Table S2: Morphologic sub-type and chromosome abnormalities in
advertisement

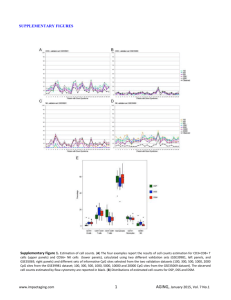
SUPPLEMENTARY FIGURE LEGENDS Figure S1. Clusters generated from the maturation-responsive CpG discriminated best between normal CD34+ cells and total bone marrow, despite 7 to 8-fold more CpG sites in the ‘no methylation change’ category, when unbiased hierarchical cluster analysis was used to analyze methylation data from an independent study downloaded from reference (8). Figure S2. The classification of promoter CpG based on direction of change in methylation during normal myeloid maturation in our dataset was applied to an independent dataset of promoter CpG methylation (independent data from (8)). Promoter CpG sites were classified into 3 categories based on our dataset and then applied to analyze this independent dataset: CpG that undergo a significant (p<0.001, t-test) increase in methylation from normal CD34+ cells (NCD34+) to normal mature cells (NBM) (95 CpG present in this dataset, ‘hypomet. in NCD34+’) (red box plots); CpG that undergo a significant (p<0.001, t-test) decrease in methylation from NCD34+ to NBM (157 CpG present in this dataset, ‘hypermet. in NCD34+’) (blue box plots); CpG that do not undergo a change in methylation between NCD34+ and NBM (1157 CpG present in this dataset, ‘no met. change during n. diffn.’) (grey box plots). A) Distribution of -values within each group of bone marrow samples. Methylation levels are represented by a -value between 0 (unmethylated) and 1 (fully methylated). NCD34 = CD34+ cells isolated from normal bone marrow (n=4), NBM = normal total bone marrow (n=4), MDS/MPD = bone marrow from MDS and MPD patients (n=13), AML = bone marrow from AML patients (n=116). Box-plot boundaries = inter-quartile range, horizontal line = median, ‘+’ = mean, whiskers = range of values, small boxes = out-lying values. Asterixes represent statistically significant differences between the median in the sample group compared to the NCD34+ group (median one-way). pvalues: ‘Hypo-met in NCD34’ NCD34 v NBM p=0.0137; ‘Hyper-met in NCD34’ NCD34 v NBM p=0.0131; ‘No change in met’ NCD34 v AML p=0.012; all other significant p-values <0.0001). B) -values of individual CpG sites in each sample. Each row represents a CpG site, with 95 CpG in the Hypo-Met in NCD34+ group, 157 CpG in the Hyper-Met in NCD34+ group and 1157 CpG in the No Change in Met group. Each column represents an individual bone marrow sample. 1 Figure S3. Myeloid maturation-responsive CpG sites (defined by analysis of our dataset) were overrepresented in the CpG sites that were most and least methylated in AML cells in the independent dataset (independent data from reference (8)). A) Of the 25 CpG sites that underwent a >20% increase in methylation from normal CD34+ cells to AML cells, 11 were from the 95 ‘hypo-met in NCD34’ CpG sites (black arrows) and 14 were from the 1157 ‘no change in methylation’ CpG sites. B) Of the 6 CpG sites that underwent >20% decrease in methylation from normal CD34+ cells to AML cells, all were from the 157 ‘hypermet in NCD34 category’ CpG sites (black arrows). Figure S4. Decitabine (DAC) 0.5µM did not induce apoptosis in Kasumi-1 cells, however, colonyforming ability (an assay for progenitors and stem cells) was substantially attenuated. A) No increase in apoptosis in Kasumi-1 cells treated with decitabine 0.5µM. Annexin and 7AAD staining measured by flow-cytometry on Day 5 after decitabine or AraC addition on day 1 and day 4. substantially decreased colony forming ability of Kasumi-1 cells. B) Decitabine 0.5µM Kasumi-1 cells were treated with decitabine 0.5µM on day 1 and day 4 and plated into semi-solid media which did not contain decitabine on day 5. Figure S5. The pattern of expression of key myeloid lineage-specifying and late-differentiation transcription factors (TF) in AML cells suggests impaired differentiation in lineage-committed cells. A) CD34+ AML cells (n=10) express higher levels of CEBPA (CEBP), and a higher CEBPA/HOXB4 ratio, than normal CD34+ cells (n=11). HOXB4 is a stem cell associated TF. Gene expression was analyzed by microarray. Expression levels are represented by heat-maps. p-values Wilcoxon two-sample test. Raw data extracted from GEO Datasets (1;2) B) CD34+CD38- cells from AML patient bone marrow (n=9) express higher levels of CEBPA, and a higher CEBPA/HOXB4 ratio, than CD34+CD38- cells from normal bone marrow (n=4). p-values median one-way test. Raw data extracted from GEO Datasets (6). C) CEBPA (lineage-specifying TF) and CEBPE (CEBP) (late- differentiation TF) expression in AML myeloblasts (n=318) compared with normal CD34+ cells, bone marrow and peripheral blood (n=38), and normal myeloblasts (n=3). p-values Wilcoxon two-sample test. Raw data extracted from GEO Datasets (3;4). 2 Figure S6. Putative RUNX1 and CEBPA (C/EBPa) binding sites were located in proximity to differentiation-responsive CpG in the CEBPE promoter. The genomic coordinates for the CEBPE promoter region analyzed for methylation are chr14:23,588,987-14:23,589,309. Putative TF binding sites in proximity to the three differentiation-responsive CpG sites identified were identified by TFSEARCHv1.3 ("TFSEARCH: Searching Transcription Factor Binding Sites", http://www.rwcp.or.jp/papia/)(9). Figure S7. Promyelocytic leukemia cells express higher levels of CEBPE (CEBP) than other AML subtypes. Gene expression analysis by microarray, data downloaded from GDS1064 (10). Figure S8. Decitabine treatment of the AML cell line THP1 decreased methylation levels of repetitive DNA elements (but not to the same extent as hypomethylation of CEBP promoter CpG). LINE-1 promoter methylation changes after a single decitabine treatment of 0.5 µM. DNA harvested 48h after treatment. 3