SECTION B INTRODUCTION
advertisement

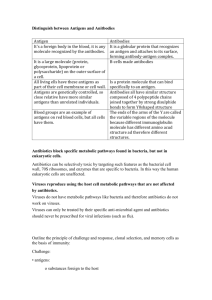
SECTION B STRAIN IDENTIFICATION INTRODUCTION Rhizobia that have dramatic differences in such important traits as host specificity, infectiveness (invasiveness) and effectiveness are indistinguishable from each other under the microscope. However, there are many circumstances in which recognition of a particular rhizobial strain and monitoring its occurrence following introduction to a soil environment is important in ecological studies. Indirect procedures are available for this purpose. Serological markers Any substance which provokes an immune response when introduced into the tissue of an animal or human is referred to as an antigen. In work with rhizobia, rabbits are commonly used for immunization and the antigens are rhizobial cell preparations. As a result of antigen injections, complex immunological reactions result in the rabbit producing special proteins called globular antibodies (immunoglobulins). These antibodies are found in the serum portion of the blood, and the study of the reactions of the immune serum with the antigens outside the animal is known as serology. Antigen-antibody reactions are highly specific in that the antibody reacts only with the antigen that elicited its formation. As in other bacteria, antigens of rhizobia can be categorized into somatic, flagellar, and capsular, depending on their derivation. Somatic antigens are closely related to the rhizobial cell-wall and usually designated by the letter "O". Some somatic antigens may be tightly bound to the cell wall, in which case they are not removed by washing of the cells; therefore, these antigens are only detected when whole cells of rhizobia react with the antibody as in agglutination or immunofluorescence. The somatic antigens that are soluble and easily removed by washing are detected by precipitation in gel, as in the Ouchterlony double-diffusion process. are also heat stable. Somatic antigens They are the most specific of the three groups of antigens. The tiny whip-like appendages (flagella) of the rhizobia are also antigenic and appropriately called flagellar or H-antigens. They are heat labile and are commonly detected by agglutination or immunofluorescence. The capsular (extracellular) antigens are surface antigens and are found outside the cell itself. They are usually designated by the letter "K." In rhizobial serology, both cultured cells and nodule antigens (bacteroids) are used for strain identification. Basic concepts on some serological methods for identification of rhizobia are described below. Agglutination. The process in which the antigens are linked together by their corresponding antibodies is called agglutination. The linked antigens may be microscopically or macroscopically visible as clumps, agglutinates or aggregates. The agglutination reaction depends on a firm structural relationship between an exposed bacterial antigen and the antibody. Linus Pauling's lattice hypothesis (Figure B.1) is the widely accepted concept for explaining the agglutination reaction. Pauling postulated that the antibody is bivalent and the antigen is multivalent, and that the antigen-antibody complexes are molded into a lattice or framework of alternating antigen-antibody particles. Figure B.1. Lattice formation in an antigen-antibody reaction Precipitin reaction. In recent years the precipitation reactions of somatic antigens have been used extensively for work with rhizobia. The precipitation reaction occurs when certain soluble antigens are brought into contact with the corresponding antibody. Precipitation differs from agglutination in that the precipitating antigens are not whole bacterial cells (cellular) but are proteins or polysaccharide molecules in solution. In the double-diffusion technique, gels, usually clarified agar, are used as matrices for combining diffusion with precipitation. The reactants simply diffuse through the gel towards each other and precipitation results when the equivalence points have been reached. Antigen preparation of a rhizobial strain will give rise to one or more lines of precipitation in the presence of the homologous antibody. When two antigens are present in a system, they behave independently of one another. The different types of precipitation reactions are illustrated in Figure B.2. Rhizobial strains that share some of or all their antigens will cross-react with respective antisera. These cross-reactions may be encountered in both agglutinations and precipitations. Immunofluorescence. Certain chemical dyes (fluorescein isothiocynate and lissamine rhodamine) have the property of fluorescing when excited by near ultraviolet light. Rhizobial antibodies developed in rabbits can be conjugated to these fluorescing chemical dyes or fluorochromes. In work with rhizobia, the chemical dye commonly used for labeling the specific antibody is fluorescein isothiocyanate (FITC) which has an apple-green fluorescence upon irradiation with blue light. In practice, a smear of rhizobial cells (cultured, or from a nodule) is made on a microscope slide, and this smear is allowed to react or "stain" with the specific antibody labeled with FITC. After appropriate washing to remove uncombined and excess labeled antibody, the smear may be viewed through a UV-microscope fitted with