Glucose, anhydrous
advertisement

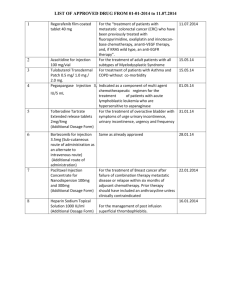
18. March 2015 SUMMARY OF PRODUCT CHARACTERISTICS for Nanocoll, kit for radiopharmaceutical preparation 1. NAME OF THE MEDICINAL PRODUCT NANOCOLL 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Human albumin colloidal particles 500 micrograms/vial At least 95 % of human albumin colloidal particles have a diameter 80 nm. NANOCOLL is prepared from human serum albumin derived from human blood donations tested according to the EEC Regulations and found non reactive for: Hepatitis B surface antigen (HBsAg) Antibodies to human immunodeficiency virus (anti-HIV 1/2) Antibodies to hepatitis C virus (anti-HCV) NANOCOLL is reconstituted with Sodium Pertechnetate (99mTc) Injection (not included in this kit) to prepare technetium-99m albumin nanocolloid injection. Technetium-99m decays with the emission of gamma radiation with an energy of 140 keV and a half life of 6 hours to technetium-99 which can be regarded as quasi stable. Excipients with known effect: The reconstituted injection contains 0.24 mg/ml sodium. For a full list of excipients, see section 6.1 3. PHARMACEUTICAL FORM Kit for radiopharmaceutical preparation Powder for solution for injection 4. CLINICAL PARTICULARS 4.1 Therapeutic indications This medicinal product is for diagnostic use only. After radiolabelling with Sodium Pertechnetate (99mTc) solution, the product is indicated in adults, neonates and children aged 1 to 18 years for: 106732761 Page 1 of 11 Intravenous administration: - Bone marrow scanning. (The product is not suitable to study the haematopoietic activity of the bone marrow). - Inflammation scanning in areas other than the abdomen. Subcutaneous administration: Lymphoscintigraphy to demonstrate integrity of the lymphatic system, also to identify the sentinel node in malignant diseases such as melanoma, breast, prostate, penile, head and neck, female pelvic (cervix and vulvar) cancer and to differentiate between venous and lymphatic obstruction. 4.2. Posology and method of administration Posology Adults Recommended activities in adults are as follows: Bone marrow scanning: 185-500 MBq. Inflammation imaging: 370-500 MBq. Lymphoscintigraphy: The recommended activity by single or multiple subcutaneous (interstitial) injection ranges from 18.5-110 MBq per injection site. For identifying sentinel node, see below. The injection site is given subcutaneously, after checking by aspiration, that a blood vessel has not been inadvertently punctured. - Melanoma: Intradermal injection 20-120 MBq, administered in four doses in the vicinity of the primary lesion or biopsy scar. - Breast Cancer: 20 - 370 MBq in several doses each of 5-20 MBq to be injected intradermally, subdermally, or sub/periareolary (superficial tumours) and intratumourally or peritumourally (deep tumours). - Prostate Cancer: 65 - 400 MBq a median of 250 MBq in one to four doses it recommended to be injected intra prostate under ultrasound guidance. - Penile Cancer: 40 - 131 MBq in several doses each of 20 MBq to be injected intratumourally or peritumourally (deep tumours). - Head and Neck Cancer: 30 - 100 MBq in one-more doses to be injected intratumourally or peritumourally (deep tumours). - Female pelvic Cancer (cervix and vulvar): 60-120 MBq to be injected peritumourally (deep tumours). In cervical cancer the injection is given subepithelially around the tumour. Renal and hepatic impairment Careful consideration of the activity to be administered is required since an increased radiation exposure is possible in these patients. 106732761 Page 2 of 11 Paediatric population The use in children and adolescents has to be considered carefully, based upon clinical need and assessing the risk/benefit ratio in this patient group. The activity for children may be calculated from the recommended range of adult activity and adjusted according to body weight or surface area. However the Paediatric Task Group of the European Association of Nuclear Medicine (EANM) recommends calculating the administered activity from the body weight according to the following table. Fraction of adult dose: 3 kg = 0.10 4 kg = 0.14 6 kg = 0.19 8 kg = 0.23 10 kg = 0.27 12 kg = 0.32 14 kg = 0.36 16 kg = 0.40 18 kg = 0.44 20 kg = 0.46 22 kg = 0.50 24 kg = 0.53 26 kg = 0.56 28 kg = 0.58 30 kg = 0.62 32 kg = 0.65 34 kg = 0.68 36 kg = 0.71 38 kg = 0.73 40 kg = 0.76 42 kg = 0.78 44 kg = 0.80 46 kg = 0.82 48 kg = 0.85 50 kg = 0.88 52-54 kg = 0.90 56-58 kg = 0.92 60-62 kg = 0.96 64-66 kg = 0.98 