Vocabulary
advertisement

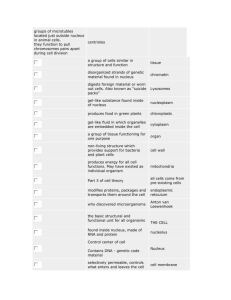
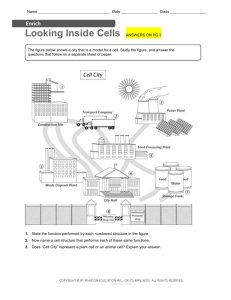
EOG Vocabulary General Dependant variable – depends on independent variable (very dependable so does not change on its own) Independent variable- “I” Change Control- compare variables to this Placebo- fake drug Hypothesis- If…then statement to predict what you think will happen Inference- come to a conclusion without proper evidence Qualitative- written data; “l” stands for “letters” Quantitative- numerical data; “n” stands for “numbers Evidence- supports your conclusion Technology- creation and use of technical means and how they interact with life Artifact- something from ancient times Mean- average Microlife Microbe- bacteria Contagion- an infection that spreads Mutagen- something that causes a genetic mutation Anti-microbial- a drug to get rid of bacteria Biotechnology- jobs that deal with the making of drugs, environmental management, and recycling Bacteria- a microorganism that can be considered good or bad Virus- an infectious agent that only replicates in a living host cell Cell wall- gives support and protection to plant and bacteria cells (police) Cell Membrane- surrounds the cell and decides what goes into and out of the cell (police or fence) Cytoplasm- jelly substance that fills up the cell (people of city) Nucleus- control center (governor) Vector- an organism that gives a disease to a bigger organism (mosquito) Host- a parasite gets its nutrients from a plant or animal host cell Carrier- an organism that carries a disease Parasite- something that lives on or in another species in order to survive Pathogen- any agent that makes diseases Carcinogen- causes cancer Multicellular- made of more than one cell Unicellular- made only of one cell Homeostasis- maintain internal stability (temperature, pH) Prokaryote- no true nucleus Eukaryote- true nucleus Photosynthesis- plants use energy from the sun to make their own food Cellular respiration- process cells use to create energy for the cell Permeable- allows substances to go through Diffusion- substances moving into and out of a cell from high concentration to low concentration Osmosis- water moving into and out of a cell Chemistry Natural substance- organic things not made from a factory or processed Synthetic substance- man-made; not found in nature Proton- positive particle found in nucleus Neutron- neutral particle found in nucleus Electron- negative particle found in energy shells around nucleus Nucleus- made of protons and neutrons in the center of atom Atom- the smallest unit of matter Atomic number- number of protons (electrons are the same number unless ion) Atomic mass- protons + neutrons Family- vertical columns on PT; families have similar characteristics Period- horizontal rows on PT Mendeleev- created first PT Mass- the amount of matter (how much of a substance) Volume- the amount of space something occupies State of matter- solid, liquid, gas, plasma Solid- particles do not move Liquid- particles are free flowing Gas- particles move quickly throughout entire area Change of state (physical change)- when a substance changes between states of matter Sublimation- solid to a gas (dry ice) Boiling point- when molecules move fast enough to go between liquid and gas Freezing point- when molecules move slow enough to go from liquid to solid Melting point- when molecules move fast enough to go from solid to liquid Compound- a mixture with more than one element (H2O, HCl) Mixture-things mixed together to create one substance Homogeneous- the same throughout Heterogeneous- can see the different parts throughout a substance Solution- one thing dissolved in another (sugar water) Specific heat- energy required to raise 1g of a substance by 1 degree celsius pH- potential of hydrogen; how acidic or basic something is Universal solvent- water Dissolved gases- gases dissolved in water Density- mass/volume Concentration- the strength of a solution Precipitate- mixing two liquids creates a solid (milk and vinegar) Malleability- able to hammer into sheets Chemical change- changing around the atoms and making a new substance (cannot go back to original state) Physical change- changing the appearance of something but keeping the same chemicals (can go back to original state) Products- right side of the equation Reactants- left side of the equation Coefficient- number in front of element or compound Conservation of matter- number