Gabapentin Nycomed capsule, hard ENG SmPC
advertisement

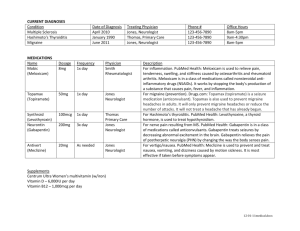
SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Gabapentin Nycomed 100 mg, 300 mg and 400 mg capsules, hard 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each capsule contains 100 mg, 300 mg respectively 400 mg gabapentin. Excipient with known effect: Lactose monohydrate For the full list of excipients, see section 6.1. 3 PHARMACEUTICAL FORM Capsule, hard Gabapentin Nycomed 100 mg capsules, hard, white, no imprint. Gabapentin Nycomed 300 mg capsules, hard, yellow, no imprint. Gabapentin Nycomed 400 mg capsules, hard, orange, no imprint. 4 CLINICAL PARTICULARS 4.1 Therapeutic indications As adjunctive treatment for partial epilepsy with or without secondary generalised seizures in patients refractory to standard antiepileptics. Painful diabetic neuropathy and post-herpetic neuralgia. 4.2 Posology and method of administration Dosage is settled by the treating doctor according to individual tolerance and efficacy. For individual adjustment of dose the strengths of 100 mg, 300 mg and 400 mg are available. Epilepsy The effective dose of gabapentin is 900-3600 mg per day. Adults and adolescents (from 12 years of age): The treatment starts with gabapentin 300 mg capsules, hard. Titration to an effective dose can progress rapidly and can be accomplished over a few days by administering 300 mg once a day on day 1, 300 mg twice a day on day 2 and 300 mg three times a day on day 3, using the following treatments: Day 1 (300 mg gabapentin/day) Day 2 (600 mg gabapentin/day) morning dose dose at noon – – 1 Gabapentin Nycomed 300 mg capsule, hard – evening dose 1 Gabapentin Nycomed 300 mg capsule, hard 1 Gabapentin Nycomed 300 mg capsule, hard Day 3 (900 mg gabapentin/day) 1 Gabapentin Nycomed 300 mg capsule, hard 1 Gabapentin Nycomed 300 mg capsule, hard 1 Gabapentin Nycomed 300 mg capsule, hard Alternatively an initial dose of 300 mg gabapentin three times per day (corresponding to 900 mg gabapentin per day) is recommended. Thereafter, the dose can be increased to 1200 mg per day given in three divided doses, and if necessary, further titration can occur using increments of 300 mg per day given in three divided doses to a maximum of 3600 mg per day. If a high dose is used and titration is rapid, the risk of dizziness during the titration period increases. It is not necessary to monitor the gabapentin plasma concentrations to optimise the therapy. The time between the evening dose and the following morning dose should not be more than 12 hours, in order to prevent new seizures. Paediatric population The safety and efficacy of gabapentin in children aged less than 12 years of age has not yet been established. Painful diabetic neuropathy and post-herpetic neuralgia Effect is usually obtained in the dose interval 1800-2400 mg per day, but in individual cases it may be necessary to increase the dose to a maximum of 3600 mg per day. Adults: The treatment starts with Gabapentin Nycomed 300 mg capsules, hard. Titration to an effective dose can be accomplished over a few days by administering 300 mg once a day on day 1, 300 mg twice a day on day 2 and 300 mg three times a day on day 3, using the following treatments: Day 1 (300 mg gabapentin/day) Day 2 (600 mg gabapentin/day) Day 3 (900 mg gabapentin/day) morning dose dose at noon – – 1 Gabapentin Nycomed 300 mg capsule, hard 1 Gabapentin Nycomed 300 mg capsule, hard – 1 Gabapentin Nycomed 300 mg capsule, hard evening dose 1 Gabapentin Nycomed 300 mg capsule, hard 1 Gabapentin Nycomed 300 mg capsule, hard 1 Gabapentin Nycomed 300 mg capsule, hard Thereafter, during one week the dose can be increased to 1800 mg per day given in three divided doses. In the following week, the dose can be increased if needed to 2400 mg per day given in three divided doses. If necessary, further titration can occur using increments of 300 mg per day given in three divided doses to a maximum of 3600 mg per day. Titration to maintenance dose should be done slowly to reduce the risk for adverse reactions. The time between the evening dose and the subsequent morning dose should not be more than 12 hours. The safety and efficacy of gabapentin in this indication for more than 5 months is not documented. Advice for all fields of application: Older people and patients with renal impairment: A reduced dose is recommended in patients with impaired renal function. Because the renal function has a tendency to decline in elderly patients, these patients may need an adjustment of the dose, too (see section 5.2 "Pharmacokinetic properties"). The following doses are recommended: Creatinine Clearance (ml/min) > 80 50-79 30-49 15-29 < 15 1 2 Total daily dose1 (mg) 900-3600 600-1800 300-900 1502-600 1502-300 The total daily dose is divided in 3 doses. 