Supplementary material - orna elroy stein laboratory
advertisement

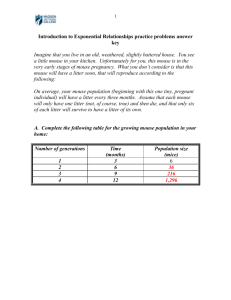
Mouse model for eIF2B-leukodystrophy Supplementary material Materials and Methods (continued) eIF2B activity assay Cerebrums of 3-week-old mice were homogenized by brief sonication in ml ice-cold buffer consisting of 45 mM HEPES, pH 7.4, 0.375 mM magnesium acetate, 95 mM potassium acetate, 10% glycerol, 2% triton X-100, 1mM DTT, EDTA-free protease inhibitor cocktail (Complete; Roche), 1gml pepstatin, 1gml leupeptin, 0.3microcystin, 10 mM NaF, 10 mM -glycerolphosphate followed by centrifugation at 10,000·g for 10 min at 40C. The resulting supernatants were assayed for exchange of [3H]GDP bound to eIF2 purified from rat liver for unlabelled GDP, as described previously (Kimball et al., 1989). Briefly, 5l supernatant was added to a mixture containing eIF2[3H]GDP complex and a 100-fold excess of non-radiolabeled GDP. The mixture was placed in a water bath at 300C and at 0, 2, 4, 6 min incubation times, 60l aliquots of the reaction mixture were removed and filtered through a nitrocellulose filter disk. Radiation associated with the filter was measured by liquid scintillation spectrometry. Each assay was performed in triplicates. Polysomes profile analysis Polysomal profiles were performed as described previously (Sivan et al., 2007) with modifications. Specifically, 1 ml of buffer containing 18 mM Tris, pH 7.5, 50 mM KCl, 10 mM MgCl, 10 mM NaF, 20 mM -glycerolphosphate, 4 g/ml pepstatin, 2 g/ml leupeptin, EDTA-free protease inhibitor cocktail [Complete; Roche], 140 g/ml cycloheximide, 2.5 mM dithiothreitol, and 1 mg/ml heparin, was added to each P1 mouse brain lacking the cerebellum. The brain was homogenized by 30 strokes in an eppendorf tube followed by addition of Triton X-100 and deoxycholate to a final concentration of 1.2% each and incubation on ice for 5 min. Followed 3 min spin at 20,000xg in a cold eppendorf centrifuge, twenty three optical density units (260 nm) of the supernatant were loaded on each 10-50% sucrose gradient. After centrifugation of 2 hr in 39,000 rpm at 4oC, the gradients were resolved using Teledyne ISCO model UA6 at sensitivity of 0.5. Mouse model for eIF2B-leukodystrophy Electron Microscopy Three 3-week-old mice from each group were sacrificed by a lethal dose of 10% Ketamine and 6.6% Xylazine in phosphate-buffered saline (PBS) and were transcardial perfused with PBS followed by fixation buffer containing 2% Glutaraldehyde, 1% paraformaldehyde in PBS. Brains were removed from the skull and postfixed in fixation buffer for over 48 hours at 4°C prior to dissection of 2 different areas the internal capsule tissue from each lobe. The tissues were fixed in 1% osmium tetroxide in PBS for 2 hours at 4°C, dehydrated in an ascending ethanol series (25%, 50%, 75%, 95% and 100%) and embedded in glycidether (Serva Feinbiochemica Heidelberg, Germany). Serial ultrathin sections cut with a Leica ultramicrotome (70-100 nm) were mounted on copper grids and treated with 2% uranyl acetate and 0.3% led citrate in 30% ethanol. Sections were visualized by JEOL 1200EX (Japan) transmission electron microscope (TEM) using magnification of X6000. The cross-section area of 3287 (WT) and 3809 (KI) axons was measures using Scion Image software (alpha 4.0.3.2). The software was employed to calculate the inner diameter (axon without myelin) and the diameter of the entire fiber containing the myelin sheaths. The G-ratio (axon/fiber diameter) was calculated using only axons whose cross section was circular. 