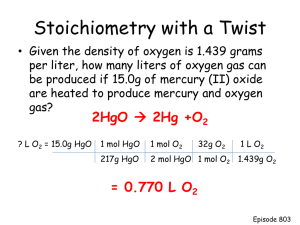Balancing Redox Reactions: Half-Reaction Method
advertisement

The Half-Reaction Method for Balancing Equations for Oxidation-Reduction Reactions Occurring in an Acidic Solution Write the equations for the oxidation and reduction half-reactions. For each half-reaction: 1. Balance all of the elements except hydrogen and oxygen. 2. Balance oxygen using H2O 3. Balance hydrogen using H+ 4. Balance the charge using electrons 5. Cancel like-species and combine remaining species Example: MnO4-(aq) + Os4+(aq) OsO4(s) + MnO2 (s) Write each half-reaction MnO4- Os4+ MnO2 OsO4 Step 1. Balance all the elements except H and O MnO4- Os4+ MnO2 OsO4 Since there are the same number of Mn and Os on each side of the reaction, we skip this step. Step 2. Balance oxygen using H2O---we have to understand that we are adding a new molecule “out of no-where,” but we have to remember that these reactions are done in water, and water can be part the reaction, it must be under these conditions. MnO4- MnO2 + 2H2O We add 2@H2O’s to the product side so we balance the O’s on each side of the reactions. 4H2O + Os4+ OsO4 We do the same for the other half reaction. Step 3. Balance hydrogen by using H+ 4H+ + MnO4- MnO2 + 2H2O We add 4H+’s to the reactant side to give 4@H’s on each side. 4H2O + Os4+ OsO4 + 8H+ Add 8H+’s to the Os half-reaction At this point ALL atoms are balanced. Step 4. Balance the charge using electrons 4H+ + MnO4- 4+ + MnO2 + 1- 0 2H2O + = 3+ 0 =0 We balance the charges of each side of the reaction using electrons. In the above case we do the following: 3e- + 4H+ + MnO4- 3- + 4+ + MnO2 + 1- 0 = 0 2H2O + 0 =0 Now the charge is balanced on both sides of the reaction. We know look at the other half-reaction. 4H2O + Os4+ 0 + 8H+ OsO4 + 4+ 0 + = 4+ 8+ = 8+ Balance the charges using electrons. 4H2O + Os4+ 0 + 4+ = 4+ OsO4 + 0 + 8H+ + 4e8+ + 4= 4+ We are not quit done with balancing the charges. Don’t forget that this is a Redox reaction and we need the same number of electrons for each oxidation and reduction reaction. 4[3e- + 4H+ + MnO43[4H2O + Os4+ MnO2 + OsO4 + 2H2O] 8H+ + 4e-] This goes to: 12e- + 16H+ + 4MnO412H2O + 3Os4+ 4MnO2 + 3OsO4 + 8H2O 24H+ + 12e- We now have the same number of electrons leaving and going into this Redox reaction. Step 5. Cancel like-species and add the two half reactions 12e- + 16H+ + 4MnO44H2O 12H2O + 3Os4+ 4H2O + 3Os4+ + 4MnO4- 4MnO2 + 3OsO4 + 4MnO2 + 8H2O 8H+ 24H+ + 12e3OsO4 + 8H+ Check to make sure all elements balance on each side of the reaction. The Half-Reaction Method for Balancing Equations for Oxidation-Reduction Reactions Occurring a Basic Solution Follow the same steps above for balancing in an acidic solution, there is an extra step at the very end. Example: Fe(OH)2 + CrO4-2 Fe2O3 Fe(OH)2 CrO4-2 + Cr(OH)4- Fe2O3 Cr(OH)4- Step 1. 2Fe(OH)2 Fe2O3 CrO4-2 Cr(OH)4- Step 2. 2Fe(OH)2 Fe2O3 + H2O CrO4-2 Cr(OH)4- Step 3. 2Fe(OH)2 Fe2O3 + H2O + 2H+ 4H+ + CrO4-2 Cr(OH)4- Step 4. 2Fe(OH)2 Fe2O3 + H2O + 2H+ 0 =0 0 + 0 = +2 + 2+ Add e2Fe(OH)2 Fe2O3 + H2O + 2H+ + 2e- =0 =0 4H+ + CrO4-2 Cr(OH)4- 4+ + 2= 2+ 3e- + 4H+ 3- + 4+ = 1- 3[2Fe(OH)2 + CrO4-2 1= 1 + 2- Cr(OH)4- 1= 1- Fe2O3 + H2O + 2H+ + 2e-] 2[3e- + 4H+ + CrO4-2 Cr(OH)4-] This goes to: 6Fe(OH)2 3Fe2O3 + 3H2O + 6H+ + 6e6e- + 8H+ + 2CrO4-2 2Cr(OH)4- Step 5. 6Fe(OH)2 3Fe2O3 + 3H2O + 6H+ + 6e2H+ 6e- + 8H+ + 2CrO4-2 2H+ + 6Fe(OH)2 + 2CrO4-2 2Cr(OH)43Fe2O3 + 3H2O + 2Cr(OH)4- Step 6 (new for balancing in a basic solution) Take the original reaction, to the side with H+, and add the same number of OH-‘s, then add the same number of OH- to the other side of the reaction: 2H+ + 6Fe(OH)2 + 2CrO4-2 + 2OH- 3Fe2O3 + 3H2O + 2Cr(OH)4+ 2OH- When we do this we now see, in this case 2H+ and 2OH- which we know forms H2O, so rewrite the equation: 2H2O + 6Fe(OH)2 + 2CrO4-2 3Fe2O3 + 3H2O + 2Cr(OH)4- + 2OHCancel waters that appear on both sides of the reaction: 2H2O + 6Fe(OH)2 + 2CrO4 -2 H 2O 3Fe2O3 + 3H2O + 2Cr(OH)4- + 2OH- Then write the finished reaction: 6Fe(OH)2 + 2CrO4-2 3Fe2O3 + H2O + 2Cr(OH)4- + 2OH- Check that you have the same number of atoms on each side.