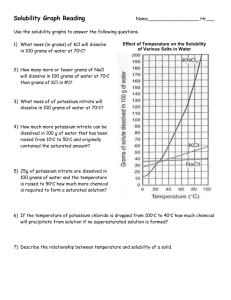
SOLUTIONS PROBLEMS - Parkway C-2
advertisement

SOLUTIONS PROBLEMS! 1. The solubility of carbon dioxide in water is .161 g CO2 in 100 mL of water at 200C and 1.00 atm. A soft drink is carbonated with CO2 at 5.5 atm pressure. What is the solubility of carbon dioxide in water at this pressure? 2. What is the solubility of nitrogen from the air in pure water at 250C, if the solubility constant of nitrogen is 6.8 x 10-4 mol/L atm? The pressure of nitrogen in the air is .78 atm. 3. Pure carbon dioxide readily leaves soda, as it has a solubility in water of .034 mol/L at 1 atm of pure CO2 pressure and 250C. If the partial pressure of carbon dioxide in the air is only .030 atm, then what is the solubility of CO2 in water from the air under atmospheric conditions? 4. What is the solubility of nitrogen in human blood at 370C due to the air, if the solubility constant for nitrogen is 7 x 10-4 mol/L atm? Assume the pressure of nitrogen in the air is .781 atm. 5. Nitrogen is soluble in blood and can cause intoxication at sufficient concentrations. For this reason, Navy divers using compressed air are not allowed to go below 125 feet. If the solubility of nitrogen gas is 1.75 x 10-5 g/mL of blood at .78 atm of nitrogen, what is the solubility of nitrogen in blood at 3.75 atm of nitrogen, the pressure at this depth? 6. Acetylene is an explosive gas that can be transported safely in acetone. The solubility of acetylene in acetone is 27g per 1 liter at 1.0 atm of pressure. If the partial pressure of the acetylene is increased to 12 atm, what is its solubility in acetone? 7. How would you prepare 145 grams of an aqueous solution that is 2.50% by mass of potassium iodide? 8. How would you prepare a solution that is .5M lithium nitrate? 9. If I wanted 35 grams of sodium acetate, how much of a 3M solution of sodium acetate would I need? 10. I have 34.9 grams of calcium hydroxide, and 750 mL of water. What is the molarity of the solution? 11. I want to prepare 500 mL of a 6 M solution of phosphoric acid. How many grams of acid do I need? 12. 68.5 grams of silver are melted into 1500 grams of gold. What is the molality of the alloy? 13. How many mL of a 12 M solution of hydrochloric acid are needed to make 3 L of a 2 M solution? How would you perform the dilution? 14. If 150 mL of a 18 M solution of sulfuric acid are used to make 1.5 L of a new solution, what is the molarity of the new, diluted solution? 15. How many grams of glucose are there in a hospital bag that contains 500 mL of a .1M solution of glucose? (Glucose is C6H12O6) 16. Vanillin, C8H8O3, occurs naturally in vanilla extract and is used as a flavoring agent. A 37.2 mg sample of vanillin was dissolved in 168.5 mg of diphenyl ether (C6H5)2O. What is the molality of the vanillin in solution? 17. Lauryl alcohol, C12H25OH, is prepared from coconut oil; it is used to make sodium lauryl sulfate, a synthetic detergent. What is the molality of lauryl alcohol in a solution containing 15.6 grams of lauryl alcohol dissolved in 148 grams of ethanol? 18. Caffeine, C8H10N4O2, is a stimulant found in tea and coffee. A sample of caffeine was dissolved in 45.0 grams of chloroform, CHCl3, to give a .0946 m solution. How many grams of caffeine were in the sample? 19. An antiseptic solution contains hydrogen peroxide, H2O2, in water. The solution is .655 m. What is the mole fraction of hydrogen peroxide? 20. How many liters of water are needed to produce a 2M solution of hydrochloric acid using 2.2 kilograms of hydrochloric acid? 21. A solution of vinegar is .763 M in acetic acid. The density of the solution is 1.004 g/mL. What is the molality of the acetic acid? 22. A beverage contains tartaric acid, H2C4H4O6, a substance obtained from grapes during wine making. If the beverage is .271 M in tartaric acid, what is the molality? The density of the solution is 1.016 g/mL. 23. The legal amount of lead in drinking water is .015 ppm. Express this concentration in ppt. 24. The EPA limit for arsenic in drinking water is .002 ppm. What is this value in ppb? 25. Concentrated nitric acid is 70% HNO3 by mass. How many grams of HNO3 are in 500 grams of the acid? 26. If wine is 10% ethanol, how many milliliters of ethanol are present in a bottle containing 750. mL of the wine? 27. A liter of water at 250C dissolves .0404 g O2 when the partial pressure of the oxygen is 1.00 atm. What is the solubility of oxygen from air, in which the partial pressure of O2 in 159 mm Hg? 28. How would you prepare 425 g of an aqueous solution containing 2.40% by mass of sodium acetate? 29. 5.67 grams of glucose (C6H12O6) is dissolved in 25.2 grams of water at 250C. The vapor pressure of water at 250C is 23.8 mm Hg. What is the new vapor pressure of this solution? 30. Napthalene, C10H8, is used to make mothballs. Suppose a solution is made by dissolving .515 g of naphthalene in 60.8 grams of chloroform, CHCl3. The original vapor pressure of chloroform at 200C is 156 mm Hg. What is the new vapor pressure of the solution? Napthalene can be assumed to be non-volatile compared with chloroform. 31. An aqueous solution is 2.22 m glucose. What are the boiling point and freezing point of this solution? 