300 mg every other day. Patients on haemodialysis: An initial dose of 300-400 mg is given, and thereafter a maintenance dose of 200-300 mg gabapentin after every 4 hours of haemodialysis. On dialysis-free days, no treatment with Gabapentin Nycomed should take place. High risk patients: In patients with a poor general condition, low body weight, after organ transplantation etc., dose should be titrated only step by step, using Gabapentin Nycomed 100 mg capsules, hard. The duration of use is determined by clinical necessity. The treatment of epilepsy is usually long-term treatment. The attending doctor shall decide whether or not a single forgotten dose of gabapentin (i.e. the last intake time point was more than 12 hours ago) should be balanced by a later additional dose. Gabapentin Nycomed is administered orally with or without food, but with fluid. The capsules should not be chewed. 4.3 Contraindications Hypersensitivity to the active substance or to any of the excipients listed in section 6.1. Acute pancreatitis. 4.4 Special warnings and precautions for use Gabapentin Nycomed is usually not considered effective in the treatment of absence seizures and may aggravate these seizures in some patients. Therefore, gabapentin should be used with caution in patients with mixed seizures including absences. The risk of seizures improves with gabapentin in some former therapy-resistant patients. If a satisfactory effect is not achieved, gabapentin should be gradually withdrawn. Abrupt withdrawal can lead to a risk of an increase in the number of seizures or even precipitate status epilepticus. Dose reduction, discontinuation, addition of or replacement by another antiepileptic should be performed gradually over minimum one week. The dose of gabapentin has to be adjusted in case of renal impairment (see section 4.2 "Posology and method of administration" and section 5.2 "Pharmacokinetic properties"). There is insufficient experience with respect to the efficacy and safety of gabapentin. Therefore gabapentin is not recommended for the treatment of epilepsy in children < 12 years and for the treatment of painful diabetic neuropathy and post-herpetic neuralgia in children and adolescents <18 years (see section 4.2. “Posology and method of administration”). Gabapentin has not been systematically studied in patients older than 65 years. Experiences from clinical investigations do not indicate that adverse effects in patients older than 65 differ from adverse effects in younger patients. Following the treatment with gabapentin, hemorrhagic pancreatitis has been reported. Therefore, treatment with gabapentin has to be stopped, when symptoms of pancreatitis occur (continuing abdominal complaints, nausea, repeated vomiting). Besides a thorough clinical examination, relevant laboratory parameters should be examined for the early diagnosis of pancreatitis. No sufficient data are available concerning the use of Gabapentin Nycomed in patients with chronic pancreatitis. Caution is recommended in patients with a history of psychotic illness. On commencing gabapentin therapy, psychotic episodes have been reported in some patients with, and rarely without, a history of psychotic illness. Most of these events resolved when gabapentin was discontinued or the dosage was reduced. Drug Rash with Eosinophilia and Systemic Symptoms (DRESS) Severe, life-threatening, systemic hypersensitivity reactions such as Drug rash with eosinophilia and systemic symptoms (DRESS) have been reported in patients taking antiepileptic drugs including gabapentin (see section 4.8). It is important to note that early manifestations of hypersensitivity, such as fever or lymphadenopathy, may be present even though rash is not evident. If such signs or symptoms are present, the patient should be evaluated immediately. Gabapentin should be discontinued if an alternative etiology for the signs or symptoms cannot be established. Suicidal ideation and behaviour have been reported in patients treated with anti-epileptic agents in several indications. A meta-analysis of randomised placebo controlled trials of antiepileptic drugs has also shown a small increased risk of suicidal ideation and behaviour. The mechanism of this risk is not known and the available data do not exclude the possibility of an increased risk for gabapentin. Therefore patients should be monitored for signs of suicidal ideation and behaviours and appropriate treatment should be considered. Patients (and caregivers of patients) should be advised to seek medical advice should signs of suicidal ideation or behaviour emerge. This medicinal product should not be administered to patients with rare hereditary problems of galactose intolerance, the Lapp lactase deficiency or glucose-galactose malabsorption. 