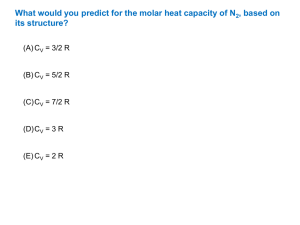Thermodynamics Worksheet: High School Physics
advertisement

12H Physics Thermodynamics worksheet 08 SECTION A 1. Which of the following statements regarding the internal energy of a gas is correct? A. The internal energy of an ideal gas is associated with the potential energy of its molecules. B. The internal energy of an ideal gas is entirely kinetic. C. The internal energy of a gas is given by U = Q + W. D. The internal energy of an ideal gas decreases as it undergoes a free expansion very quickly. E. The internal energy of an ideal gas at constant temperature varies inversely with volume. 2. A B. C. D. E. When ice at 0 oC melts to form water at the same temperature, which of the following is correct? U Increases No change No change Increases Increases W on system on system by system by system no change Q supplied to system supplied to system. supplied to system. supplied to system. supplied to system. 3. NOT THIS TIME 4. The p-V diagram represents a cycle of changes for a fixed mass of an ideal gas. p Isothermal change V For the complete cycle, which of the following is/are zero? A. 1,2 only B. 2,3 only C. 1 only 1. U 2. Q D. 3 only 3. W E. None of them 5. The molar gas constant is 8.3 J mol-1 K-1. The molar heat capacity at constant volume of an ideal gas is 12.5 J mol-1 K-1 . The heat transfer in J required to raise the temp. of 1 mol of the gas by 10K at constant pressure is A. 42 B. 83 C. 125 D. 0 E. 208 SECTION B 1. Calculate the change in internal energy when 40 g of ice changes to water under normal atmospheric pressure. Given density of ice is 920 kgm-3, density of water = 1000 kgm-3 latent heat of fusion of ice = 3.5 x IO5 Jkg - 1. 3. Figure 1 shows two processes abc and cda. For process abc, 7 J of work is done by the gas and 10 J of heat is absorbed by it. If the area enclosed by abcda corresponds to an energy difference of 4 J, what is the heat exchanged in carrying out the process cda? 4. 2 mole of an ideal gas occupying a volume of 0.5 m3 is heated at constant pressure of 6.64 kPa until the temperature of the gas changes from 200 K to 350 K. The gas is then cooled under constant volume until it reaches the initial temperature. During the cooling process, the internal energy changes by 3.75 kJ. What was the work done by the gas in the entire process? 5. Distinguish between the operation of a heat engine and a heat pump.