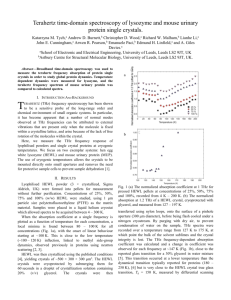
2. Methodology
advertisement

DEVELOPMENT OF TAGLESS BIOSENSORS F0R DETECTING THE PRESENCE OF PATHOGENS SHORT TITLE: TAGLESS BIOSENSORS JING–YIN CHEN*,1, JOSEPH R. KNAB1, SHUJI YE, YUNFEN HE2 AND ANDREA G. MARKELZ3 1 State University of New York at Buffalo, Department of Physics, 239 Fronczak Hall, Buffalo, NY, USA 14260 1 Second address… ABSTRACT. The vibrational modes corresponding to protein tertiary structural motion lay in the far infrared or terahertz frequency range. These collective large scale motions depend on global structure and thus will necessarily be perturbed by ligand binding events. We discuss the use of terahertz dielectric spectroscopy to measure these vibrational modes and the sensitivity of the technique to changes in protein conformation, oxidation state and environment. A challenge of applying this sensitivity as a spectroscopic assay for ligand binding is the sensitivity of the technique to both bulk water and water bound to the protein. This sensitivity can entirely obscure the signal from the protein or protein-ligand complex itself, thus necessitating sophisticated sample preparation making the technique impractical for industrial applications. We discuss methods to overcome this background and demonstrate how terahertz spectroscopy can be used to quickly assay protein binding for proteomics and pharmaceutical research. Keywords: biosensors, terahertz spectroscopy, far-infrared, lysozyme, tri-N-acetyl-Dglucosamine, biomolecular sensing, ligand binding 1. Introduction A key component to pharmaceutical development is the determination of protein-protein and protein-ligand binding. Rapid optical assays to determine this binding could have a significant impact, however not all binding events Terahertz Frequency Detection and Identification of Materials and Objects © 2007 Springer. Printed in the Netherlands 124 TAGLESS BIOSENSORS * To whom correspondence should be sent: J.Y. Chen, State University of New York at Buffalo, Department of Physics, 239 Fronczak Hall, Buffalo, NY, USA 14260; email jchen9@buffalo.edu will result in changes to UV/Vis absorbance or fluorescence since binding may occur at a site remote from the optically active region. In the case of mid infrared vibrational spectroscopy, while binding at any given site will locally change the MIR vibrational modes involving on the order of 5-10 atoms, this local change may have little effect on the dense overlapping modes in the amide I and II regions. Global collective vibrational modes which involve large scale structural motions on the other hand would be affected by any binding event within the molecule. These vibrational modes associated with protein tertiary structure lie in the far infrared or terahertz frequency range (0.03 – 6 THz, 1 – 200 cm-1). Methods of measuring these low frequency modes include inelastic neutron scattering1 (INS) and terahertz dielectric spectroscopy2-5. Recently there have been several reports of sensing biomolecular binding using these techniques. For example, INS measurements demonstrated this binding sensitivity in the case of binding of methotrexate with dihydrofolate reductase which indicating an increase in the vibrational density of states at these frequencies1. INS however is not a technique that is amenable to rapid parallel assays for drug discovery. 2. Methodology 2.1 Subtitle-1 Hen egg white lysozyme (HEWL) lyophilized power (Sigma Aldrich L6876) is dissolved in trizma buffer (pH 7.0, 0.05 M) and the final concentration of the lysozyme solution is 200 mg/ml. Tri-N-Acetyl-D-glucosamine (N, N’, N”Triacetylchitotriose; 3NAG) lyophilized power is purchased from Toronto Research Chemicals, Inc. (T735000). The HEWL+3NAG binding solution is made by dissolving 3NAG powder into the 200 mg/ml lysozyme solution and the molar ratio of lysozyme to 3NAG is 1 to 1. The solution sample cell for the transmission measurements is composed of two brass plates sandwiching two quartz windows (Spectrocell Inc.) with a ~ 250 m spacer10. The top plate of the cell has two identical apertures respectively for the sample and reference. About 14 µl of the protein solution is pipetted to fill the bottom aperture and the upper aperture is left empty as a reference. The entire measurement was performed for two different sets of the lysozyme and HEWL+3NAG binding solutions and results were found to be excellent agreement. TAGLESS BIOSENSORS 125 The ligand binding for the solutions was verified by the fluorescence measurements before terahertz transmission measurements. The fluorescence measurements are taken by using the SLM8100 spectrofluorimeter. The excitation wavelength for both types of solutions is 280 nm. We found the fluorescence peak of lysozyme solutions is about 343 nm and that of HEWL+3NAG binding solutions is 332 nm as expected for bound and unbound HEWL11,12. 