4.5 Interaction with other medicinal products and other forms of interaction Antacids reduce the bioavailability of gabapentin with up to 24%. It is recommended that gabapentin is taken at the earliest two hours after any administration of antacids. Gabapentin has not been observed to induce cytochrome P450 enzymes. No pharmacokinetic interactions have been observed between gabapentin and phenytoin, valproic acid, carbamazepine or phenobarbital. Gabapentin has no influence on contraceptives based on norethisteron- and / or ethinylestradiol. Combined with other antiepileptic medicinal products which are known to decrease the contraceptive efficacy, failure of contraception has to be taken into consideration. Renal excretion of gabapentin is not altered by the administration of probenecid. The slight decrease in renal excretion of gabapentin has been observed when co-administered with cimetidine which is not likely to be of clinical importance. Because false-positive readings were reported with dipstick test when gabapentin was added to other anticonvulsant medicinal products, the more specific sulphosalicylic acid precipitation procedure is recommended to determine urinary protein. The intake of alcohol or illicit drugs may aggravate some CNS related side effects of gabapentin, e.g. drowsiness, ataxia. Food intake has no influence on the pharmacokinetic of gabapentin. 4.6 Pregnancy and lactation Pregnancy Safe use in human pregnancy has not been established, as experience of use in pregnant women is limited. Animal studies yielded no indications of teratogenic effects of gabapentin, however, embryotoxic effects were observed (see section 5.3. Preclinical safety data). Generally, use of antiepileptic medicinal products during pregnancy increases the risk of malformations 2-3 times compared with children of non-epileptic women. The most frequent defects registered are within the central nervous system, heart and skeleton deformations, urinary tract and orofacial defects (cleft lip/palate). Mental retardation or delayed mental and motoric development can occur in children. Impairment or discontinuation of seizure prophylaxis may lead to a considerable risk for both the mother and the unborn child and this risk may be more serious than the risk of malformations. Generally, the risk of injury to the embryo/foetus is least when monotherapy is used, when the lowest possible dose is chosen and folic acid is given before and during the pregnancy. Potential risks when used in combination with other antiepileptics should be considered. Gabapentin should only be given during pregnancy if the benefit outweighs a possible risk. Breast-feeding Gabapentin is excreted in maternal milk, and the average milk/plasma ratio is 0.73. The calculated dose the child ingests is 1.2 mg/kg/day. The effect on breast-fed infants is unknown and it cannot be excluded that they could be affected. Gabapentin should therefore not be used during lactation: a decision should be made to discontinue breast-feeding or to discontinue the medicinal product, taking into account the importance of the medicinal product to the mother. 4.7 Effects on ability to drive and use machines Gabapentin may have minor or moderate influence on the ability to drive and use machines. It acts on the central nervous system and may produce somnolence, drowsiness, dizziness, or other related symptoms and could be potentially dangerous in patients driving or operating machines. This has to be considered especially in case of initiation or increase of the medicinal product. 4.8 Undesirable effects Undesirable effects are in general described as light to moderate. The most frequent possible undesirable effects are: disturbances in the central nervous system, reduced alertness, ability to concentrate, somnolence, fatigue, ataxia, dizziness, headache, nausea, vomiting, weight increase, nervousness, insomnia, nystagmus, paraesthesia, loss of appetite. In combination with other antiepileptics undesirable effects have been reported in ca. 50% of patients. The below list shows the undesirable effect, arranged after system organ class and frequency, Very common (≥ 1/10), Common (≥1/100 to <1/10), Uncommon (≥1/1,000 to <1/100), Rare (≥1/10,000 to <1/1,000), Very rare (<1/10,000), not known (cannot be estimated from the available data) Blood and the lymphatic system disorders: Common :Leucopenia Uncommon :Blood glucose fluctuations Rare :Thrombocytopenia, changes in glucose in patients with diabetes Psychiatric disorders: Common: Nervousness, depressive mood, disorientation, confusion and emotional lability, anxiety; Additionally, in clinical studies in children under 12 years of age, aggressive behaviour and hyperkinesia