68 kg = 0.99 In very young children (up to 1 year) a minimum dose of 20 MBq (bone marrow scanning) is necessary in order to obtain images of sufficient quality. In children, it is possible to dilute the product, see section 12. This product is not intended for regular or continuous administration. Method of administration: This medicinal product should be reconstituted before administration to the patient. For instructions on reconstitution of the medicinal product before administration, see section 12. This agent is not intended for regular or continuous administration. Intravenous administration Bone marrow scanning: Images may be acquired 45-60 minutes after administration. Inflammation scanning: Dynamic imaging is performed immediately. Static imaging comprises an early phase, 15 minutes post-injection and a washout phase, 30-60 minutes post-injection. Subcutaneous administration Lymphoscintigraphy: Is given by single or multible subcutaneous (interstitial) injection (18.5-110 MBq per injection site) and depends on the anatomical areas to be investigated and upon the time interval between injection and imaging. The injected volume should not exceed 0.2 - 0.3 ml. A maximum volume of 0.5 ml per injection is critical. The injection is given subcutaneously, after checking by aspiration, that a blood vessel has not been inadvertently punctured. When imaging the lower limbs, dynamic pictures are taken immediately following injection and static imaging 30-60 minutes later. In parasternal lymph scanning, repeated injections and additional images may be required. 106732761 Page 3 of 11 4.3 Contraindications Hypersensitivity to the active substance(s), to any of the excipients listed in section 6.1 or to any of the components of the labelled radiopharmaceutical. In particular, the use of 99m Tc-human albumin colloidal particles is contraindicated in persons with a history of hypersensitivity to products containing human albumin. During pregnancy, lymphoscintigraphy, including sentinel node detection, involving the pelvis is strictly contraindicated due to the accumulation in lymph nodes. 4.4 Special warnings and special precautions for use Pregnancy, see section 4.6 Paediatric population Paediatric population, see section 4.2. Individual benefit / risk justification For each patient, the radiation exposure must be justifiable by the likely benefit. The activity administered should in every case be as low as reasonably achievable to obtain the required diagnostic information. Lymphoscintigraphy is not advised in patients with total lymphatic obstruction because of the potential radiation hazard at injection sites. Renal impairment and hepatic impairment Careful consideration of the benefit risk ratio in these patients is required since an increased radiation exposure is possible. Patient preparation The patient should be well hydrated before the start of the examination and urged to void as often as possible during the first hours after the study in order to reduce radiation. Potential for hypersensitivity or anaphylactic reactions The possibility of hypersensitivity including serious, life-threatening, fatal anaphylactic/ anaphylactoid reactions should always be considered. If hypersensitivity or anaphylactic reactions occur, the administration of the medicinal product must be discontinued immediately and intravenous treatment initiated, if necessary. To enable immediate action in emergencies, the necessary medicinal products and equipment such as endotracheal tube and ventilator must be immediately available. Specific warnings It is strongly recommended that the product name and batch number are stated every time Nanocoll is given to a patient, in order to maintain a connection between the patient and the product’s batch number. Standard measures for preventing transmission of infections from pharmaceuticals made of human blood or plasma, include selection of donators, test of individual donators and plasma pools for finding specific infective agents, and effective manufacturing steps for inactivation/elimination of virus as a part of manufacturing process as well. In spite of that, the risk of transmission of infectious agents cannot be eliminated completely, as long as pharmaceuticals made of human blood or plasma are used. This also applies to new virus of unknown nature and other pathogens as well. 106732761 Page 4 of 11 There are no reports of virus transmission in connection with albumin, made in accordance with specifications in Ph. Eur. and in accordance