of reactants must be equal to number of products of each element Reaction- mixing compounds/elements to create a new substance Dose- how much of a substance you get Exposure- how often you are introduced to the substance Potency- how strong a substance is Susceptibility- how likely you are to be affected by a substance Ductile- being able to stretch into wire Conductor- allows flow of electrons to produce electricity Metalloid- having factors of metals and nonmetals; on the staircase on PT Molecule- smallest unit of an element Crop yield- how much can be produced from a crop Food preservative- put on food so that it will last longer Hydrosphere Distribution of water on earth- saltwater97%; freshwater- 3% Local water availability- water available for us to use found mostly in groundwater and rivers Estuaries- where a river meets an ocean and is brakish River basins- land drained into a river Upwelling- cold water from the deep ocean rises to the surface bringing nutrients Groundwater- where most of our drinking water comes from Polar ice- ice frozen in the poles Glaciers- frozen freshwater; most freshwater found here Marine ecosystem- things living in the sea Terrestrial Food web- shows relations among land organisms Aquatic food web- shows relations among water organisms Cohesion- water likes itself (water going up straw) Adhesion- water likes other molecules (water going up paper towel) Polarity- molecule that has a positive and negative end Buoyancy- being able to float Hydrosphere- all of earth’s water Dissolved oxygen- the amount of oxygen in water; high temp. low oxygen & vice versa Turbidity- how many particles are in water Run-off- water that runs off land Nitrates- nitrogen found in water from fertilizers Phosphates- nutrients found in water from fertilizers Algal bloom- algae that comes from too many nutrients in water; takes oxygen out of water Point source- pollution we know the exact source (pipe) Non-point source- pollution we don’t know origination (run off) Solute- “U” dissolve Solvent- does the dissolving (usually water) Surface Tension- tightness across top of water (water on penny head) Condensation- gas to liquid (clouds) Evaporation- liquid to gas Precipitation- when clouds become too heavy and water falls as rain, snow, sleet, etc Watershed- large area around a river that brings water to that river Aquifer- water stored undergound Intertidal Zone- between high and low tide Neritic Zone- extending from low tide out Benthic Zone- lowest level of ocean Hydrothermal Vent- crack in ocean floor where many marine animals lives SONAR- uses sound to map sea floor Eutrophication- lots of nutrient build up causing fish to die Bio-indicator- animals used to monitor health of ecosystem Geographic Information Systems (GIS)used to capture and store data and create maps (google earth) Remote Sensing- finding information through satellites Ground Truthing- finding info about land by physically going there and measuring data Evolution Law of superposition- rock layers on top are younger than ones below it Natural selection- Charles Darwins theory that says the best features of an animal are passed on to the next generation Intrusion- igneous rock that juts up through other rock layers Geologic time scale- a system to measure our time periods Period- smaller than an era Era- long period of time Cambrian Boundary- time period of increase in plants and animals Ice cores- sample of ice removed from an ice sheet Index Fossil- used to identify geological periods and relative age Extinction- a species that is no longer alive Fossil Record- record of all the fossils ever found on earth Relative Age- approximate age Absolute Age- exact age Radioactive Dating- a way to use radioactive material to find the relative age of a fossil Half-life- a way to find the age of a fossil Plate Tectonics- theory that the land and water on earth sits on plates that float on magma Continental Drift- theory that all the continents have drifted apart from Pangaea Evolution- change over time Adaptation- changing based on environmnet Mutation- change in genetic make-up (always at birth) Divergent boundary- plates that separate/divide Convergent boundary- plates that push together/ converge Transform boundary- plates that slide past each other Mechanical weathering- changing the physical features (water eroding surface) Chemical weathering- changing the chemical features (rusting, turning to limestone)