100 axons per group were used for G-ratio calculation, followed by t-test. Motor function assays Rotarod: male mice were pretrained for 4 consecutive days starting from the age of 3 weeks and then subjected at the age of 4, 6, 8, 13, 16 and 40 weeks to a rotarod (ENV-576M, Med Associates Inc.) assay, using 16 rpm without acceleration at 480 sec limit. Open field: 3-months old mice were acclimated for a week in a quiet room with 12h light / 12h dark cycle and then videotaped (Ikegami B/W ICD-47E infrared-sensitive video camera) for 30 minutes in a 2m x 2m arena enclosed with a 0.5m-high opaque Plexiglas walls and a blue PVC floor. All testing took place between 18-22 hrs, which were the first hours of the dark phase. During testing, the room was illuminated with an infrared light (Tracksys, IR LED Illuminator; UK) with 830 nm filter light that emit light not visible to rodents. Video recording were then analyzed using a tracking system (Ethovision, by Noldus , NL) yielding the overall distance traveled by each mouse. Fat mass analysis 2 Mouse model for eIF2B-leukodystrophy Fat mass was measured using Double X-ray absorption (DXA; Lunar Piximus II) using 5 WT and 7 mutated 90-weeks old male mice which were anaesthetized with inhaled isoflurane & oxygen as previously described (Gutman et al., 2006). Histochemistry and Immunostaining Male mice were anesthetized with ketamine/xylazine and perfused transcardially with PBS followed by PFA fixation solution (4% paraformaldehyde in 0.1 M phosphate buffer, pH 7.4). Brains were removed, incubated overnight in PFA fixation solution followed by incubation in 30% sucrose for 48h. Frozen coronal sections (30 m) were cut using a sliding microtome and collected serially. For Luxol Fast Blue (LFB) staining, the free-floating sections were mounted on slides and gradually dehydrated by 2 min incubations in each of 50%, 70% and 95% ethanol. 0.1% LFB (solvent blue 38; Sigma, #S3382) in 95% ethanol-0.5% glacial acetic acid was then added for further incubation overnight at 56oC, followed by rinse in 95% ethanol and then in water. The slides were differentiated by sequential 1.5 min incubations in 0.05% lithium-carbonate in water, followed by 70% ethanol and finally in water. The slides were gradually dehydrated by 5 min incubations in each of 70%, 95% (twice), 100% (twice) ethanol and then in Xylen, followed by application of histomount (Invitrogen #00-8030) and cover slips. For immunostaining, the free-floating sections were washed with 100 mM PBS pH 7.4 prior to the addition of antibodies diluted in PBST (PBS containing 0.1% Triton X-100) and 2% horse or goat serum. The following antibodies were used: 1:200 AA3 monoclonal anti-PLP (residues 264–276) (Yamamura et al., 1991); 1:500 anti-MBP Ab1 (Neomarkers); 1:500 anti-NG2 (Chemicon); 1:2000 anti GFAP (Dako). Initial 30 min incubation at 37oC was followed by overnight incubation at 4°C. After PBST rinse, sections were incubated for 60 min at room temperature with 1:200 diluted secondary antibody. For immunofluoresence, Cy3-conjugated affinity pure goat anti rabbit IgG (Jackson) was used, followed by rinse in PBST and mounting with fluoromount (Sigma). Images were visualized by LSM 510 META (Zeiss). For immunohistochemistry, biotynilated antimouse or anti-rabbit IgG, (Vector Labs) were used and after several PBST rinses, sections were further incubated for 30 min in avidin-biotin-horseradish peroxidase complex (ABCElite; Vector Laboratories) in PBST. After PBST rinse, sections were soaked in diaminobenzidine chromagen solution (Vector Labs). The reaction was monitored visually and stopped by PBS rinses. To minimize 3 Mouse model for eIF2B-leukodystrophy variability, sections from all animals were stained simultaneously. Following dehydration and mounting using histomount (Invitrogen), the peroxidase-immunostained sections were photographed using a 40X, 10X and 2X objectives and a Nikon DS-FI1 camera (Nikon Instech, Tokyo, Japan), followed by measurements of integral optical density (IOD) and total stained area using Image-Pro Plus software (version 5.1 Media Cybernetics, Silver Spring, MD). Other than moderate adjustments of contrast and brightness, the images were not manipulated. Statstical comparison was performed using student's t-test (1 tailed distribution with equal variance). Western blot analysis Proteins were