with routine processes. Precautions with respect to environmental hazard are in Section 6.6. The human albumin contained in Nanocoll corresponds to the requirements "Note for Guidance on Plasma Derived Products", CPMP/BWP/269/95, rev. 2. For the production of Nanocoll only human albumin of plasma of donators was used whose blood on the occasion of every donation was checked individually with suitable methods for HBsAg and antibody against HIV-1, HIV-2 and HCV and was found not reactive. Blood or plasma of donators from a land in which several cases vCJK have appeared is not used for the production of the human albumin. Before reconstitution this medicinal product contains less than 1mmol sodium (23mg) per vial, i.e. essentially ‘sodium-free’. The reconstituted injection contains 0.24 mg/ml sodium. This needs to be taken into consideration for patients on a controlled sodium diet. 4.5 Interaction with other medicinal products and other forms of interaction Iodinated contrast media used in lymphoangiography may interfere with lymphatic scanning using technetium-99m albumin nanocolloid. 4.6 Pregnancy and lactation Woman of childbearing potential When an administration of radiopharmaceuticals to a woman of childbearing potential is intended, it is important to determine whether or not she is pregnant. Any woman who has missed a period should be assumed to be pregnant until proven otherwise. If in doubt about her potential pregnancy (if the woman has missed a period, if the period is very irregular, etc.), alternative techniques not using ionising radiation (if there are any) should be offered to the patient. Pregnancy Radionuclide procedures carried out on pregnant women also involve radiation doses to the foetus. Only essential investigations should therefore be carried out during pregnancy, when the likely benefit far exceeds the risk incurred by mother and foetus. Dose to the uterus arising for intravenous administration of 500 MBq of technetium-99m albumin nanocolloid is 0.9 mGy. Dose to the uterus above 0.5 mGy will be regarded as a potential risk to the foetus. During pregnancy the subcutaneous administration of technetium-99m albumin nanocolloid for lymphoscintigraphy, including sentinel node detection, is strictly contraindicated, due to the possible accumulation in pelvic lymph nodes, see section 4.3. Breastfeeding Before administering radiopharmaceuticals to a mother who is breast-feeding consideration should be given to the possibility of delaying the administration of radionuclide until the mother has ceased breast-feeding and as to what is the most appropriate choice of radiopharmaceutical, bearing in mind the secretion of activity in breast milk. If the administration is considered necessary, breastfeed should be interrupted for 13 hours post injection and the expressed feeds discarded. 106732761 Page 5 of 11 4.7 Effects on ability to drive and use machines No studies on the effects on ability to drive and use machines have been performed. 4.8 Undesirable effects The frequencies of undesirable effects are defined as follows: Very common (≥1/10), common (≥1/100 to <1/10), uncommon (≥1/1,000 to <1/100), rare (≥1/10,000 to <1/1,000), very rare (<1/10,000) and not known (cannot be estimated from the available data) Congenital, familial and genetic disorders Frequency not known (cannot be estimated from the available data) Neoplasms benign, malignant and unspecified (including cysts and polyps) Frequency not known (cannot be estimated from the available data) Immune system disorders Frequency not known (cannot be estimated from the available data) Hereditary defects. Cancer induction. Hypersensitivity reactions (including very rare life-threatening anaphylaxis). Occasionally hypersensitivity reactions (including very rare life-threatening anaphylaxis) may occur. Exposure to ionising radiation is linked with cancer induction and a potential for development of hereditary defects. As the effective dose is 2,3 mSv when the maximal recommended activity of 500 MBq is administered these adverse events are expected to occur with a low probability. For safety with respect to transmissible agents see section 4.4. Reporting of suspected adverse reactions Reporting suspected adverse reactions after authorisation of the medicinal product is important. It allows continued monitoring of the benefit/risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions via: Sundhedsstyrelsen Axel Heides Gade 1 DK-2300 København S Websted: www.meldenbivirkning.dk E-mail: sst@sst.dk 