2) Percent relative uncertainty
percent relative uncertainty =
uncertainty
x 100
measurement
b. Multiplication and Division
(1) percent relative uncertainty
%e=
(%e1)2 + (%e2)2 + (%e3)2 ...+...(%en)2
(2) Relative uncertainty
Page 10
c. Mixed operations
e Number of Significant figures
The first uncertain figure of the answer is the last significant figure
Page 11
Chapter 4
Part III. Statistical Evaluation of Data
I. Background
1. Samples are representative of a population/Universe
2. Statistical equations are based on mathematical evaluations of data sets.
II. DISTRIBUTION OF EXPERIMENTAL RESULTS- Review
1. Mean (average) =
>
=
∑ xi
n
∑i xi = x1 + x2+ x3+ x4+.....+ xi
Where:
n = number of values
2. Median
a. For an odd number of data points the median is the middle data point.
11.2 12.3 11.4 12.1 10.9
b. For an even number of data points the median is the value above and below in which there is an equal
number of data points.
10.01 10.08 10.10 10.13
NOTE: For a good set of data the mean will approximately equal the median
3. Accuracy
Accuracy shows the closeness to the true value
a. (Absolute) error = E = Measured value - true value
Page 12
b. Relative error from the true value
Error
(1) % (pph) = true value x 100
Error
(2) ppt = true value x 1000
4. Precision
Precision shows the reproducibility of measurement
a. (1) Absolute deviation = d = |Xvalue - x|
∑d
(2) Average deviation = n
b. (1) Standard deviation (s)
s=
∑ (xi - x)2
n-1
Where:
n = number of values
x = mean
n - 1 = degrees of freedom
s
(2) Relative standard deviation ( ppt) = x x 1000
c. Variance = s2
d. Range (Spread) = w = largest value - smallest value
Page 13
5. Sample Calculations
True value = 34.37 %
% Cl
Absolute deviation
from the mean
1.
34.34
0.02
0.0004
- 0.03
2.
34.31
0.05
0.0025
- 0.06
3.
34.33
0.03
0.0009
- 0.04
4.
34.39
0.03
0.0009
+ 0.02
5.
34.43
0.07
d2= 0.0096
0.0049
+ 0.06
∑ = 171.80 ∑ d = 0.20∑
d2
n=5
a. Mean =
b. Median =
c. Relative error, in ppt =
d. Standard Deviation, s
e. RSD (relative standard deviation), ppt
f. Range
Page 14
Absolute error
III. Popuation mean, µ and sample mean , x
SampleSETS are representative of a population/Universe
IV. Standard deviation
A. S....for sample sets:
s=
∑ (xi - x)2
n-1
1. n is small
2. A representation of a population.
∑ (xi - µ)2
B.
for a population: =
n
1. n is very large
2. A true standard deviation of a population.
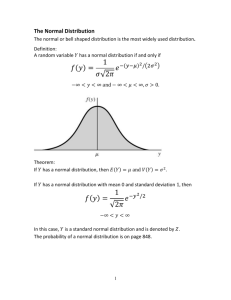
V. Normal distribution curves
Statistically,s is an estimator of .
A. Gaussian curves:
∑ (xi - µ)2
1. =
n
2. Integrals under the curve show:
68.3 % of the results are with in ± 1 of the population mean
95.5 % of the results are within ± 2 of the population mean
99.7 % of the results are with in ± 3 of the population mean
Page 15
is a good estimator of for small sets of data
Example #1
0.5
relative frequency
i
n-1
0.4
dev
0.3
0.2
0.1
-3
-2
-1
0
1
2
deviation from mean
3
Example #2
0.5
relative frequency
∑ (x - x)2
3. Statistically, s =
0.4
dev
0.3
0.2
0.1
-2
Page 16
-1
0
1
deviation from mean
2
VI. Student's t
A. Confidence Intervals,(Confidence levels, C L)
From a limited number of measurements , it is impossible to find the true population mean, µ,or the true standard
deviation, We can determine x and s, the sample mean and the sample standard deviation.
The confidence interval is an expression stating that the true population mean is likely to fall within a certain interval
from the sample mean.
