SOL REVIEW QUESTIONS
advertisement

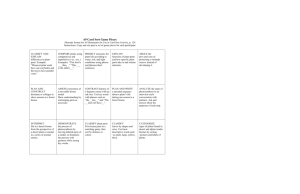
3SOL REVIEW QUESTIONS - SHORT ANSWER 1. What are the four planets closest to the Sun? Mercury, Venus, Earth, Mars 2. What are the four outer planets? Jupiter, Saturn, Uranus, Neptune 3. What is used to be the last planet in our solar system? Pluto 4. What is the largest planet? Jupiter 5. What is the smallest planet? Mercury 6. What is the coldest planet? Neptune 7. What is the warmest planet? Mercury 8. What is found between Mars and Jupiter? Asteroid Belt 9. What do you call a planet’s trip around the Sun? Revolution 10. How long does it take Earth to make a revolution? One Year 11. What is it called when a planet spins on its axis? Rotation 12. How long does it take Earth to make a rotation? One Day 13. What causes day and night on the Earth? Earth’s Rotation 14. What time is it when the Sun is directly overhead? Noon 15. What causes the seasons on the Earth? Tilt of the Earth’s Axis 16. The N. Hemisphere is tilted towards the Sun. What season are we having? Summer 17. The N. Hemisphere is tilted away from the Sun. What season are we having? Winter 18. What causes the tides on the Earth? Moon’s gravity 19. How often do we have high tide during a day? Twice 20. Are the tides stronger or weaker if the moon and Sun are aligned? Stronger 21. Name the eclipse when you can’t see the Sun. Solar 22. Name the eclipse when you can’t see the moon. Lunar 23. What instrument do astronomers use to see outer space? Telescope 24. What causes the phases of the moon? Moon’s Revolution 25. How long does it take the moon to complete its phases? About one month 26. What measures the amount matter in an object? Mass 27. What measures the amount of space an object takes up? Volume 28. What measures the matter in a given space? (Mass divided by Volume) Density 29. What does gravity determine? Weight 30. Do you weigh less on the Earth or moon? Why? Moon, less gravity 31. What number is the metric system based on? 10 32. One kilogram equals how many grams? 1000 33. Seconds measure? Time 34. Degrees Fahrenheit measures? Temperature 35. Kilometers and centimeters measure? Distance 36. Kilograms and milligrams measure? Mass 37. Milliliters, Liters, and Cubic centimeters measure? Volume 38. What does g/ml (grams per milliliter) or g/cm3 (grams per cubic centimeter) measure? Density 39. A stopwatch measures? Time 40. A thermometer measures? Temperature 41. A metric ruler measures? Distance 42. A graduated cylinder measures? Volume 43. A triple beam balance measures? Mass 44. Length x Width x Height measures? Volume of a solid 45. Mass divided by volume measures? Density 46. There’s 3 mL of water in a graduated cylinder. The water level rises to 5 mL when a ring is dropped in. What is the volume of the ring. 2 mL 47. What is an “if-then” statement or an educated guess to the solution of a problem? hypothesis 48. What group is used to test the hypothesis? Test group 49. What group is used for a comparison? Control Group 50. What are things that do NOT change in an experiment called? Constant 51. What are things that do change in an experiment called? Variables 52. How many variables should you have when you start an experiment? One INDEPENDENT OR DEPENDENT VARIABLE (53-59) 53. Located on the x-axis? Independent 54. Located on the y-axis? Dependent 55. Located after “if” in a hypothesis? Independent 56. Located after “then” in a hypothesis? Dependent 57. It is the first thing to change in an experiment? Independent 58. It is sometimes called the cause? Independent 59. It is sometimes called the effect? Dependent An experiment is testing how the height of a ramp effects the distance a car will roll. (60-62) 60. What is the independent variable? Height of the ramp 61. What is the dependent variable? Distance a car will roll 62. Should you use the same car or different cars? Same car 63. What is the purpose of repeating an experiment? Prove or verify your results, reduce errors 64. What graph measures changes over time? Line 65. What graph measures percents? Pie 66. What graph measures total amounts or averages? Bar 67. Resources limited in their supply are called? Nonrenewable 68. Resources not limited in their supply are called? Renewable 69. Identify a nonrenewable resource. Fossil Fuels 70. Identify a renewable resource. Wood, Wind, Water, Sun, Etc 71. What is the major source of pollution in the United States? Burning Fossil Fuels 72. Where do fossil fuels come from? Decomposed plants and animals over millions of years 73. Give an example of a fossil fuel. Gasoline, Oil, Petroleum, Coal 74. What are changes over time in order to survive? Adaptations 75. What is the process where animals select mates with traits best suited for survival? Natural Selection 76. What term describes when a species completely dies out? Extinction Complete each statement about the cell theory. 