Supplementary material Nickel-doped zinc aluminate oxides: starch
advertisement

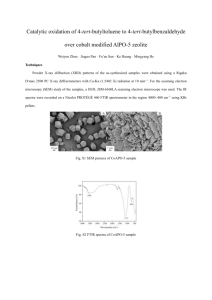
Supplementary material Nickel-doped zinc aluminate oxides: starch-assisted synthesis, structural, optical properties and their catalytic activity in oxidative coupling of methane Diana Visinescu, Florica Papa, Adelina C. Ianculescu, Ioan Balint and Oana Carp Diana Visinescu, Florica Papa, Ioan Balint and Oana Carp Romanian Academy, Institute of Physical Chemistry “Ilie Murgulescu”, Splaiul Independentei 202, 060021 Bucharest, Romania; e-mail: Adelina C. Ianculescu University Polytechnica of Bucharest, Gh. Polizu Street no.1-7, 011061 Bucharest, Romania FTIR spectra of the (Zn,Ni,Al)-starch gel-precursors The overlaid infrared spectra of starch for two (Zn,Ni,Al)-starch gel precursors, corresponding to an oxide composition of Ni0.6Zn0.4Al2O4 and Ni0.8Zn0.2Al2O4 are depicted in Fig. S1. The structured medium intensity band observed on 1000-1050 cm-1 region is attributed to the stretching vibrations of the C-C and adjacent C-O bonds originated from starch [1]. Significant differences between the two precursors samples are remarked on 1300-1480 cm-1 region, a zone specific to the δ(OCH), δ(COH), δ(CCH) deformations and also to nitrate characteristic bands. For x = 0.6 an intense and sharp absorption centred at ca. 1384 cm-1 is present, while for a richer nickel sample (x = 0.8) the band is clearly broadened. The distinct pattern of the two spectra suggest that the nickel content has determined the assembly of two different and complicated supramolecular systems in which the (Zn,Ni,Al)-starch incipient complexe(s) is/are covered and connected through hydrogen bonds to the carbohydrate in excess. The polysaccharide transformations is more evident for lower quantities of Ni(II) cations. In both cases is remarked the triplet characteristic for M-O and M-O-M’ bonds of the aluminate spinels [2] as low-intensity bands , which represents an additional prove that the nucleation centres of the mixed oxides are already formed in gel-precursors. Water is identified by a medium intensity band centred at 1639 (for x =0.6) and 1623 (for x = 0.8) cm-1, covering 3000-3500 cm-1 region. The sharp absorption located at 825 cm-1, observed for both precursors, corresponds to nitrate anions most probably embedded in the polysaccharide matrix. Fig. S1. Overlay of the FTIR spectra of starch and (Zn,Ni,Al)-starch gel precursors of NixZn1-xAl2O4 (x = 0.6, 0.8) References [1] K. Nakamoto, Infrared and Raman Spectra of Inorganic and Coordination Compounds, Part B, Application in Coordination, Organometallic, and bioinorganic chemistry 6th Ed., 2009. [2] (a) G.A. Siegel, R.A. Bartlet, D. Decker, M.M. Olmstead, P.P. Power, Charge delocalization in ruthenium-quinone complexes. Structural characterization of bis(bipyridine)(3,5-di-tertbutylsemiquinonato)ruthenium(II) perchlorate and trans-bis(4-tert-butylpyridine)bis(3,5-di-tertbutylquinone)ruthenium, Inorg. Chem.26 (1987) 1769-1773; (b) R.W. Adams, R.L. Martin, G. Winter,Possible ligand field effects in metal-oxygen vibrations of some first-row transition metal alkoxides, Aust. J. Chem.20 (1967) 773-774; (c) A. Kruger, G. Winter, Magnetism, electronic spectra, and structure of transition metal alkoxides. VIII. Nickel halo methoxides,Aust. J. Chem.23 (1970) 1-14; (c) C.G. Barraclough, D.C. Bradley, J. Lewis, I.M. Thomas,The infrared spectra of some metal alkoxides, trialkylsilyloxides, and related silanols, J. Chem. Soc. (1961) 2601-2605. FTIR spectra for NixZn1-xAl2O4 oxides FTIR spectra of the corresponding oxides reveal the presence of a broad three-structured band attributed to the spinelic phase that overlaps the 500-800 cm-1 region (fig. S3). For the mixed substituted oxides, NixZn1-xAl2O4 (x = 0, 0.1, 0.3, 0.5, 0.7, 0.9, 1) the highest energy peak of the triplet has been associated with vibration of the tetrahedral group (ZnO4), while the bands located at lower energies are related to tetrahedral (ZnO4)/(NiO4) and octahedral [AlO6] vibrations [3]. For the two oxide samples, Ni0.6Zn0.4Al2O4 and Ni0.8Zn0.2Al2O4, the differences remarked for an increased content of nickel, namely the changed pattern of absorptions attributed to the spinelic phase is most probably resulted from a distinct cations distributions, especially regarding the Ni(II) metal ions. The weak and sharp band, observed for both samples at 418 cm-1, is an additional prove that indicates that nickel oxide coexist with the spinelic oxide. Specific bands of starch are still visible in the FTIR spectra of the calcined samples independent on calcination temperature and time (800oC/1h, for x =0.6 and x = 0.8 and 1000oC/5h for x = 1, see fig. S4), meaning that the polysaccharide has been strongly associated in (Zn,Ni,Al)-starch gel precursors and the heating treatment could not remove completely the carbohydrate. References [3] L.K.C. de Souza, J.R. Zamian, G.N. da Rocha Filho, L.E.B. Soledade, I.M.G. dos Santos, A.G. Souza, T. Scheller, R.S. Angélica, C.E.F. da Costa, Blue pigments based on CoxZn1−xAl2O4 spinels synthesized by the polymeric precursor method, Dyes and Pigments 81 (2009) 187-192. Fig. S2. Overlay of the FTIR spectra of NixZn1-xAl2O4 (x = 0.2, 0.8) oxides Fig. S3. FTIR spectra of stoichimetric NiAl2O4 calcined at 1000oC for 5h Fig. S4. SEM images for Ni0.2Zn0.8Al2O4 oxide resulted after calcination at 800oC for 1 h Fig. S5. The NIR-UV-Vis spectra of the nickel-doped zinc aluminates oxides, Ni0.2Zn0.8Al2O4 for samples calcinated at 800oC and 1000oC for 1h Table S1. Color parameters for Ni0.2Zn0.8Al2O4 samples calcinated at 800 and 1000oC. Ni0.2Zn0.8Al2O4 L* a* b* T = 800oC 20 -4 -2 T = 1000oC 21 -6 -5 (a) (b) Fig. S6. The nitrogen adsorption-desorption isotherms for: (a) Ni0.2Zn0.8Al2O4 and (b) Ni0.2Zn0.8Al2O4 oxides