.
ex. Form a Cl- analysis:
66.7 % Cl ± ________
The uncertainty would show a s that is 98%, 95.5%,90%,....etc
A. Confidence intervals
1. Degrees of Freedom = n-1
2. Confidence Interval = µ =
>
±
(t) (s)
n
See Table 4.2 on page 74 for t values
Problem: Calculate the confidence level at 95% probability for 4 samples. The known standard
deviation, s = 0.08% and XCl- = 21.70%
NOTE: With fewer measurements (n is smaller) the standard deviation increases.
B. Comparision of Confidence Intervals
_
50.00% Cl
50 % Confidence Interval
95 % Confidence Interval
99% Confidence Interval
49.00
50.00
Page 17
51.00
Case 1 Comparing a Measured Result to a "Known" Value
Case 2 Comparison of two Means (Comparing Replicate Measurements
There are many occasions when chemists must determine if two independently obtained results are
essentially the same. For this purpose we perform the t test using the following:
x1 - x2
t= s
pooled
spooled =
n1n2
n1 + n2
∑set1(xi - x1)2 + ∑set2(xj -x2)2
n1 + n2 - 2
Problem: Two chemists on two different instruments gave the following % Zn results. Is there any significant
difference of the means at the 90% confidence level between the two instruments?
% Zinc
Chemist #1 ...... 92.61,92.84,92.77,92.61,92.65,92.69
Chemist #2 ...... 93.08,92.87,92.91,93.03,93.06
VIII Rejection of Outliners, Q test
The Q test determines whether or not to reject a questionable test.
Page 18
Qexp =
|xq - xn|
w
Where:
xq = questionable result
xn = nearest neighbor
w = spread
1. Arrange results in increasing magnitude
2. Determine Q
3. Compare result with Q table
------------------------------------------------------------------------Critical Values for Rejection Quotient Q
Number of
Observations
90 %
Confidence
96 %
Confidence
99 %
Confiden
3
0.94
0.98
0.99
4
0.76
0.85
0.93
5
0.64
0.73
0.82
6
0.56
0.64
0.74
7
0.51
0.59
0.68
8
0.47
0.54
0.63
9
0.44
0.51
0.60
0.48
0.57
10
0.41
-------------------------------------------------------------------------Problem:
For the following data, determine if the outlining result can be rejected at 96 % confidence .
19.5
20.0
20.5
20.2 18.0
Page 19
Chapter 5
Finding the "Best" Straight line- Method of least Squares
This is a method for drawing the best straight line through a set of data points. By using this method, a better
fit of the straight line and a more accurate slope and intercept can be obtained.
Of the many possible straight lines that can be drawn, the "best fit" line is the one drawn minimizing the sum of
the squared deviations.
y = mx + b
y
x
Page 20
DATA
x
y
xy
x2
y2
_____________________________________________________________________________________________
1.00
2.96
5.96
1.00
8.76
2.00
5.05
10.10
4.00
25.50
3.00
7.03
21.09
9.00
49.42
4.00
8.92
35.68
16.00
79.57
5.00
10.94
54.70
25.00
119.68
_____________________________________________________________________________________________
x=15.00
y=34.90
xy=124.53
x2=55.00
y2=
282.93
Slope
Slope = m =
xy - (xy) =
nx2) - ( x)2
n
Intercept
Intercept = b =
x2)y - xy)x
nx2) - (x)2
=
EQUATION OF THE LINE
y = mx + b
Page 21
Chapter 26
Part IV Gravimetric and combustion Analysis
GRAVITMETRIC ANALYSIS
I. Background
Analysis of a sample by weight-A product of a chemical reaction of an analyte is dried and weighed. Stoichiometry
is used to determine amount of reactants.
II. Requirements for a Sucessful Gravimetric Determination
1. Product must be of known composition:
2. Product must be pure, stable and easily filtered.
3. Analyte must be completely precipitated as the product
Equilibrium must favor the product - >99.9% yield
III. Formation of Precipitate
A. Precipitation of a solute can begin when the concentration of the solute exceeds the solubility limit.
Supersaturation occurs once the solute concentration exceeds solubility limit without crystal formation (no ppt)
IV. Mechanism of Precipitation
Step. 1 - Induction Period - The time period after the solubility limit is exceeded until crystal growth
Step. 2 - Nucleation - Formation of very tiny particles of ppt called nuclei (seeds)
Step 3 - Growth - Particle growth in 3 dimensions into a lattice.