77. All cells come from _____________. Other cells 78. Cells can carry out all of _____________. Life’s functions 79. All _____________ are made of cells. Living things Identify the parts of the cell. (80-89) 80. Creates protein. Ribosomes 81. Transports things throughout the cell. ER (Endoplasmic Reticulum) 82. Store food, water, and waste. Vacuoles 83. Powerhouse of cell; supplies energy. Mitochondria 84. Control center of the cell. Nucleus 85. Jelly-like substance. Cytoplasm 86. What two things are found only in plant cell? Chloroplast (Chlorophyll) and Cell Wall 87. Absorbs sunlight in plants? Chloroplast (Chlorophyll) 88. Protects plant cells? Cell Wall 89. Protects plant and animal cells plus allows things to move in and out of the cell? Cell Membrane 90. What can plant cells do that animal cells can’t? Make their own food 91. What is the process where new cells are created through cell reproduction? Meiosis 92. What is the process where new cells are created through cell division? Mitosis 93. Groups of cells create? Tissues 94. Groups of tissues create? Organs 95. Groups of organs create? Systems 96. What is the process where plants create their own food? Photosynthesis 97. What source of energy is needed for photosynthesis? Sunlight 98. What two resources are the reactants during photosynthesis? Water and Carbon Dioxide 99. What two resources are the products of photosynthesis? Glucose and Oxygen 100. What do plants use glucose for? Food 101. What is the two most important reasons photosynthesis is necessary for life on Earth? Food and Oxygen 102. Who is the Father of Genetics? Mendel 103. Identify a characteristic you inherited. Eye color, Hair color, Nose shape, etc. 104. Identify a characteristic determined by your environment. Personality, Intelligence, Language Skills 105. Your individual traits are called? Genes 106. What creates DNA and contains all of your individual traits? Chromosomes 107. What is your complete genetic code called? DNA 108. What word describes the strong trait? Dominant 109. What word describes the weak trait? Recessive 110. What is the name of the chart that shows possible phenotypes and genotypes? Punnett Square 111. What is the basis of all food chains? Producers (Plants) Identify each as a producer, consumer, or decomposer. (112 – 114) 112. A grasshopper is a _____________. Consumer 113. Wheat is a _____________. Producer 114. A bacteria spore is a _____________. Decomposer 115. Contrast a predator and prey. Predator eats Prey 116. Give an example of a predator – prey relationship. Ex. Wolf eats Rabbit 117. Give an example of a producer – consumer relationship. Ex. Rabbit eats Grass 118. What does a herbivore eat? Plants 119. What does a carnivore eat? Meats 120. What does omnivore eat? Plants and Meat Identify each symbiotic relationship. (121-123) 121. One thing benefits while the other is harmed. (Tick on a Dog) Parasitism 122. One thing benefits while the other isn’t effected. (Vultures feed on the scrapes left by a Lion) Commensalism 123. Both things benefit. (Sharks are cleaned by a smaller fish) Mutualism You’re a raccoon. What will happen to your population in each case below? (124131) 124. If the size of the habitat increases? Population increases 125. If the size of the habitat decreases? Population decreases 126. If the number of wolves decrease? Population increases 127. If the number of wolves increase? Population decreases 128. If the food supply decreases? Population decreases 129. If the food supply increase? Population increases 130. If the number of squirrel (competition) increase? Population decreases 131. If the number of squirrel (competition) decrease? Population increases 132. What is the warmest biome? Desert 133. What is the coldest biome? Tundra 134. What biome gets the most rain? Rainforest 135. What biome gets the least rain? Desert 136. What biome do the animals need to be able to run fast? Grassland 137. What biome do animals and plants need to store water? Desert 138. What biome do plants have very large leaves? Rainforest 139. What biome is white fur an advantage? Tundra 140. What biome is being able to climb or fly important? Rainforest 141. What biome would you find trees with needles and cones? Coniferous Forest (Taiga) 142. What biome would you find oak and maple trees? Deciduous Forest 143. What biome is large feet an advantage? Tundra 144. Are there more plants or animals in the grassland or rainforest? Why? Rainforest,more rain & stable climate 145. What biome would you find corn and wheat growing? Grassland 146. What is an abiotic factor? Nonliving factor 147. Give an example of an abiotic factor. Rock, water, climate, temperature, etc. 148. What is a biotic factor? Living factor 149. Give an example of a biotic factor. Trees, animals, cells, etc. 150. The flow of electrons creates? Electricity 151. What is created when domains align in certain metals? Magnetism 152. What are the two ways you can strengthen an electromagnet if the battery is kept the same? Bigger nail, more coils of wire around the nail 153. What do opposite charges or poles do? Attract 154. What do like charges or poles do? Repel 155. A positive and negative will? Attract 156. A North pole and North pole will? Repel 157. Where is a magnet the strongest? Poles 158. What is a circuit with only one path called? Series 159. What is a circuit with more than one path called? Parallel 160. Which circuit will shut off if there is one break in the circuit? Series 161. A parallel circuit will still work as long as electricity can do what? The current can travel from and