2.2 Subtitle-2 The solution sample cell is mounted inside a continuous-flow cryostat (Cryo Industries). A silicon diode temperature sensor is located on the sample stick adjacent to the solution cell. Liquid nitrogen is used as the cryogen and the fluctuation at each temperature is lower than 0.01 K. Each temperature measurement is taken at least 10 minutes after the temperature is stable. At each temperature, measurements were repeated more than three times in order to make sure that the sample reached the thermal equilibrium. Terahertz time domain spectroscopy (THz-TDS) was used to determine the terahertz dielectric response. The terahertz electric field is generated by a hertzian dipole antenna and detected by the electro-optical detection13,14. The bandwidth for this study is good from 0.2 THz to 1.6 THz at room temperature and from 0.2 THz to 2.0 THz at low temperatures. A Ti-sapphire laser (65 fs, 82 MHz) is used to generate and detect terahertz electric field. The entire terahertz spectroscopy system is continuously purged with dry nitrogen gas in a plexiglass box prior to and during the measurements to avoid the atmospheric water absorption. Each measurement consists of measuring the relative transmission through the reference and sample by toggling between the two apertures of the sample cell. THzTDS measures the complex field transmission. We normalize our measurements to the reference transmission yielding a net field transmission with transmitted field amplitude, |t|, and the phase, : t Esample Ereference i te e ikd ( ns 1) e d 2 (1) where k is the frequency in wavenumbers (cm-1), d is the thickness of sample, ns is the refractive index of sample, and is the absorption coefficient. TAGLESS BIOSENSORS 126 3. Data & Result The temperature dependent terahertz transmission for HEWL and trizma buffer solutions is shown in Figure 1 for several frequencies. The temperature dependence of the transmission of HEWL solution is relatively flat for T < 200 K, and then decreases more rapidly above 200 K. Above 273 K, the transmission for both protein and buffer solutions dramatically drops due to the strong absorbance for bulk liquid water. … 1.2 1.0 |t| 0.8 0.6 0.4 0.2 Lysozyme solution 0.52 THz 0.84 THz 1.17 THz 1.31 THz Trizma Buffer 0.51 THz 0.83 THz 1.17 THz 1.32 THz 0.0 100 150 200 Temperature (K) 250 300 Figure 1 Transmission, |t| of the 200 mg/ml lysozyme and trizma buffer solution as a function of temperature at several frequencies. The transmission of the lysozyme solution shows a dramatic increase at 280 K at each frequency. Below 280 K, the transmission of HEWL solution increases with the decreasing temperature. In comparison with the lysozyme solution, |t| of the trizma buffer has a big jump at 273 K. Besides, |t| of buffer also increases with the decrease temperature. We now turn to the question of whether the terahertz dielectric response can distinguish between HEWL and 3NAG+HEWL. The native state and binding of HEWL and HEWL+3NAG solutions were verified by the fluorescence measurements. For each sample solution, the transmission at 295 K is much less than that at ~ 270 K. For both temperatures shown, the transmission decreases with increasing frequency for the entire frequency range and oscillates with a period ~ 0.8 THz. TAGLESS BIOSENSORS 127 4. Conclusions The terahertz dielectric response of the lysozyme and HEWL+3NAG binding solutions was measured as a function of frequency at room temperature and at 270 K. The terahertz transmission of the lysozyme solution at 295 K cannot be distinguished from that of the HEWL+3NAG binding solution due to the large contribution from the solvent. After the solvent was frozen, the protein dominates the terahertz spectrum. … ACKNOWLEDGEMENTS This work was supported by the American Chemical Society (PRF 39554AC6) and the National Science Foundation (NSF CAREER PHY-0349256, NSF IGERT DGE0114330 and NSF REU DMR-0243833). References 1. B.Erika, B.Torsten, O.Martin, L.Ruep, D.Roy, F.John and C.S.Jeremy, Physical Review Letters 93(2):028103 (2004). 2. M.Brucherseifer, M.Nagel, P.H.Bolivar, H.Kurz, A.Bosserhoff and R.Buttner,. Applied Physics Letters 77(24):4049-4051 (2000). 3. J.Y.Chen, J.R.Knab, J.Cerne and A.G.Markelz,. Physical Review E (Statistical, Nonlinear, and Soft Matter Physics) 72(4):040901 (2005). 4. J.R.Knab, J-Y.Chen and A.G.Markelz,. accepted by Biophysical Journal (2005). 5. A.Menikh, S.P.Mickan, H.Liu, R.MacColl and X.C.Zhang,. Biosensors and Bioelectronics 20(3):658(2004). 6. etc…. NOTE: this is the example template for ARW SMECS that is distributed only between contributing participants of the Workshop. It is based on the real article in which the part of the text was removed. It can be used for template purposes only.