were reported. Uncommon: Depression, psychoses/hallucinations, hostility Nervous system disorders: Common: Ataxia, nystagmus, tremor, amnesia, sensitations such as paraesthesia, twitches, amplified, alleviated or missing reflexes, reduced alertness, reduced ability to concentrate, disturbances of thinking, speech-disturbances, memory impairment, dysarthria, abnormal gait Uncommon: Confusion, hypesthesia Rare: Movement disorders (e.g. choreoathetosis, dyskinesia, dystonia) Eye disorders: Common: Visual disturbances such as amblyopia, diplopia, reduced sight Ear and labyrinth disorders: Rare: Tinnitus Cardiac disorders: Common: Vasodilatation, hypertension Rare: Palpitations Respiratory, thoracic and mediastinal disorders: Common: Rhinitis, pharyngitis, cough Uncommon: Dyspnoea Rare: Upper respiratory tract infection Gastrointestinal disorders: Common: Vomiting, nausea, diarrhoea, dry mouth, dyspepsia, constipation, abdominal pain, dental anomalia, gingivitis, feeling unwell Uncommon: Flatulence Skin and subcutaneous tissue disorders: Common: Pruritus, purpura, rash Rare: Urticaria, alopezia, angioedema Not known: Drug rash with eosinophilia and systemic symptoms (see section 4.4) Musculoskeletal, connective tissue and bone disorders: Common: Arthralgia, myalgia, low back pain, bone fractures Renal and urinary disorders: Common:Incontinence, urinary tract infection Rare:Acute renal failure Reproductive system and breast disorders Common: Impotence General disorders and administration site conditions: Common: Drowsiness, tiredness, sleepiness, somnolence, fatigue, dizziness, headache, insomnia, weight gain, anorexia, peripheral oedema or oedema of the whole body, asthenia, increased appetite Uncommon:Accidental injury, vertigo Rare: Withdrawal reaction (pain, sweating thoracic pain) : Under treatment with gabapentin allergic reactions (Stevens-Johnson-Syndrome and Erythema multiforme) were reported very rarely (< 1/10,000). Cases of haemorrhagic pancreatitis (see 4.4 Special warnings and precautions for use), hypotension, bradycardia, syncope, atrial fibrillation, electrocardiographic abnormalities and maculopapular rashes have been reported in patients receiving gabapentin. Clinical manifestation on laboratory parameters In combination with other antiepileptic medicinal products elevated activity of hepatic enzymes were reported. A possible dose dependent increase in the frequency of seizures has been found in a few patients. Dose-related repeated seizures of an uncharacteristic type have also been reported. Reporting of suspected adverse reactions Reporting suspected adverse reactions after authorisation of the medicinal product is important. It allows continued monitoring of the benefit/risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions via the national reporting system listed in Appendix V. 4.9 Overdose Symptoms of overdose are dizziness, ataxia, diplopia, slurred speech, dysarthria, somnolence, lethargy, apathy and mild diarrhoea. Acute life-threatening toxicity has not been observed with overdoses of gabapentin with up to 49 g per day. Gabapentin can be eliminated by haemodialysis. Previous experience indicates, that it is not usually required. However, in patients with renal impairment, haemodialysis may be indicated. 5 PHARMACOLOGICAL PROPERTIES 5.1 Pharmacodynamic properties Pharmacotherapeutic group: Antiepileptics / Analgesics ATC code: N03AX12 / NO2BG Mechanism of action The mechanism of action of gabapentin is not clear. Gabapentin is structurally related to the neurotransmitter GABA (gamma amino butyric acid), but its mechanism of action is different from that of several other active substances that interact with GABA-synapses including valproate, barbiturates, benzodiazepines, GABA-transaminase-inhibitors, GABA-uptake inhibitors, GABA-agonists, and GABA-prodrugs. In clinically relevant concentrations gabapentin is not bound to other cerebral receptors for common active substances or neurotransmitters including receptors for GABAA, GABAB, benzodiazepine, glutamate, glycine, or N-methyl-d-aspartate. Gabapentin does not interact with sodium channels in-vitro and differs hereby from phenytoin and carbamazepine. Gabapentin causes a slight reduction of the release of monoamine neurotransmitters invitro. Gabapentin administered to rats increases the GABA-metabolism in several cerebral regions in a similar way as sodium valproate, however, in other cerebral regions. 