32. Ethylene glycol is the main component of antifreeze. How many grams of ethylene glycol, CH2OHCH2OH, must be added to 37.8 g of water to give a freezing point of -5.500C? 33. Safrole is contained in oil of sassafras and was once used to flavor root beer. A 2.39 g sample of safrole was dissolved in 103.0 g of diphenyl ether. The solution had a melting point of 25.700C. Calculate the molar mass of safrole. The freezing point of pure diphenyl ether is 26.840C, and the Kf is 8.000C/m. Saffrole is a covalent compound. 34. Butylated hydroxytoluene (BHT) is used as an antioxidant in processed foods. It prevents fats and oils from becoming rancid. A solution 2.500 g of BHT in 100.0 g of benzene had a freezing point of 4.8800C. What is the molar mass of the BHT? Benzene has a normal freezing point of 5.50C, and a Kf of 5.12 oC/m. BHT is covalent. 35. Urea, (NH2)2CO, is dissolved in 100.0 grams of water. The solution freezes at -2.50C. How many grams of urea were dissolved to make this solution? Assume urea has an i=3. 36. A solution was prepared by dissolving 9.15 grams of sulfur, S, in 100.0 grams of acetic acid. Calculate the freezing point and boiling point change of this solution. 37. Salt is used to de-ice the streets in the winter. If we need to lower the melting point of ice on the streets by 60C on a given evening, how many grams of salt (NaCl) need to be added to say, 200 kg of ice on a given street? 38. What is the freezing point of a .91 m aqueous sodium phosphate solution? Use the formula of the compound to obtain i. 39. The vapor pressure of pure benzene is 100.0 mm Hg at 26.10C. Calculate the vapor pressure of a solution containing 24.6 grams of camphor (C10H16O) dissolved in 98.5 g of benzene (C6H6). 40. How many grams of Urea (H2NCONH2) must be added to 450 g of water to give a solution with a vapor pressure of 2.50 mm Hg less than that of pure water at 300C? (The vapor pressure of pure water at 300C is 31.8 mm Hg). 41. In beer making, often times the carbon dioxide is produced through yeast fermentation, naturally in the bottle. Glucose is consumed in the bottle by yeast to produce ethanol and carbon dioxide. What is the solubility of the carbon dioxide in beer at 250C, if the pressure of the CO2 produced inside the bottle is 15 atm? The solubility constant for CO2 is .0325 mol/L atm at 250C. 42. A solution of .85 grams of the organic compound mesitol in 100.0 grams of benzene is observed to have a freezing point of 5.160C. What is the molality of the mesitol solution? What is the molar mass for mesitol? Benzene has a normal freezing point of 5.50C. 43. Pheromones are compounds secreted by the females of many insect species to attract males. One of these compounds contained 80.78% C, 13.56% H, and 5.66% O. A solution of 10.0 g of this pheromone in 8.50 g of benzene freezes at -1.611 degrees C. What are the molecular formula and molar mass of the compound? Benzene has a normal freezing point of 5.50C. 44. Dichloromethane (CH2Cl2) is a popular non-polar solvent used in the manufacturing of pharmaceutical drugs. It is highly volatile, and so it must be handled with extreme care. It has a vapor pressure of .53 atm at 24.10C. 35 grams of an unknown chemical with pain relieving qualities was dissolved in 145 grams of dichloromethane at 24.10C. The vapor pressure was reduced to .47 atm. What is the molar mass of the unknown compound? 45. What is the molarity of a solution that contains 245 grams of hexane (C6H14) dissolved in 4500 grams of carbon tetrachloride? What would be the molality of the same solution? The density of CCl4 is 1.594 g/mL. 46. An unknown chemical is found at a drug bust, and 3.55 grams of the substance are dissolved in 48.0 grams of diethyl ether (C4H10O) at the crime lab. After analysis, it is found the boiling point of the solution is now 39.0 0C. What is the molar mass of the chemical found at the crime scene? Diethyl ether normally boils at 34.6 0C, and has a Kb of 2.11 oC/m. Assume the unknown chemical is covalent. 47. A solution contains .0653 g of a compound in 9.75 g of ethanol. The molality of the solution is .0368 m. Calculate the molar mass of the compound. 48. Carbon disulfide, used to make rayon, is volatile and extremely toxic to the central nervous system. It has a high vapor pressure of 352.6 mm Hg at 250C. When 4.567 grams of an unknown powder that appears to be testesterone is taken from a baseball player and dissolved in 6.46 grams of CS2 at 250C, the vapor pressure drops to .391 atm. What is the molar mass of the unknown powder? Look up the molar mass of testosterone – is he guilty? 49. A sample of a chemical that looks like Ritalin was taken from a college student suspected of taking it to get high. 140.28 grams of the powder was dissolved in 250 grams of water, and the freezing point dropped to –3.900C. What is the molar mass of the unknown substance? Assume the unknown chemical is a covalent substance! 50. Sodium phosphate is a compound frequently used as a laxative before colonoscopies. What is the boiling point of an aqueous solution of sodium phosphate that contains .30-mole fraction of this substance?