extracted from each individual brain by brief sonication in lysis buffer containing 20mM Hepes pH 7.7, 100mM KCl, 10% glycerol, 2% Triton X-100, 20mM -glycerolphosphate, EDTA-free protease inhibitor cocktail [Complete; Roche], and 1 mM dithiothreitol. Concentration was determined by BCATM protein assay kit (Thermo scientific), followed by analysis of equal amounts of total proteins on 15% SDS-PAGE. Western blot analysis was performed using 1:100 AA3 monoclonal anti-PLP (residues 264–276) (Yamamura et al., 1991); 1:200, anti MBP Ab1 (Neomarkers), anti -tubulin (Sigma #T4026), 1:10,000 p38 MAPK (Sigma #M0800), and 1:1000 GFAP (BD Pharmingen #556330). Analysis of Eif2b5 was performed following separation on 10% SDS-PAGE using antibody #SC28854 from Santa Cruz. MRI Mice were scanned in a 7T/30 spectrometer (Bruker) under ~1.5% isoflurane anesthesia. The MRI protocol included conventional anatomy sequences (T2) and diffusion tensor imaging (DTI). T2 Protocol: Multi-slice Multi-Echo (MSME) sequence acquired with TR of 3000 ms and multi TE starting from 10ms and incremented in 10ms intervals up to 120ms. Geometrical parameters: 12 coronal slices with resolution of 0.075x0.120x1 mm3 (FOV of 19.2 mm2 and matrix of 256x160) were acquired. The images were reconstructed to final image dimensions of 256x256 with a resolution of 0.075x0.065x1 mm3. DTI protocol: Spin-echo diffusion-weighted echo planar imaging (DW-EPI) sequence acquired with TR of 2500ms, TE of 22ms, =10/4ms, b value of 1000 s/mm2 measured along 16 gradient directions. Geometrical parameters: 12 coronal slices with a resolution of 4 Mouse model for eIF2B-leukodystrophy 0.240x0.240x1 mm3 (FOV of 19.2 mm2 and matrix of 80x80). Total acquisition time was about 30 minutes per mouse. Image Analysis: Correction of head motion image artifacts, normalization and statistical analysis were performed using the statistical parametric mapping (SPM) software (version 2, UCL, London, UK). The image analysis routine includes the following steps: 1. DTI analysis from which FA, ADC, axial (1) and radial (3) diffusivity maps were computed (Pierpaoli and Basser, 1996); 2. Spatial normalization using 12-parameter affine non-linear transformation to mouse brain template (Paxinos and Franklin, 2001); 3. Spatial smoothing with 0.3mm full width half maximum Gaussian kernel. The normalization and smoothing were applied on each of the DTI output maps (FA, ADC, 1 and 3). Statistical Analysis for MRI: following transformation of the images into a standard space (spatial normalization), detection of regionally specific differences was done on a voxel-byvoxel basis (Ashburner and Friston, 2000; Crum et al., 2003). In this study, ANOVA was used to compare WT and mutant groups at different ages, combined with a correlation test within each group with age as covariate. Using these analyses, we estimated areas of specific differences between the groups (ANOVA) and areas of developmental signal changes (correlation with age). Following the voxel-based analysis, in clusters of voxels that passed the statistical threshold of p<0.01 (that were within the territory of internal capsule or cerebral peduncle), voxel of interest (VOI) analysis was performed. The VOI were chosen, per each cluster, centered at the voxel with the highest t-value and collected from voxels around it in a 0.4mm sphere (~20 voxels). The DTI indices (FA, ADC or principal diffusivities) were averaged within each VOI and used to graphical presentation and further analysis. 5 Mouse model for eIF2B-leukodystrophy Supplemental Figure 1: Polysomal profiles of WT and mutant mice brain. Each brain (lacking the cerebellum) of P1 WT or mutant (Mut) mice was homogenized as detailed in Materials and Methods, followed by analysis of 23 optical density units (260 nm) on 10-50% sucrose gradient. The gradients were resolved using Teledyne ISCO model UA6 at sensitivity of 0.5. The 40S, 60S, 80S and polysomes peaks are indicated. 6