appropriately complementary filters, and an apple-green fluorescence of the bacterial (rhizobial) cells would mean that the antigen smear has reacted with the FITC-labeled antibody. There are two types of fluorescent antibody techniques, namely the direct- and indirect-immunofluorescence. In the direct method the specific antiserum is conjugated and is used as a "stain" in the procedure. This is different from the indirect method where the unconjugated (unlabeled) specific or primary antibody is first reacted with the antigen smear, and after sufficient time is allowed for antigen-antibody reaction, the smear is then washed free of excess antiserum. This step is followed by "staining" with the FITC-labeled secondary antibody. In serological work with rhizobia, the specific or primary antibody against the rhizobial strain is most often developed in rabbits. The secondary antibody is developed by immunization of goats or sheep with purified rabbit immunoglobulins from a previously unimmunized rabbit. Thus, the rabbit immunoglobulin serves as an antigen for immunization of the goat or sheep. Therefore, the antibody produced in the goat or sheep will not only react with the rabbit antiserum, but also with rhizobial Figure B.2. Precipitation reactions antigen with specific unlabeled rabbit antibody attached when the indirect procedure is employed. Though the results are the same, the indirect method is considered more sensitive. The indirect method requires the labeling of only the immune serum from the goat or sheep, and involves two reaction steps; but the indirect method is also known to give more nonspecific staining reactions. In the direct method, each rabbit antiserum developed against each rhizobial strain must be conjugated. The two methods are illustrated diagrammatically in Figures B.3 and B.4. Antibiotic resistance markers When high density inocula of a rhizobial strain are inoculated into media containing an antibiotic, a few cells may exhibit resistance as a result of spontaneous genetic changes or mutations. The resistance of a rhizobial strain to a particular antibiotic is a useful marker. If the mutant strain is used to inoculate a legume then nodules occupied by that strain may be identified by plating nodule isolates on media containing the respective antibiotic. The mutant rhizobial strain will grow on the antibiotic media and other bacteria will be suppressed. It is important that antibiotic-resistant mutants that are selected for inoculation experiments have not lost their infectiveness (ability to form nodules) nor their effectiveness (ability to fix nitrogen) in the symbiosis with the host plant. The symbiotic capacity of the mutant should be compared with its parent culture from time to time. The mutant should be stable throughout the steps of infection, nodulation, nitrogen-fixation and subsequent re-isolation. Streptomycin resistance is frequently used as a marker for rhizobia. Mutants resistant to this aminoglycoside are stable, have a low incidence of cross-resistance, and infrequently lose their symbiotic capacity. Besides streptomycin, spectinomycin and rifampicin have also been used. Highly resistant mutants with single or double markers (streptomycin-spectinomycin or streptomycin-rifampicin) can be obtained with one exposure of the rhizobia to low concentrations of these antibiotics or by successive selection for resistance. Figure B.3. Direct immunofluorescence Cross-resistance is a phenomenon whereby a bacterium develops resistance to a second antibiotic as a result of resistance to the first. This may happen if the antibiotics are closely related. The parallel use of antibiotic and serological markers, both relatively stable in themselves, provides a means of confirming the stability of each marker independently in ecological research with rhizobia. Compared to the serological marker techniques (fluorescent antibody, enzyme immunoassays, gel-diffusion and agglutination) the development and use of antibiotic resistant Figure B.4. Indirect immunofluorescence markers is relatively inexpensive and does not require sophisticated equipment. Bacteriophage markers: phage typing Viruses that infect bacteria (bacteriophage) were independently discovered by Twort and by d'Herelle in 1917 and 1919, respectively. Since then, the processes of infection and multiplication have been well defined. The first step involves the adsorption of the virus to specific receptors on the bacterial cell (somatic), flagella or pilli. This is followed by the injection of the viral nucleic acid into the bacteria. The nucleic acids utilize the machinery of the host cell to replicate, leading to the accumulation of several copies of the viral nucleic acid. These nucleic acids are packaged newly-synthesized viral coat protein and are then released by lysis of the host cell, liberating many infective viruses. Susceptibility of a certain bacterial strain to a particular bacteriophage forms the basis for phage-typing. One approach in a phage-typing scheme is the use of a group of phages with different host-specificities. The bacteria can then be placed into groups (lysotypes) if they are susceptible to some of the phages and not others. Through this means, bacteriophage-marked rhizobia can be indirectly traced in soil, in isolates from nodules and in laboratory experimentation.