5.2 Pharmacokinetic properties Absorption: Gabapentin plasma concentrations are dose proportional between dosages of 300 mg and 400 mg, administered every 8 hours. The absorption of gabapentin is saturable and non-linearly related to dose. The bioavailability of gabapentin declines at higher doses, it is about 60% with a dose of 300 mg and about 42% after 800 mg. Food has no effect on the pharmacokinetics of gabapentin. The pharmacokinetic of gabapentin is not influenced by repeated doses, and steady state plasma concentrations may be predicted on the basis of a single dose. Distribution: Maximum plasma concentrations (e.g. 4-5.5 µg/ml following repeated oral daily doses of 900-1200 mg) of gabapentin are achieved within 2 to 3 hours with therapeutic doses. The percentage of gabapentin bound to plasma proteins is less than 3%. It has a volume of distribution of 57.7 litre. In epileptic patients the concentration of gabapentin in the central nervous system is app. 20% of the corresponding lowest plasma concentration at steady-state. Biotransformation: Gabapentin is not metabolised in humans and does not induce drug metabolising cytochrome P450 enzymes. Elimination: Gabapentin elimination from plasma was described by linear pharmacokinetics. Elimination half-life of gabapentin is dose independent and ranged from 5 to 7 hours. In elderly patients the age related changes in renal function cause a decrease in the plasmaclearance and an increase in the elimination half-life of gabapentin. In reduced renal function, renal excretion rate is directly proportional to creatinine clearance. Gabapentin is cleared from plasma through haemodialysis (see 4.2 Posology and method of administration). 5.3 Preclinical safety data Acute toxicity: After administration of doses up to 8000 mg/kg to mice and rats the oral lethal dose could not be estimated. Toxicity after repeated dosing: Investigations in non-rodents and rodents have shown the kidney and liver as toxicological target organs. Fertility and reproduction: No adverse effects on fertility or reproduction were observed in rats at doses up to 2000 mg/kg. In mice, the no-effect level was 500 mg/kg/day. There was an increased incidence of hydroureter, hydronephrosis and delayed ossification in rat offspring after maternal doses of more than 500 mg/kg/day. In a teratology study in rabbits, an increased incidence of postimplantation fetal loss occurred in dams exposed to more than 60 mg/kg/day. Mutagenesis: Gabapentin has no genotoxic potential. It was not mutagenic in the Ames bacterial plate incorporation assay or at the HGPRT locus in mammalian cells in the presence or absence of metabolic activation. Gabapentin did not induce structural chromosome aberrations in mammalian cells in vitro or in vivo, and did not induce micronucleus formation in the bone marrow of hamsters Carcinogenesis: Gabapentin was fed to mice (up to 2000 mg/kg body weight/day) and to rats (up to 2000 mg/kg body weight/day) in 2-year studies. In male rats a statistically significant increase in the incidence of acinic cell tumours of the pancreas was observed in the highest dose group. In rats receiving 2000 mg/kg body weight daily, the maximum plasma concentration of the active ingredient and the area under the concentration-time curve were 10 times higher than the plasma concentrations in humans taking a daily dose of 3600 mg. Acinic cell tumours of the pancreas in male rats have a slight carcinogenic risk, had no influence on survival times, were not metastasising or invasive into the surrounding tissue and are similar to those of the control groups. The relevance of these acinic cell tumours of the pancreas in male rats for a carcinogenic risk in humans is therefore uncertain. 6 PHARMACEUTICAL PARTICULARS 6.1 List of excipients Capsules 100 mg: lactose monohydrate, maize starch, talc, gelatin, titanium dioxide (E171). Capsules 300 mg: lactose monohydrate, maize starch, talc, gelatin, titanium dioxide (E171), yellow iron oxide (E172). Capsules 400 mg: lactose monohydrate, maize starch, talc, gelatin, titanium dioxide (E171), yellow and red iron oxide (E172). 6.2 Incompatibilities Not applicable. 6.3 Shelf life 3 years. 6.4 Special precautions for storage Store below 30 ºC. 6.5 Nature and content of container Blister pack of PVC/aluminium. Gabapentin Nycomed capsules 100 mg; 20 and 100 caps. Gabapentin Nycomed capsules 300 mg; 50, 100 caps. Gabapentin Nycomed capsules 400 mg; 50, 100 caps. HDPE-container Gabapentin Nycomed capsules 100 mg; 100 caps. Gabapentin Nycomed capsules 300 mg; 100 caps. Gabapentin Nycomed capsules 400 mg; 100 caps. Not all pack sizes may be marketed. 6.6 Special precautions for disposal No special requirements. Any unused medicinal product or waste material should be disposed of in accordance with local requirements. 7 MARKETING AUTHORISATION HOLDER To be completed nationally 8 MARKETING AUTHORISATION NUMBER(S) Capsules 100 mg: Capsules 300 mg: Capsules 400 mg: 9 DATE OF FIRST AUTHORISATION/RENEWAL OF THE AUTHORISATION 2004-02-20/2009-02-20 10 DATE OF REVISION OF THE TEXT 2015-07-03