Retro Orbital Injection - UCSF Animal Care and Use Program
advertisement

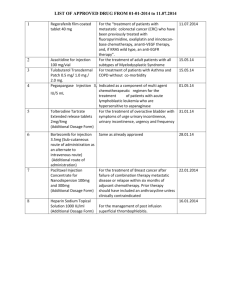
THE INSTITUTIONAL ANIMAL CARE AND USE COMMITTEE (IACUC) IACUC / LARC STANDARD PROCEDURES RETRO-ORBITAL INJECTION IN MICE Description of procedure: The retro-orbital injection is an acceptable alternative to the tail vein injection route, if technicians receive proper training. The route designated by the protocol should therefore be based on the researcher’s level of training and proficiency with the retro-orbital or tail vein injection. When administering a retro-orbital injection, anesthesia is required. When performed proficiently, this procedure will take less than five minutes. In which case, a heat source is not required. The injectable solution is limited to 200µL. Cell suspensions are filtered or agitated prior to injection to prevent cell clumping. A mouse receives no more than one injection per day. When more than one injection is required, alternate between eyes and allow 1-2 days between injection sites. Do not exceed two injections per eye in a mouse. Supplies: Anesthesia Gloves Needle (needle not to exceed ½” to prevent trauma) Gauze sponges 1. Anesthetize the mouse. 2. Position the mouse on its side. Restrain it with the thumb and middle finger of the nondominant hand, pulling back the loose skin over the shoulders and behind the ears. 3. Use the index finger of the non-dominant hand to draw back the skin above the eye and the thumb to draw back the skin below the eye. The eye will protrude slightly. 4. Insert the needle approximately at a 45° angle to the eye, lateral to the medial canthus, through the conjunctival membrane. There is a degree of resistance, which causes the eye to retreat back into the sinus, until the needle pierces through the conjunctiva. The needle will be positioned behind the globe of the eye in the retrobulbar sinus. A sharp cutting needle is preferred as it results in reduced tissue distortion and damage. 5. 6. 7. 8. 9. 10. Inject into the retrobulbar sinus. Remove needle gently to prevent injury to the eye. Close the eyelid and apply mild pressure to the injection site with a gauze sponge. Monitor the mouse during the recovery process. Examine the injection site for swelling or other visible trauma. If no adverse effects are observed, return the mouse to its home cage, once it regains its righting reflex. Updated June/2010 Please contact IACUC Training and Compliance at IACUCTrainer@ucsf.edu for training. Literature search words required: Literature search was performed for refinement of this Standard Procedure on May 26, 2010: Key Words Retro orbital injection Retro orbital tail vein comparison Retro orbital injection complications Search Site Pubmed PubMed Years Covered Full database up to 2010 Full database up to 2010 Agents: This procedure requires inhalant anesthesia (isoflurane). All agents administered to animals should be listed in the “Agents” section of RIO. Adverse Effects: Procedure, Agent or Phenotype Incorrect placement of the needle Tumor cells injection Monitoring Parameters General appearance and signs of trauma around injection site Grooming habits, skin color, lethargy, general appearance/activity level Adverse Effects Potential Adverse Effects Swelling around injection site, proptosis of the eye or eye trauma Tumor growth around injection site/behind the eye Monitoring Parameters Frequency During and after procedure, following day and prior to next injection. During and after procedure, following day and prior to next injection. Management Consult a LARC veterinarian or euthanize. Consult a LARC veterinarian or euthanize. Change injection site for the other animals in the study (e.g. tail vein injection). PI/Lab will Document Will document only if problem is identified. Will document only if problem is identified. Describe the conditions, complications, and criteria (e.g. uncontrolled infection, loss of more than 15% body weight, etc.) that would lead to removal of an animal from the study, and describe how this will be accomplished (e.g. stopping treatment, euthanasia). Euthanize if untreatable or permanent eye trauma occurs at the injection site or uncontrolled bleeding is observed in the mouse. In addition, if injecting tumor cells and the tumor rapidly grows around injection site/behind the eye, euthanize the animal. For all investigators housing animals with tumor formation, skin lesions, neurological deficits, or that are in Category E, list the expected characteristics/clinical presentations and endpoints of the animal model and the criteria for euthanasia. Note: The IACUC also requires such lists to be posted in the respective animal rooms and monitored by the IACUC compliance staff and LARC, to assure PI adherence to the endpoints listed. Updated June/2010 Updated June/2010