to the battery or the circuit is complete 162. What is a current that flows back and forth? Alternating 163. What is a current that flows in one direction? Direct 164. What is the unit for electric power? Watts 165. What is the unit for electric energy consumed? KWh (kilowatt hours) 166. What is the energy (juice) of the current measured in volts? voltage 167. What is an example of magnetism creating electricity? Hydroelectric plant (Hoover Dam) or Generator 168. Can sound or light waves travel in outer space? Light 169. What is an example of longitudinal wave? Sound 170. What is an example of transverse wave? Light 171. What is the top of a light wave? Crest 172. What is the lowest point of a light wave? Trough 173. What is the distance from the line of origin to the crest or trough in a light wave? Amplitude 174. What is the pressed together portion of a sound wave? Compression 175. What is the section where a sound wave spreads apart? Rarefaction 176. What is the distance between two similar parts of a wave? Ex. Crest->Crest, Trough->Trough, Compression->Compression Wavelength 177. What is it called when a wave bounces? Reflection 178. What is it called when a wave bends as it changes speeds? Refraction 179. What is it called when sound waves are used to locate objects under the water? Sonar 180. What is it called when animals use sound waves to locate things? Echolocation 181. What wave property determines intensity (loudness)? Amplitude 182. What wave property determines pitch? Frequency 183. What two things will happen to the particles in a substance when it is heated? Move faster & farther apart 184. What two things will happen to the particles in a substance when it is cooled? Move slower & closer together 185. What is the coldest possible temperature when there is no kinetic energy called? Absolute zero 186. Heat transfer by contact is called? Conduction 187. Heat transfer by fluids is called? Convection 188. Heat transfer by the Sun to the Earth is called? Radiation Identify the phase change. (189 – 193) 189. Solid changes to a Liquid. Melting 190. Liquid changes to a Gas. Boiling, Evaporation, Vaporization 191. Gas changes to a Liquid. Condensation 192. Liquid changes to a Solid. Freezing 193. Are phase changes physical or chemical changes? Physical Identify the state of matter. (194-196) 194. Has shape and volume. Solid 195. Has volume, but has no shape. Liquid 196. Has no shape or volume. Gas 197. Why does an object appear to be blue? It reflects blue light 198. How do you remember the order of colors in the electromagnetic spectrum? ROY G BIV 199. What do you call an object that you can’t see through? Opaque 200. What do you call an object that you can see clearly through? Transparent 201. What do you call an object that produces a blurry image? Translucent 202. What do you call a push or pull? Force 203. In order for you to do work what two things must happen? Force & Motion (Movement) 204. What is Newton’s 1st Law? Law of Inertia 205. What is Newton’s 2nd Law? Force = Mass x Acceleration 206. What is Newton’s 3rd Law? For every action there is an equal and opposite reaction 207. A ramp or screw is what type of simple machine? Inclined Plane 208. What type of simple machine is used to operate a flagpole? Pulley 209. What type of simple machine is a rake, scissors, and see-saw? Lever 210. What type of simple machine always contains a fulcrum? Lever 211. What is a device made of more than one simple machine? Compound Machine 212. Give an example of a compound machine. Bike, car, pencil sharpener, etc. 213. Why can’t Mechanical Efficiency ever be 100%? Friction or Heat 214. What does meters per second or kilometers per hour measure? Speed 215. What does velocity tell you that speed does not? Direction 216. What is the ability to do work? Energy 217. What is the metric unit for energy and work? Joules 218. What is the metric unit for force? Newton 219. What is anything that has mass and takes up space? Matter 220. What is the basic building block for all matter? Atoms 221. What is pure substance made of only one kind of atom? Elements 222. What are two or more elements chemically combined? Compounds 223. What are two or more substances physically combined? Mixtures 224. When an acid reacts with a base you get? Salt and Water Identify each as an acid, base, or neutral. (225-237) 225. pH of 7. Neutral 226. pH 0-6. Acid 227. pH 8-14. Base 228. Turns litmus blue. Base 229. Turns litmus red. Acid 230. Has no effect on litmus. Neutral 231. Tastes bitter. Base 232. Tastes sour. Acid 234. Starts with H. Acid 235. Ends with OH. Base 236. Feels slippery. Base 237. Burns your skin. Acid 238. Chemical properties tell you how something ______. Reacts 239. What are two ways can you identify a physical property? Uses your senses, can be measured 240. Malleable, Ductile, and Luster are all physical properties of what type of substance? Metal Identify each as a characteristic of a physical or chemical change. (241-246) 241. Something new produced. Chemical 242. Can be change back. Physical 243. Change in size or shape. Physical 244. Energy is produced. Chemical 245. Results in gas bubbles or a color change. Chemical 246. Phase change or mixture. Physical 247. What is