Cynthia Ju, Ph.D.
CURRICULUM VITAE
Cynthia Ju, Assistant Professor
Department of Pharmaceutical Sciences
School of Pharmacy
University of Colorado Denver
CONTACT INFORMATION
Mail Stop C238-P15
Research Complex 2
12700 East 19th Avenue, Room P15-3007
Aurora, CO 80045
Phone: 303-724-4019
Fax: 303-724-7266
E-mail: Cynthia.Ju@UCDENVER.EDU
EDUCATION
1991
1995
1999
B.Sc. Chemistry, Peking University, Beijing, China
M.Sc. Bio-organic Chemistry, University of Victoria, British
Columbia, Canada
Ph.D. Drug Metabolism & Toxicology, University of Toronto,
Ontario, Canada
PROFESSIONAL EXPERIENCE
1999 - 2002
2002 – present
Post-doctoral Research Scientist, Molecular and Cellular
Toxicology Section, Laboratory of Molecular Immunology,
NHLBI, National Institutes of Health, Bethesda, Maryland.
Assistant Professor, Department of Pharmaceutical Sciences,
University of Colorado Health Sciences Center, Denver, Colorado.
ACADEMIC APPOINTMENTS
2002 – present
2005 – present
Assistant Professor, Department of Pharmaceutical Sciences,
University of Colorado Health Sciences Center, Denver, Colorado.
Faculty member, Integrated Department of Immunology, National
Jewish Medical and Research Center, Denver, Colorado.
CONSULTING ACTIVITY
06/2006
10/2004
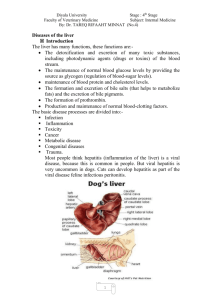
Discussion on current understanding of drug-induced liver
injury, Merck Inc., West Point, PA and Raway, NJ.
Discussion on studying the mechanisms of idiosyncratic drug
reactions. Pfizer Inc, Ann Arbor
Cynthia Ju, Ph.D.
10/2004 – present
Discussions on developing strategies to evaluate drug
candidates’ potential of causing immune reactions. Pfizer
Inc., Monthly conference calls
TEACHING ACTIVITY
Professional Program Courses
2003 – present
PHRD 4750, Integrated Organ System VI
2003 – present
PHRD 4200, Instructional Methods II
Graduate Program Courses
2003 – present
TXCL 7322, Reactive Metabolites and Immunology
2004 – present
TXCL 7325, Current Topics in Toxicology Research
2007 – present
PHSC&TXCL 7310, Fundamentals of Pharmaceutical Sciences
ADVISING ACTIVITY
Postdoctoral Fellows/Professional Research Assistants
2004 – present
Qiang You (Ph.D.)
2004 – present
Linling Cheng
Thesis Advisor for Graduate Students
2004 – present
Hao Yin
2004 – present
Michael Holt
2007 – present
Brittany Martin
Graduate Student Committee Membership
2003 – 2008
Benjamin Stewart
2004 – 2005
Katie Peebles
2004 – 2005
Ileana Vargas
ADMINISTRATIVE ACTIVITY AND UNIVERSITY SERVICE
2006 – present
2008
2007 – present
2003 – 2006
2003 – 2006
Associate Director of the Toxicology Graduate Program, DOPS
Member, search committee, Associate Dean for Professional
Education, SOP
Member, Student Affairs Committee, SOP
Member, Research Committee, DOPS
Member, Toxicology Graduate Program Curriculum, DOPS
PROFESSIONAL ACTIVITY AND PUBLIC SERVICE
Membership in Professional Organizations
2002 – present
American Association of Colleges of Pharmacy
2002 – present
Society of Toxicology
2003 – present
American Association of Immunology
2005 – present
Society of Chinese Biological Science Investigators
Journal Referee (ad hoc)
Cynthia Ju, Ph.D.
Hepatology; Chemical Research in Toxicology; American Journal of Pathology;
Chemico-Biological Ineraction; Experimental Biology and Medicine; International
Immunopharmacology; Biochemical Pharmacology
SPEAKING ENGAGEMENTS
On Campus
12/2002
Department of Clinical Immunology Seminar Series (UCHSC),
Denver, CO. Title: Potential Role of the Liver in Preventing Adverse
Reactions Against Drug-Protein Adducts.
6/2003
Department of Pharmaceutical Sciences Annual Retreat (UCHSC),
Winter Park, CO. Title: The Protective Role of Hepatic Kupffer Cells
in Idiosyncratic Drug Reactions.
National and International
06/2008
Society of Toxicology Pathology, San Francisco, CA. Title: Role of
Hepatic Macrophages in Regulation of Idiosyncratic Drug Reactions.
05/2007
Discussion Leader, Experimental Biology, Washington D.C. Topic:
Mechanisms of Idiosyncratic Drug Reactions
06/2007
Gordon Conference on Adverse Drug Reactions, Waterville, MN.
Title: Role of Hepatic Macrophages in Drug-Induced Liver Injury.
08/2007
American Society of Chemistry, Boston, MT. Title: Role of the Innate
Immune System in Drug-Induced Liver Injury
06/2006
Merck Inc., West Point, PA and Raway, NJ. Title: Role of the Innate
Immune System in Drug-Induced Liver Injury.
03/2006
Society of Toxicology, San Diego, CA. Title: The Role of Kupffer
Cells in Immune-Mediated Adverse Drug Reactions.
