MECHANICAL ALLOYING OF Cu
advertisement

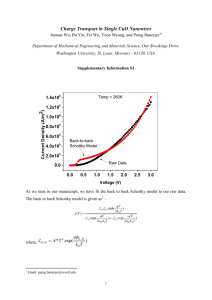
MECHANICAL ALLOYING OF Cu-Al2O3 NANOPARTICLES K. Borodianskiy1,2, A. Basov1,2, A. Gedanken2, M. Zinigrad1 1 Natural Science Faculty, Ariel University Center of Samaria, Ariel, Israel 2 Department of Chemistry, Bar-Ilan University, Ramat-Gan, Israel Abstract. Nano-sized materials formation by mechanical alloying (MA) is the interest of this article. Results of Mechanical Alloying treatment of Al and CuO are presented. Reduction of CuO by aluminum is very exothermic reaction where Cu9Al4 phase forms first and it is the possibility of explosion during the reaction. To avoid Cu9Al4 phase formation and explosion possibility, pre-milling of CuO was prepared. This pre-milled oxide was milled with Al for different times and after 15 h of milling, Cu and Al2O3 nanoparticles with an average size of 125 nm are formed. INTRODUCTION Mechanical alloying has been one of the novel non-equilibrium methods of a solid state powder processing technique involving repeated deformation, welding and fracturing of powder particles. This technique was developed in the middle of 1960s by John S. Benjamin at the Paul D. Mercia research laboratory of the International Nickel Company to produce nickel-based oxide dispersion strengthening (ODS) superalloys. This technique is an alternative for the traditional method where materials production is carried out by high temperature synthesis. The method of MA has been widely used to synthesize a various solid state materials, such as amorphous alloys [1], quasicrystalline and nanocrystalline alloys [2], intermetallic compounds [3], borides [4], or carbides [5]. Nanostructured materials may be formed by MA. During the process, the grain size of materials decreases with milling time and reaches a saturation level when a balance is established between the fracturing and cold welding events. The minimal grain size is different depending on the material and milling conditions. This way Ye et al [6] synthesized TiC with a grain size of 9 nm, Tavoosi et al [7] synthesized Al2O3 which reached grain size about 40 nm. The main advantage of these synthesis techniques are that they can be produced at relatively low temperatures and also in an economical way through fewer steps than in the conventional way. Nano-sized structures can be synthesized by a displacement reaction: MeO R Me RO , (1) where a metal oxide (MeO) is reduced by more reactive metal (reductant R) to the pure metal Me. The chemical reactions induced by high energy ball milling can be either combustion or progressive reactions. The Al2O3 synthesis from pure Al and metal oxide, ZnO [7], CuO [8] or Cu2O [9] are highly exothermic and the self propagating reactions can be avoided by reducing the activities of reactants. This can be achieved by intermetallic compounds formation. Aluminum oxide can be synthesized by aluminothermic reaction where Al uses as a reducing element at a high temperature. However, MA process is a simple and easy process to achieve homogeneous nano-sized oxide. In this study, we demonstrate the Al2O3 formation by mechanosynthesis displacement reaction avoiding intermediate phase Cu9Al4 intermetallic compound 2-141 which forms during the reaction. Additionally, we show the way to suppress a self propagating combustion reaction. EXPERIMENTAL Copper oxide powder (Acros organics, 98%) was pre-milled in planetary ball mill under air atmosphere for 5 h at 450 rpm. The ball-to-powder (BPR) ratio was 1:10. Stearic acid (Sigma-Aldrich, 95%) was used as a process control agent (PCA) with 3wt.%, added to prevent powder spreading. A milling vial of chromium hardened steel was of 125 ml. Pre-milled CuO and Al (Metallisation, 99%+) with a molar ratio of 1:1 with 3wt.