the center of the atom? Nucleus 248. What charge do protons have? Positive 249. What charge do electrons have? Negative 250. What charge do neutrons have? Neutral or no charge 251. What two parts of the atom make up of the nucleus? Protons and neutrons 252. What part of the atom is involved in chemical bonding? Valence electrons 253. Where are valence electrons located? Last or outermost energy level 254. What identifies the number of protons in an atom? Atomic number 255. What identifies the number of protons plus neutrons? Atomic mass 256. What are atoms that gained or lost electrons and give them a positive or negative charge? Ions 257. What are atoms of the same element with different numbers of neutrons? Isotopes 258. What part of the atom never changes? Protons 259. What makes every element of the periodic table different? Protons or Atomic Number 260. What are the 18 columns on the periodic table called? Groups 261. What are the 7 rows on the periodic table called? Periods 262. A covalent bond is between what two things? Nonmetal and nonmetal 263. An ionic bond is between what two things? Metal and nonmetal 264. Where are the metals on the periodic table? Left of the staircase 265. Where are nonmetals on the periodic table? Right of the staircase 266. Where are the metalloids on the periodic table? On the staircase 267. Are there more metals, nonmetals, or metalloids on the periodic table? Metals 268. What is it called when an atom splits? Nuclear Fission 269. What is it called when two atoms combine? Nuclear Fusion 270. The left side of a chemical equation is called? Reactants 271. The right side of a chemical equation is called? Products 272. What identifies the # of atoms in a chemical bond? Subscripts 273. What identifies the # of molecules in a chemical reaction & can be used to balance an equation? Coefficients 274. What is stored energy? Potential energy 275. What is energy of motion? Kinetic energy Identify the examples of energy. (276-280) 276. Fire, Friction, Radiant Energy. Heat Energy 277. Sun, Homer Simpson’s Power Plant. Nuclear Energy 278. Food, Fuel, Batteries, Plants. Chemical Energy 279. Simple Machines, Wind, Water. Mechanical Energy 280. Circuits, Lightening, Solar Cells. Electrical Energy What energy transformation takes place in each. (281-282) 281. Photosynthesis. Light to Chemical 282. Flashlight. Chemical to Electrical to Light 283. The stomach is the major organ in what system? Digestive 284. The lungs are the major organs in what system? Respiratory 285. The brain is the major organ in what system? Nervous 286. The heart is the major organ in what system? Circulatory 287. What is the function of digestive system? Breakdown food 288. What is the function of respiratory system? Take in oxygen 289. What is the function of nervous system? Controls other body systems and muscles 290. What is the function of circulatory system? Move blood and oxygen throughout the body 291. What is the function of red blood cells? Carries oxygen 292. What is the function of white blood cells? Fights infections & diseases 293. What is the function of platelets? Clots the blood 294. What is the blood plasma? Blood fluid 295. What are three ways to increase how fast something dissolves? Heat it up, Cut it up, or stir it 296. Phototropism is when plants grow towards ________. Its light source or the Sun 297. What are two alternatives to using fossil fuels for power? Wind, Water, Nuclear, Solar 298. What is the problem with solar power? Sun doesn’t always shine 299. What is the problem with hydroelectric power? Everyone doesn’t live near a large water supply 300. What is the problem with wind power? Wind doesn’t always blow 301. What is the problem with nuclear power? Storing nuclear waste 302. What element do organic compounds always contain? Carbon 303. What are reactions that absorb or take in energy? Endothermic 304. What are reactions that release or let our energy? Exothermic 305. What determines the gravitational pull of an object? The object’s mass 306. What is an animal’s niche? Role or job in life 307. What is the wise use of natural resources called? Conservation 308. What is a science periodical? Magazine 309. What is it called when an animal sleeps through the winter to save energy? Hibernation 310. Where does an aquatic animal live? In the water 311. What is called when an animal blends in with their surroundings? Camouflage Aluminum has an atomic number of 13 and an atomic mass of 26.98 312. How many protons does Aluminum have? 13 313. How many neutrons does Aluminum have? 14 314. How many electrons does Aluminum have? 13 315. If an atom gains an electron the atom will have what charge? Negative 316. If an atom loses an electron the atom will have what charge? Positive 317. If the mass is 10 grams and the volume is 5 mL. What is the density? (Density = Mass/Volume) 2 g/mL 318. A cube has sides that measure 2 centimeters. What is the volume? (V = L x W x H). 8 cubic centimeters 319. What is the speed of a runner who travels 100 meters in 10 seconds? (Speed = Distance/Time). 10 m/s 320. How does most of the water in the water cycle get into the air? Evaporation 321. How does most of the water in the water cycle return to the Earth? Precipitation