For good gravimetric analysis, large crystals are needed
V Rules for Forming Large Filterable Crystals
1. Von Weimarn found that the rate of nucleations is more dependant on relative supersaturation then particle
growth. Therefore in a highly supersaturated solution nucleation proceeds faster than particle growth and very tiny
particles (colloidal) will result.
Relative supersaturation =
(Q-S)
S
Where Q is the actual concentration of solute and S is the
concentration at equilibrium
To keep the relative supersaturation to a minimum:
a. Add precipitating reagent slowly
b. Stir continuously during reagent addition
c Dilute the precipitating agent
d Elevating the T°
* Note: After the initial burst of nucleation, the rate of crystal growth is faster that the rate of nucleation
VI Ionic Crystal Theory - Colloids
Colliods are particles which have a diameter from 1 nm to 1µm
Problem: Colloids are unfilterable!
Page 22
A. Consider the formation of AgCl
Results of colloidal formation:
1.
2.
3.
B. Coagulation of Colloids......"How to Destroy a Colloid"
Coagulation-forming larger particles from smaller colloidal particles
1. Heating/Digestion
2. Increase ionic strength (µ) by adding an electrolyte
The double layer is shrunk/destroyed with an addition of an electrolyte
C. Peptization
The coagulated precipitate reverts back to a colloidal state
1. Cause:
2. Prevention:
Page 23
VII Precipitation from Homogeneous solutions
Formation of the precipitating reagent "in situ"
Solute is formed:
1. at a dilute concentration
2. homogeneously
KEY: This gives a small Q in the Relative Supersaturation Ratio. Therefore......Large crystals are formed
Ex. 1 Formation of Fe(OH)3
Rxn. A
The Solution:
Rxn. B
VIII. Coprecipitation = Contamination/impurities
Inclusion of ions, which are normally soluble, in the lattice (PPT)
TYPES:
A. Adsorption
Trapping of ions on the surface of the lattice
Solution: Wash with HNO3. the H+ will replace the Ag+. HNO3 is volatile and is lost during
drying
B. Inclusion
Impurities are randomly occupying lattice points in the lattice matrix
C. Occlusion in Voids
Large pockets of impurities is trapped in the middle of crystal
D. Postprecipitation
IX. Digestion
X. Reprecipitating/Recrystallization
Page 24
The ppt is filtered off and redissolved into fresh solvent and reprecipitated.
XI. Composition of Product
The final product must have a known, stable composition
XII. Gravimetric calculations
A. What mass of AgI can be produced from a 0.240 g sample that assays 30.6 % MgI2
B. A sample of uranyl nitrate, UO2(NO3)2, contains an unknown quantity of water. To determine the amount of
the sample that is uranyl nitrate, a portion is ignited to produce the oxide, U3O8. It is found that 5.00 g of the
sample are ignited, 2.78 g of U3O8 are formed. Calculate the percent uranyl nitrate in the sample.
Page 25
C. A 0.1948 g sample of Ag and Cu was treated to produce a mixture of AgIO3 and Cu(IO3)2. The two Iodates
weigh 0.7225 g. What is the % by mass of the two metals?
Page 26
COMBUSTION ANALYSIS
Combustion analysis is used to determine the carbon, hydrogen, nitrogen and sulfur content in organic compounds.
Organic compounds are volatilized and oxidized to water, carbon dioxide, sulfur di and trioxide (nitrogen remains
unoxidized) and are collected selectively on weighed absorbents. The increase in weight of the absorbents gives the
component weights.
C,H -------> CO2 + H2O
and
C,H,N,S
-------> CO2 + H2O + N2 + SO2 + SO3
-------> CO2 + H2O + N2 + SO2
N2
CO
2
H2 O
Time
Page 27
SO
2