grl29472-sup-0002-txts01
advertisement

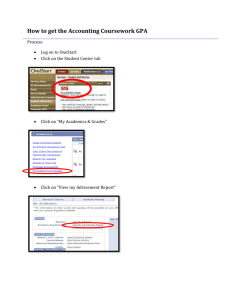
Auxiliary Materials: Tsuno et al. (2012) – Geophys. Res. Lett. vol. 39; doi: 10.1029/2012GL052606 Flux of carbonate melt from deeply subducted pelitic sediments – geophysical and geochemical implications for the source of Central American volcanic arc Kyusei Tsuno1, Rajdeep Dasgupta1, Lisa Danielson2, Kevin Righter3 1 Department of Earth Science, Rice University, 6100 Main Street, MS 126, Houston, TX 77005, USA 2 ESCG, Mail Code JE23, 2101 NASA Parkway, Houston, TX 77058, USA 3 NASA Johnson Space Center, 2101 NASA Parkway, Houston, TX 77058, USA Multi anvil experiments Experiments at 4-7 GPa and 900-1100 °C were performed using a Walker-type multi-anvil (MA) apparatus at NASA-JSC, and a COMPRES G2 cell assembly [Leinenweber et al., 2012]. The MA pressure calibration was conducted using the quartz-coesite transformation at 3.1 GPa and 1000 °C [Bose and Ganguly, 1995], Fe2SiO4 olivine-spinel transition at 5.8 GPa and 1200 °C [Yagi et al., 1987], and CaGeO3 garnet-perovskite transition at 6.1 GPa and 1000 °C [Ono et al., 2011]. Page 1 of 5 Auxiliary Materials: Tsuno et al. (2012) – Geophys. Res. Lett. vol. 39; doi: 10.1029/2012GL052606 Supplementary Table 1: Compositions of the sedimentary starting materials (in wt.%) SiO2 TiO2 Al2O3 FeO* MnO MgO CaO Na2O K2O P 2O5 H2O CO2 Mg# Total HPLC3a 54.0 0.7 15.0 7.3 0.2 2.9 9.6 2.2 2.0 - 1.0 5.0 41.9 100.0 HPLC4a 54.3 0.7 15.0 7.4 0.2 3.0 9.7 2.2 2.0 - 0.5 5.0 42.0 100.0 HPLC2b 60.0 0.6 12.5 6.1 0.2 2.5 8.0 2.2 2.0 - 1.0 5.0 42.1 100.0 TS08, GS11c 47.6 - 22.8 9.2 - 2.0 6.8 2.4 3.6 - 1.1 4.8 28.1 100.0 10GC-90GDMd 51.5 0.5 10.7 5.4 0.2 2.1 6.9 1.9 1.7 0.1 14.6 4.3 41.5 100.0 a Fluid present (HPLC3) and fluid-absent (HPLC4) starting compositions used in this study, which simulate bulk compositions from which siliceous water vapor has been extracted in shallow part of subduction. b The starting composition used in Tsuno and Dasgupta [2012], which simulates significant dehydration of 10GC-90DGM during shallow part of Central American subduction, on a volatile-free basis, is also similar to the bulk composition of the sediment columns that subduct at the trenches of Vanuatu, E. Sunda, and Makran (see Figure 1 of Tsuno and Dasgupta [2012]). c The starting composition used in Thomsen and Schmidt [2008] and Grassi and Schmidt [2011a, b], which is similar to the alumina-rich, Fe-calcareous clay composition that subducts at the trench of Lesser Antilles [Plank and Langmuir , 1998]. d The model composition, a mixture of 10 wt.% Guatemalan carbonated (GC) unit and 90 wt.% Guatemalan diatomaceous mud (GDM) unit that subducts at the Central American trench (see Plank and Langmuir [1998]). Supplementary Table 2. Phase compositions from partial melting experiments on HPLC3 and HPLC4 at 5-7 GPa a Run No. P (GPa) T (°C) Phases (HPLC3: pelite + 1 wt.% H2O and 5.0 wt.