Science 10 - Mr. Laura/ Ms. Reynolds Fleetwood Park Secondary
advertisement

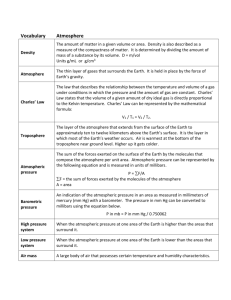
Science 10 - Mr. Laura/ Ms. Reynolds Name:_______________ Date:_______________ Fleetwood Park Secondary Block:____ Section 10.2 Notes Atmospheric Pressure (p. 443- 446) - What is weather? Weather is: The ______________________________in a specific place and at a specific time o Describes all aspects of the _______________, including: ______________________________ ______________________________ ______________________________ ______________________________ Closely connected to _______________in the atmosphere because convection moves ______________________________throughout the troposphere, o This causes the _______________we experience - Atmospheric Pressure In general, Pressure= _______________/area o Air pressure = ______________________________= pressure exerted by the _______________about any point on Earth’s surface. o In other words, it is a measure how many air molecules are colliding and exerting a _______________ on you Atmospheric Pressure is affected by these 3 factors: o _______________ o _______________ o _______________ Measuring Atmospheric Pressure: Is measured using a _______________which was invented by ______________________________ o He used an upside glass tube filled with _______________, and found that the _______________of mercury changed (went up or down) with changes in weather _______________atmospheric pressure made the mercury rise _______________atmospheric pressure made the mercury fall o Newer barometers are called: ______________________________ Contains a capsule of ______________________________ When the ____________________________changes, it causes the capsule to expand or contract The SI unit for atmospheric pressure = _______________ (Pa) o 1 pascal = 1 N/m2 (_______________/area) o Most measurements are given in kPa (kilopascals) = 1000 Pa o At sea level, the atmospheric pressure is _______________, or _________/cm2 which is similar to having ______ of air pushing down on each square centimetre of your body Pressure is affected by altitude and temperature Altitude Increased altitude = _______________pressure Decreased altitude = _______________pressure Temperature - Increased temperature = ______________pressure Decreased temperature = ______________pressure Kinetic molecular theory says that as particles gain ______________________________, they move around more quickly o because the molecules take up ________space, ______________air = less dense than ______________air Pressure is also affected by Humidity ______________- the measurement that describes the amount of ______________in the air. Water molecules take up more space than ___________________________, but their mass is less o This makes them ______________dense ____________________________- humidity expressed as the number of grams of water vapour in 1kg of air ____________________________- the point where air becomes saturated- it can’t hold any more ______________at a certain ______________ o When specific humidity= the ability of air to hold water ____________________________- compares the amount of water vapour in the air with the amount that the air ______________hold if it was ____________________________ Read Pages 447 and 448, take some notes on your own. Try to explain High and Low pressure in your own words. Air Masses: High Pressure systems: Low Pressure Systems: - KEY Section 10.2 Notes Atmospheric Pressure (p. 443- 446) - What is weather? Weather is: The condition of the atmosphere in a specific place and at a specific time o Describes all aspects of the atmosphere, including: Temperature Atmospheric pressure Amount of moisture in the air Wind speed and direction Closely connected to Heat transfer in the atmosphere because convection moves air and thermal energy throughout the troposphere, o This causes the weather we experience - Atmospheric Pressure In general, Pressure= amount of force/area o Air pressure = atmospheric pressure = pressure exerted by the mass of air about any point on Earth’s surface. o In other words, it is a measure how many air molecules are colliding and exerting a force on you Atmospheric Pressure is affected by these 3 factors: o Altitude o Temperature o Humidity Measuring Atmospheric Pressure: Is measured using a barometer, which was invented by Evangelista Torricelli o He used an upside glass tube filled with mercury, and found that the height of mercury changed (went up or down) with changes in weather Increase atmospheric pressure made the mercury rise Decrease atmospheric pressure made the mercury fall o Newer barometers are called: aneroid barometers Contains a capsule of flexible metal When the atmospheric pressure changes, it causes the capsule to expand or contract The SI unit for atmospheric pressure = pascal (Pa) o 1 pascal = 1 N/m2 (force/area) o Most measurements are given in kPa (kilopascals) = 1000 Pa o At sea level, the atmospheric pressure is 101.3 kPa, or 1 Kg/cm2 which is similar to having 1kg of air pushing down on each square centimetre of your body Pressure is affected by altitude and temperature Altitude Increased altitude = decreased pressure Decreased altitude = increased pressure Temperature - Increased temperature = decreased pressure Decreased temperature = increased pressure Kinetic molecular theory says that as particles gain kinetic energy, they move around more quickly o because the molecules take up more space, warm air = less dense than cold air Pressure is also affected by Humidity Humidity- the measurement that describes the amount of water in the air. Water molecules take up more space than oxygen and nitrogen, but their mass is less o This makes them less dense Specific Humidity- humidity expressed as the number of grams of water vapour in 1kg of air Dew point- the point where air becomes saturated- it can’t hold any more water at a certain temperature o When specific humidity= the ability of air to hold water Relative humidity- compares the amount of water vapour in the air with the amount that the air could hold if it was totally saturated