Supplementary Figures Legends (doc 33K)
advertisement

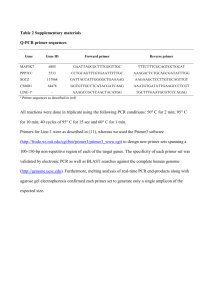
SUPPLEMENTARY INFORMATION Supplementary Figure 1. Tandem repeats in the TSTA elements are not deleted. HEK293 (293), HT-29, Hep G2, NIT-1, and -TC-6 cells were transduced with the indicated vectors. Genomic DNA was isolated and PCR was performed with primers designed to amplify the 345 bp of the two copies of the VP16 immediate early transactivator in Gal4VP2 (a) or the 170 bp fragment containing the five copies of the Gal4 DNA binding sites in G5E4T (b). Supplementary Figure 2. Detection of insulin mRNA in cell lines. RNA was isolated from the indicated cell lines or from primary islets as positive control. RT-PCR was performed using primers to detect murine insulin I or II mRNA in murine insulinoma cell lines (top panel), human insulin mRNA in human cell lines, or rat insulin mRNA in ARIP cells (bottom panel). No RT was used in some reactions containing primary islets as negative controls. Supplementary Figure 3. Diagram of names and locations of restriction enzymes used in Southern blot of Figure 2. The restriction enzymes used to digest genomic DNA for Southern blot analysis and their approximate locations are indicated on each lentiviral vector. The expected band of DNA (Kb) resulting from the enzyme digestion is shown to the right. Supplementary Materials and Methods Polymerase chain reaction. Genomic DNA was isolated using a standard phenol/chloroform extraction procedure or a DNeasy Tissue Kit (Qiagen; Valencia, CA) 1 from transduced cell lines carrying an integrated vector copy of 1 or less. To detect the 170 bp fragment encompassing the five Gal4 DNA binding sites, 1-2 L of genomic DNA was used per 50 L PCR reaction containing 2.0 mM MgCl2 using a sense primer (5’GCTCGCGAAAGCTTGCAT-3’) and an antisense primer (5’GCCAAGTGCAGAGCGAGTAT-3’) with the following conditions: 1 cycle at 94° C for 15 min, followed by 25 cycles at 94° C for 1 min, 59.2° C for 45 s, 72° C for 1 min, and a final extension at 72° C for 10 min. To detect the 345 bp fragment containing the two copies of the VP16 domain, 1-2 L of genomic DNA was used per 50 L PCR reaction containing 2.5 mM MgCl2 using a sense primer (5’-GTTGACTGTATCGCCGGAAT-3’) and an antisense primer (5’-ACAAAGATCCCAAGCTGTCG-3’) with the following conditions: 1 cycle at 94° C for 15 min, followed by 25 cycles at 94° C for 1 min, 60° C for 45 s, 72° C for 1 min, and a final extension at 72° C for 10 min. Reverse-transcription PCR. Total cellular RNA were isolated using an RNeasy Mini Kit (Qiagen, Valencia, CA), treated with DNase (Sigma-Aldrich; St. Louis, MO), and 100-200 ng RNA was reverse transcribed using a SuperScript First-Strand Synthesis System for RT-PCR Kit (Invitrogen; Carlsbad, CA) with Oligo(dT) according to manufacturer’s instruction. Five microliters of the resulting reaction containing the cDNA was subjected to PCR. To detect a 260 bp human insulin product, PCR was performed in 50 L reaction volume containing a sense primer (5’-TGTGAACCAACACCTGTG-3’), an antisense primer (5’-CGTCTAGTTGCAGTAGT-3’), and 5 L of 10X Combination Buffer (Gene Choice; Frederick, MD) with the following conditions: 1 cycle at 94° C for 15 min, followed by 25 cycles at 94° C for 1 min, 53° C for 1 min, 72° C for 2 min, and a final extension at 72° C for 10 min. To detect a 187 bp rat insulin product, PCR was performed in 50 L reaction volume containing a sense primer (5’- 2 CCTGCCCAGGCTTTTGTCAA-3’), an antisense primer (5’CTCCAGTGCCAAGTCTGAA-3’), and 5 L of 10X Combination Buffer (Gene Choice, Frederick, MD) with the following conditions: 1 cycle at 94° C for 10 min, followed by 25 cycles at 94° C for 1 min, 60° C for 45 s, 72° C for 1 min, and a final extension at 72° C for 10 min. To detect murine insulin sequences, PCR was performed in 50 L reaction volume containing 2.5 mM MgCl2, forward primer (5’-GCAAGCAGGTCATTGTTTCA-3’) and reverse primer (5’-GCTGGTAGAGGGAGCAGATG-3’) for a 329 bp murine insulin 1 product and forward primer (5’-TTTGTCAAGCAGCACCTTTG-3’) and reverse primer (5’TCTACAATGCCACGCTTCTG-3’) for a 241 bp murine insulin 2 product with the following conditions for both insulins: 1 cycle at 94° C for 15 min, followed by 35 cycles at 94° C for 30 s, 63° C for 30 s, 72° C for 1 min, and a final extension at 72° C for 10 min.. 3