web assistance
advertisement

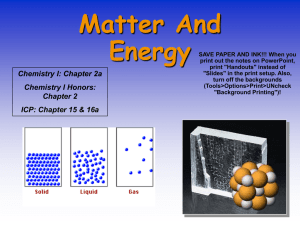
State Changes Name:_____________________ Melting point oC Substance ammonia iron lead mercury sulfur table salt water 1. Liquid -33 2750 1751 357 445 1473 100 Gas Convert the melting point of each substance to Kelvins: ammonia:196 2. -77 1535 327 -39 112 808 0 Solid Boiling point oC iron:______ lead: ______ mercury:______ sulfer: _______ Which of the substances listed above has a Boiling Point of: 373 K: water (100 + 273 = 373) 240 K:___________ 2024 K: ___________ 3. At what temperature does iron freeze?1535 4. At what temperature does iron boil?__________________ 5. Is iron a solid, liquid, or gas at 1000 oC?__________________ 6. Is lead a solid, liquid, or gas at 1000 oC? liquid (MP = 327, BP = 1751) 7. Is mercury a solid, liquid, or gas at -50 oC?__________________ 8. Is sulfur a solid, liquid, or gas at 500 oC?__________________ 9. Is table salt a solid liquid or gas at 1000 oC?__________________ 10. what is the hottest temperature that Sulfur can be a liquid?___________ 11. 12. At what temperature do the molecules of mercury in a gas state begin to remain in contact after colliding with each other?___________ What is the name of the state change described above?____________________________________ 13. Would sulfur be a solid, liquid, or gas in a pot of boiling water?______________________________ 14. What is the lowest temperature that Mercury can be a liquid? What is the melting point 15. At what temperature do the atoms in table salt begin to move past each other? ________________ 16. What is the name of the state change described above?_________________________________ 17. If you were in Antarctica and the temperature was -50 oC, would ammonia be in a solid, liquid, or gaseous state? ________________________________________ 18. In what state of matter is the hydrogen in the sun ? ___________________________________ Answer 19 – 23 with the following terms: melting, freezing, boiling, condensation, or sublimation 19. atoms begin to lock into place: _______________________________________________ 20. A direct change from solid to gas: __________________________________ 21. atoms that were in contact begin to move away from each other: _____________________________ 22. atoms begin to change positions: Melting 23. atoms begin to stick together when they touch: __________________________________ 24. What state of matter is characterized by molecules that are in contact with each other and cannot move past each other? __________________________________ 25. What state of matter is characterized by molecules not in contact with each other and can move past each other? gas 26. What state of matter is characterized by molecules that are in contact with each other but can move past each other?_______________________________ 27. Why do solids have a definite shape? _________________________________________________ _______________________________________________________________________________ 28. Why do liquids have a definite volume?________________________________________________ ________________________________________________________________________________ 29. Look at the graph above. What is the melting point of this substance? _____________ 30. Identify the substance shown on the graph above by comparing its melting point to those shown on the state change data table on the front of this sheet: _________________________ 31. How long did the melting process last once it began? it did not begin melting until the 20 second mark