% CO2) BJJB156 5 900 cpx garnet phengite calcitess BJJB176 5 950 cpx garnet phengite calcitess carbonate melt BJJB155 5 1000 cpx garnet phengite carbonate melt BJJB178 5 1050 cpx garnet BJJB157 BJJB177 5 7 1100 1000 Na2O-calcc n SiO2 TiO2 Al2O3 FeO* MnO MgO CaO Na2O K2O CO2 H2O Total 4 4 4 5 5 4 4 5 4 4 5 2 3 6 5 59.2(2) 40.2(2) 54.3(7) 0.08(2) 57.3(7) 39.6(3) 52.3(7) 0.07(7) 0.61(29) 57.5(9) 40.0(3) 52.6(5) 0.42(18) 55.9(8) 39.7(2) 0.17(3) 0.48(4) 0.90(3) 0.04(1) 0.24(4) 0.65(6) 2.0(5) 0.02(2) 0.19(5) 0.32(9) 0.68(9) 2.6(5) 0.20(12) 0.38(8) 0.47(4) 20.8(3) 22.3(1) 24.9(6) 0.02(2) 20.0(3) 22.2(1) 23.7(10) 0.04(4) 0.92(5) 19.1(5) 22.7(1) 25.0(2) 0.53(13) 18.1(4) 22.6(3) 1.3(1) 17.3(5) 1.9(1) 6.3(5) 1.6(1) 17.8(3) 2.4(3) 0.13(3) 9.3(14) 2.2(1) 16.4(5) 2.1(1) 7.8(4) 2.6(2) 17.0(3) 0.06(4) 0.79(5) 0.00(1) 0.05(5) 0.03(1) 0.86(3) 0.01(1) 0.02(3) 0.32(17) 0.06(4) 0.77(4) 0.04(1) 0.28(3) 0.01(4) 1.0(2) 3.00(2) 5.23(2) 4.2(1) 9.8(5) 3.7(1) 5.3(1) 3.8(2) 0.00(0) 5.4(1) 3.8(2) 4.7(2) 3.2(1) 3.5(5) 3.8(1) 5.2(2) 6.2(2) 14.8(4) 0.13(7) 39.1(5) 6.6(2) 15.2(3) 0.13(3) 55.6(6) 34.9(6) 8.5(2) 16.0(7) 0.06(2) 36.2(3) 9.6(1) 15.8(1) 11.0(3) 0.16(3) 0.08(1) 0.01(1) 10.6(4) 0.21(2) 0.07(2) 0.06(3) 0.81(10) 9.0(3) 0.20(3) 0.13(2) 6.0(4) 8.4(1) 0.18(6) 0.03(1) 0.04(4) 11.1(3) 0.05(4) 0.12(6) 0.12(4) 10.8(2) 0.12(4) 5.6(20) 0.13(5) 0.04(5) 11.1(5) 3.2(6) 0.04(2) 0.01(1) 45.2(4) - 4.51(1) 101.6 101.3 102.0 100.7 100.2 101.9 99.6 99.7 58.0 100.5 101.6 101.4 58.2 98.8 102.0 silicate meltb carbonate melt cpx garnet 8 2 7 7 55.3(7) 0.37(20) 56.0(4) 38.9(2) 2.1(6) 2.1(2) 0.37(5) 0.68(11) 10.3(2) 0.33(21) 19.7(4) 22.3(2) 8.7(2) 6.5(14) 2.4(2) 16.5(2) 0.28(4) 0.25(1) 0.03(3) 0.76(4) 1.6(1) 2.5(6) 3.4(2) 5.0(1) 11.2(1) 39.8(5) 8.7(3) 16.1(3) 3.2(1) 0.63(12) 8.7(2) 0.20(3) 7.4(4) 1.5(6) 0.04(2) 0.01(1) silicate meltb carbonate melt cpx garnet carbonate melt 8 2 7 7 3 51.4(3) 0.48(2) 57.0(2) 38.9(2) 1.9(15) 3.2(1) 3.0(2) 0.3(1) 0.95(3) 1.1(1) 11.9(2) 1.3(3) 20.5(4) 22.1(2) 0.97(33) 5.0(1) 8.3(1) 2.2(1) 16.8(1) 7.5(15) 0.13(3) 0.25(1) 0.05(3) 1.7(1) 0.25(10) 1.7(0) 3.2(2) 2.7(2) 2.6(3) 4.6(11) 13.5(2) 37.9(4) 6.1(3) 16.7(1) 34.3(18) 1.9(2) 0.47(4) 9.8(3) 0.13(2) 2.7(12) 11.2(3) 0.76(2) 0.06(1) 0.06(0) 5.9(10) 79.3 55.6 98.6 99.9 58.2 4.7 6 6 5 7 7 4 5 2 6 9 8 3 58.1(9) 39.9(1) 66.1(4) 52.0(4) 0.03(1) 57.5(4) 38.4(3) 52.4(3) 0.03(1) 57.0(4) 39.7(2) 0.98(73) 0.12(2) 0.36(4) 0.00 0.92(4) 0.02(2) 0.20(2) 0.64(22) 2.5(2) 0.03(3) 0.26(5) 0.62(12) 3.0(2) 20.8(4) 22.6(2) 18.44(5) 23.4(0) 0.14(3) 20.1(5) 21.9(1) 25.3(4) 0.14(1) 19.3(4) 22.6(2) 1.3(3) 1.4(1) 18.8(6) 0.15(2) 2.0(1) 8.2(7) 2.2(1) 21.0(12) 2.1(1) 5.3(4) 2.4(2) 17.1(3) 8.3(1) 0.04(3) 0.83(8) 0.00(0) 0.00(0) 0.32(4) 0.07(2) 1.1(1) 0.02(3) 0.3(4) 0.07(3) 0.82(3) 0.41(2) 2.9(2) 5.6(3) 0.00(0) 4.2(1) 11.2(3) 3.2(4) 4.1(2) 2.4(3) 7.2(1) 4.9(2) 6.1(5) 3.2(2) 5.4(4) 13.9(5) 0.13(1) 0.19(17) 35.1(5) 6.2(5) 12.1(10) 0.06(1) 42.0(6) 7.2(2) 14.5(7) 25.4(3) 11.2(2) 0.12(2) 0.31(10) 0.07(4) 0.13(4) 10.1(4) 0.23(5) 0.17(6) 0.13(4) 9.9(2) 0.19(3) 0.47(4) 0.12(2) 0.07(1) 16.6(14) 11.1(2) 0.18(7) 0.10(3) 0.19(5) 11.0(2) 0.22(6) 0.14(11) 0.03(2) 18.3(7) 100.1 102.1 101.7 98.4 100.0 99.6 99.6 100.4 99.3 101.1 101.6 61.3 7.2 (HPLC4: pelite + 0.5 wt.% H 2O and 5.0 wt.% CO2) BJJB198 5 900 cpx garnet K-feldspar phengite calcitess BJJB194 5 950 cpx garnet phengite calcitess BJJB193 5 1000 cpx garnet carbonate melt a Numbers in parentheses are one standard deviation in terms of last digit(s). For example, 59.2(2) should be read as 59.2 ± 0.2 wt.