4.9 106732761 Overdose The risk of overdose lies in an unintentional high exposure to ionising radiation. In the event of an overdose of radioactivity being administered when using technetium-99m albumin nanocolloid, no practical measure can be recommended to satisfactorily diminish tissue exposure as the label is poorly eliminated in urine and faeces. Page 6 of 11 5. PHARMACOLOGICAL PROPERTIES 5.1 Pharmacodynamic properties Pharmacotherapeutic group: Technetium (99mTc), particles and colloids ATC-code: V09DB01 At the chemical concentrations and activities used for diagnostic procedures technetium99m albumin nanocolloid does not appear to exert any pharmacodynamic effects. 5.2 Pharmacokinetic properties Distribution Reticuloendothelial cells in liver, spleen as well as in bone marrow are responsible for blood clearance after intravenous injection. A small fraction of technetium-99m radioactivity passes through kidneys and is eliminated in urine. The maximum concentration in the liver and spleen is reached after about 30 minutes, but in the bone marrow after only 6 minutes. The proteolytic breakdown of the colloid begins immediately after its uptake by the reticuloendothelial system (RES), the products of degradation being excreted through the kidneys into the bladder. Organ uptake After subcutaneous injection into connective tissue, 30-40 % of the administered technetium-99m albumin colloidal particles (less than 100 nm) are filtered into lymphatic capillaries whose main function is the drainage of proteins from the interstitial fluid back into the blood pool. The technetium-99m albumin colloidal particles are then transported along the lymphatic vessels to regional lymph nodes and main lymphatic vessels, and are finally trapped into the reticular cells of functionary lymph nodes. Elimination A fraction of the injected dose is phagocytised by histiocytes at the injection site. Another fraction appears in the blood and accumulates mainly in the RES of the liver, spleen and bone marrow; faint traces are eliminated via the kidneys. 5.3 Preclinical safety data No animal death and no gross pathological changes at necropsy were noted after intravenous injection of 800 and 950 mg in mice and rats respectively. No local reactions were observed in either mice or rats subcutaneously injected with 1g/kg. These doses correspond to the contents of several tens of vials per kg body weight, compared to the human albumin colloid dose of 7 micrograms/kg generally used in nuclear medicine for diagnosis. Mutagenicity studies and long-term carcinogenicity studies have not been carried out. 6. PHARMACEUTICAL PARTICULARS 6.1 List of excipients Stannous chloride, dihydrate Glucose, anhydrous Poloxamer 238 Sodium phosphate, dibasic, anhydrous Sodium phytate, anhydrous 106732761 Page 7 of 11 6.2 Incompatibilities This medicinal product must not be mixed with other medicinal products except those mentioned in section 12. 6.3 Shelf life Kit before reconstitution: 12 months from the date of manufacture. Reconstituted product: should be used within 6 hours after labelling. Store below 25ºC. Do not refrigerate or freeze. 6.4 Special precautions for storage Store in a refrigerator (2-8°C). Storage should be in accordance with national regulations for radioactive material. For storage conditions of the reconstituted product, see section 6.3. 6.5 Nature and contents of the container 10 ml, Type I Ph.Eur., glass vials sealed by bromobutyl rubber stoppers and metal flip off caps, placed in a polystyrene tray and a package insert, inserted in a cardboard box. Pack size: Each kit contains 5 vials. 6.6 Special precautions for disposal and other handling Radiopharmaceuticals should be received, used and administered only by authorised persons in designated clinical settings. Their receipt, storage, use, transfer and disposal are subject to the regulations and/or appropriate licences of the competent official organisation. Radiopharmaceuticals should be prepared by the user in a manner which satisfies both radiation safety and pharmaceutical quality requirements. Appropriate aseptic precautions should be taken. Contents of the vial are intended only for use in the preparation of 99m Tc-human albumin nanocolloid and are not to be administered directly to the patient without first undergoing the preparative procedure. For instructions on reconstitution of the medicinal product before administration, see section 12. The content of the kit before reconstitution is not radioactive. However, after