% of stearic acid which used as a PCA were milled in planetary ball mill at 500 rpm under Ar atmosphere. Vials and balls are made of chromium hardened steel, and the volume of the vial was 125 ml, which contains 12 balls (10 mm in diameter). The BPR was about 10:1. The milling process was interrupted at different times, i.e., 0, 5, 10, 15 h, and small amount of powders were removed from the vial for characterization. XRD measurements (X-ray powder diffractometer, D8 Bruker, 40kV, 40mA) with Cu Kα radiation (λ=0.154 nm) was used to analyze the microstructure of as-milled samples. The XRD patterns were recorded in the 2Θ range of 20-80º (step size 0.05º and time per step 3s). The microstructure of powder particles was investigated by scanning electron microscopy (SEM, FEI Inspect S). RESULTS AND DISCUSSION In this research we obtained Cu and Al2O3 nanoparticles using mechanosynthesis displacement reaction between a pure Al and CuO powders. This red-ox reaction is very high exothermic reactions: 3CuO 2 Al 3Cu Al2O3 , (2) H 1210kJ / molAl2O3 , where Cu9Al4 phase forms first. To avoid this phase formation we suggested CuO premilling where CuO becomes more reactive because of energy transformed to the powder in this process. Figure 1 shows XRD patterns of the CuO and Al powder mixture after different stages of milling. The intensity of copper oxide and pure aluminum peaks reduced within the first 5 h of the process. Pure copper phase peaks forms near 43º, 50º, and 74º. They become broader with increasing of mechanical milling time (Fig.1). As it is known from X-ray diffraction theory, peak broadening becomes as the particle size decreases to nano-size. Al2O3 (corundum) phase falls very close to that of Cu peak near 35º. Figure 2 illustrates Scanning Electron Microscopy (SEM) image of the obtained powder. On this image we can find nanoparticles which average size was calculated using a computer program “Image J”. According to this calculation, the nanoparticles size was about 125 nm. Alumina phase presence in the obtained powder was found using Energy Dispersive X-ray analysis (EDX) and shown in Fig. 3. As shown in Fig. 3, Al2O3 phase is present in the obtained powder. This is in agreement with SEM image where we can find light zones which are explained by the theory of SEM as having high charge, e.g. in ceramic materials. 2-142 Fig. 1. XRD patterns after (a) 0 h, (b) 5 h, (c) 10 h, and (d) 15 h of milling. Fig. 2. SEM image of obtained nanopowder. 2-143 Fig. 3. EDX image of obtained nanopowder. CONCLUSIONS The red-ox synthesis by mechanical alloying on the example of reaction between aluminum and copper oxide was conducted. As it is known from previous works, this reaction is very exothermic and it is chance for explosion during the process. To avoid this problem, CuO pre-milling process was prepared. The obtained pre-milled CuO powder milled with Al and nanoparticles, with an average size of 125 nm of aluminum oxide and copper, were obtained. REFERENCES (1) (2) (3) (4) (5) (6) (7) (8) (9) Jinhui Wang: Mechanical alloying of amorphous Al–SiO2 powders. J. Alloys Compd. 2008 456 139-42. C.C. Koch: The synthesis and structure of nanocrystalline materials produced by mechanical attrition. Nanostruct. Mater. 1993 2 109-29. T.Yu. Kiseleva, A.A. Novakova, T.F. Grigorieva, A.P. Barinova: Iron and indium interactions during mechanical attrition. J. Alloys Compd. 2004 383 (12) 94-7. Y. Hwang, J.K. Lee: Preparation of TiB2 powders by mechanical alloying. Mater. Lett. 2002 54 1-7. B.Zhang, Z.Q. Li: Synthesis of vanadium carbide by mechanical alloying. J. Alloys Compd. 2005 392 183-6. L.L. Ye, M.X. Quan: Synthesis of nanocrystalline TiC powders by mechanical alloying. Nanostructur. Mater. 1995 5 25-31. M. Tavoosi, F. Karimzadeh, M.H. Enayati: Fabrication of Al–Zn/α-Al2O3 nanocomposite by mechanical alloying. Mater. Lett. 2008 62 282-5. K. Wieczorek-Ciurowa, D. Oleszak, K. Gamrat: Mechanosynthesis and process characterization of some nanostructured intermetallics–ceramics composites. J. Alloys Compd. 2007 434-435 501-4. Seung J, Hwang, Jin-hyung Lee: Mechanochemical synthesis of Cu–Al2O3 nanocomposites. Mater. Sci. Eng. A 2005 405 140-6. 2-144