%. b Silicate melt compositions are reported on a volatile-free basis. The microprobe total is also shown. c Na2O contents of carbonate melts based on the mass balance calculations [Yaxley and Green , 1996] Page 2 of 5 4.5(1) 43.7(4) - 4.5(2) - - - - 4.5(1) 44.7(4) 4.5(1) 44.0(2) 9.7 11.4 84.6 53.9 99.3 100.3 Auxiliary Materials: Tsuno et al. (2012) – Geophys. Res. Lett. vol. 39; doi: 10.1029/2012GL052606 Figure S1. (a) Jadeite (NaAlSi3O8) and (b) Calcium-Tschermak component in clinopyroxene (cpx) as a function of temperature from 5 GPa melting experiments of carbonated pelite. Solid circles and squares (in red) are data from our lower-alumina bulk compositions HPLC3 and HPLC4, respectively and open diamonds are data from high aluminous bulk composition of Thomsen and Schmidt [2008] (at 5.0 GPa) and Grassi and Schmidt [2011]. Higher alumina bulk composition results in cpx with higher aluminous components at a given P-T condition which in turn elevate bulk partition coefficient of Na between pelite and carbonatitic melt, thereby minimizing the influence of Na in stabilizing a carbonate melt. Page 3 of 5 Auxiliary Materials: Tsuno et al. (2012) – Geophys. Res. Lett. vol. 39; doi: 10.1029/2012GL052606 Figure S2. Estimated (a) Na2O (wt.%), (b) K2O (wt.%), and (c) total alkali concentrations of carbonatite melt as a function of temperature from 5 GPa melting experiments of carbonated pelite. The symbols used here are the same as those in Figure S1. The higher alkali contents of carbonatite melt at lower temperatures in our study are caused by the lower bulk partition coefficient of Na between pelite and carbonatitic melt, which in turn is the result of stability of less aluminous cpx in our lower alumina bulk compositions. References Bose, K., and J. Ganguly (1995), Quartz-coesite transition revisited: Reversed experimental determination at 500-1200 °C and retrieved thermochemical properties, Am. Mineral., 80, 231–238. Grassi, D and M. W. Schmidt (2011), The Melting of Carbonated Pelites from 70 to 700 km Depth, J Petrol., 52, 765–789. Leinenweber, K. D., J. A. Tyburczy, T. G. Sharp, E. Soignard, T. Diedrich, W. B. Petuskey, Y. Wang, and J. L. Mosenfelder (2012), Cell assemblies for reproducible multi-anvil experiments (the COMPRES assemblies), Am. Mineral., 97, 353–368. Ono, S., T. Kikegawa, and Y. Higo (2011), In situ observation of a garnet/perovskite transition in CaGeO3, Phys, Chem. Miner., 38, 735–740. Thomsen, T. B., and M. W. Schmidt (2008), Melting of carbonated pelites at 2.5-5.0 GPa, silicate-carbonatite liquid immiscibility, and potassium-carbon metasomatism of the mantle. Earth Planet. Sci. Lett., 267, 17–31. Yaxley, G. M., and D. H. Green (1996), Experimental reconstruction of sodic dolomitic carbonatite melts from metasomatised lithosphere, Contrib. Mineral. Petrol., 124, 359–369. Page 4 of 5 Auxiliary Materials: Tsuno et al. (2012) – Geophys. Res. Lett. vol. 39; doi: 10.1029/2012GL052606 Yagi, T., M. Akaogi, O. Shimomura, T. Suzuki, and S.‐I. Akimoto (1987), In Situ Observation of the Olivine‐Spinel Phase Transformation in FeSiO Using Synchrotron Radiation, J. Geophys. Res., 92, B7, doi:10.1029/JB092iB07p06207. Page 5 of 5