Sodium Pertechnetate (99mTc) Injection Ph. Eur. is added, adequate shielding of the final preparation must be maintained. The administration of radiopharmaceuticals creates risks for other persons from external radiation or contamination from spills of urine, vomiting, etc. Radiation protection precautions in accordance with national regulations must therefore be taken. Normal safety precautions for handling radioactive materials should be observed. After use, all materials associated with the preparation and administration of radiopharmaceuticals, including any unused product and its container, should be decontaminated or treated as radioactive waste and disposed of in accordance with the conditions specified by the lo- 106732761 Page 8 of 11 cal competent authority. Contaminated material must be disposed of as radioactive waste via an authorised route. 7. MARKETING AUTHORISATION HOLDER GE Healthcare S.r.l. Via Galeno, 36 20126 - Milan - Italy Representative GE Healthcare A/S Park Allé 295 2605 Brøndby 8. MARKETING AUTHORISATION NUMBER DK R 1107 9. DATE OF FIRST AUTHORISATION 20. June 1995 10. DATE OF REVISION OF THE TEXT 18. March 2015 11. DOSIMETRY Technetium (99mTc) is produced by means of a (99Mo/99mTc) generator and decays with the emission of gamma radiation with a mean energy of 140 keV and a half-life of 6.02 hours to technetium (99Tc) which, in view of its long half-life of 2.13 x 105 years can be regarded as quasi stable. The radiation dose estimation for a number of organs is based on MIRD reference man and MIRD S values, and has been calculated from biological data of organ uptake and blood clearance. The radiation doses absorbed by a patient weighing 70 kg, after intravenous injection of 99m Tc-human albumin colloidal particles, are reported hereafter. Organ Liver Urinary bladder (wall) Spleen Bone marrow (red) Ovaries Testes Whole body Absorbed dose µGy/MBq 78 25 18 14 3.2 1.1 5.1 The effective dose resulting from an administered activity of 500 MBq for an adult weighing 70 kg is about 2.5 mSv. For an administered activity of 500 MBq the typical radiation dose to the critical organ (liver) is 39 mGy and the typical radiation dose to the target organ (red bone marrow) is 7.0 mGy. 106732761 Page 9 of 11 The radiation doses absorbed by a patient weighing 70 kg, after subcutaneous injection of 99m Tc-human albumin colloidal particles, are reported hereafter. Organ Injection site Lymph nodes Liver Urinary bladder (wall) Spleen Bone marrow (red) Ovaries Testes Whole body Absorbed dose µGy/MBq 12000 590 16 9.7 4.1 5.7 5.9 3.5 4.6 The effective dose resulting from an administered activity of 110 MBq for an adult weighing 70 kg is about 0.44 mSV. For an administered activity of 110 MBq the typical radiation dose to the target organ (lymph nodes) is 65 mGy and to the critical organ (injection site) 1320 mGy. 12. INSTRUCTIONS FOR PREPARATION OF RADIOPHARMACEUTICALS Method of preparation Place a vial containing the albumin colloidal particles in a convenient lead shield. Aseptically introduce into the vial 1-5 ml Sodium Pertechnetate (99mTc) Injection Ph. Eur. with a radioactivity ranging from 185 to 5550 MBq (5 to 150 mCi). In children, it is possible to dilute the product up to 1:50 with sodium chloride for injection. Do not use a breather needle. Relieve the excess of pressure in the vial by simply withdrawing an equal volume of gas in the syringe. Invert carefully a few times to dissolve the contents of the vial. Then allow standing for 30 min at room temperature (15°C -25°C). Shake before withdrawing a dose. In no case should the preparation be left in contact with air. The disposal of waste should be in accordance with national and international guidelines. Quality control A – RCP by ascending paper chromatography Support: paper Whatman No. 1 Solvent: methanol:water 85:15 v/v Time: 1 hour 99m Tc (nanocolloid): ≥ 95 % Rf [99mTc (nanocolloid)]: 0,0 % Free 99m-Tc pertechnetate migrate with Rf 0.7 + 10% 106732761 11 Page 10 of B - RCP by ascending chromatography on TLC-SA: Support: TLC-SA (2 x 12 cm strips; dispose a small drop on preparation at 2.5 cm of the bottom) Solvent: methanol:water 85:15 v/v Time: 25-30 minutes (approximately at 7 cm from the origin; remove strip from the tank and allow it to dry) 99m Tc (nanocolloid): ≥ 95 % Rf: [99mTc (nanocolloid)]: 0.0-0.1 Free 99m-Tc pertechnetate and other Technetium-hydrophilic complexes migrate with Rf 0.8 – 1.0 Do not use material if the radiochemical purity is less than 95%. 106732761 11 Page 11 of