10/2005
International Society of the Study of Xenobiotics, Maui, Hawaii.
Title: The Role of Kupffer Cells in Drug-Induced Immune-Mediated
Reactions.
06/2005
Discussion Leader, Gordon Conference on Adverse Drug Reactions,
Connecticut College, CT. Topic: Basic Immunology with Relevance
to Adverse Drug Reactions
12/2004
Shanghai Immunology Institute, Shanghai, China. Title: The
Protective Role of Kupffer Cells in Drug-Induced Liver Injury.
11/2004
Idiosyncratic Toxicity: Understanding, Prediction and Prevention,
Philadelphia, PA. Title: Protective Role of Hepatic Kupffer Cells in
Idiosyncratic Drug Reactions.
10/2004
Pfizer Inc., Ann Arbor, MI. Title: Studying the mechanisms of
Idiosyncratic Drug Reactions.
06/2004
Society of Toxicology Pathology, Salt Lake City, UT. Title:
Protective Role of Hepatic Kupffer Cells in Idiosyncratic Drug
Reactions.
05/2004
Drug Hypersensitivity Meeting, Bern, Switzerland. Title: The
Tolerogenic Role of Hepatic Kupffer Cells in Idiosyncratic Drug
Reactions.
Cynthia Ju, Ph.D.
04/2004
07/2002
Reactive Metabolites and Drug Toxicity, ACS Drug Metabolism
Discussion Group, Summerset, NJ. Title: Protective Role of Hepatic
Kupffer Cells in Adverse Reactions Against Drug-Protein Adducts.
Gordon Conference on Drug Metabolism, New Hampshire, MA.
Title: Potential Role of the Liver in Preventing Immune Reactions
Against Drug-Protein Adducts.
GRANT/RESEARCH ACTIVITY
Current Projects
RO1 ES012914
PN200509-221
F31 DK082269
Fellowship
Title: The Immunosuppressive Role of Kupffer Cells in
Xenobiotic-Induced Adverse Reactions
Agency: NIH
Principal Investigator: Cynthia Ju
Direct Costs: $190,000 / year
Period: 06/06/2005 – 03/31/2010
Title: Developing a Screen Test to Evaluate Drug Candidates’
Potential of Causing Immune Reactions
Agency: Pfizer Inc.
Principal Investigator: Cynthia Ju
Direct Costs: $150,000 / year
Period: 11/01/2005 – 10/31/2009
Title: Investigation of the Role of Damaged-Associated Molecular
Pattern (DAMP) Molecules in the Pathogenesis of Drug-Induced
Liver Injury.
Agency: NIH
Fellowship Recipient: Brittany Martin
Direct Costs: $27,404
Period: 07/01/2008 – 06/30/2013
Title: Role of Hepatic Macrophages in Drug-Induced Liver Injury
Agency: PHRMA Foundation
Fellowship Recipient: Michael Holt
Direct Costs: $20,000
Period: 08/01/2007 – 07/31/2009
Completed Projects
R21 DK068171
Title: Role of 15d-PGJ2 in the Protective Effects of Hepatic
Macrophages
Agency: NIH
Principal Investigator: Cynthia Ju
Direct Costs: $100,000 / year
Period: 7/1/04 – 6/30/06
Seed Grant
Title: Chemopreventive Effect of Silibinin against Hepatocellular
Carcinoma
Agency: Cancer Center, UCHSC
Principal Investigator: Cynthia Ju
Cynthia Ju, Ph.D.
Period: 2/1/04 – 1/31/05
Direct Costs: $13,334
Seed Grant
Title: Mechanism of Kupffer Cell-Mediated Protection Against
Acetaminophen-Induced Liver Injury in Mice
Agency:Department of Pharmaceutical Sciences, UCHSC
Prinicipal Investigator: Cynthia Ju
Period: 1/1/03 – 12/31/03
Direct Costs: $10,000
PUBLICATIONS
Refereed Journals published papers
L. Cheng, Q. You, H. Yin, M.P. Holt, C. Franklin, and C. Ju (2009) “The Effect of
PolyI:C Co-treatment on Halothane-Induced Liver Injury in Mice”, Hepatology, 49, 215226.
M.P. Holt, L. Cheng, and C. Ju (2008) “Identification and Characterization of Infiltrating
Macrophages in Acetaminophen-Induced Liver Injury”, J. Leukoc. Biol., 84, 1410-1421.
Q. You, L. Cheng, R.M. Kedl, and C. Ju (2008) “Mechanism of T cell Tolerance
Induction by Hepatic Kupffer Cells”, Hepatology, 48, 978-990.
L. Cheng, B.J. Stewart, Q. You, D.R. Petersen, J.A. Ware, J.R. Piccotti, T.T. Kawabata,
C. Ju (2008) “Covalent Binding of the Nitroso Metabolite of Sulfamethoxazole Is
Important in Induction of Drug-Specific T Cell Responses In Vivo”, Molecular
Pharmacology, 73: 1769-1775.
H. YIN, L. Cheng, R. Langenbach, and C. Ju (2007) “Prostaglandin I2 and E2
Mediate the Protective Effects of Cyclooxygenase-2 in a Mouse Model of ImmuneMediated Liver Injury”, Hepatology, 45: 159-169.
Q. You, L. Cheng, T. P. Reilly, D. Wegmann and C. Ju (2006) “Role of Neutrophils
in a Mouse Model of Halothane-Induced Liver Injury”, Hepatology, 44: 1421-1431.
C. Ju, J.P. McCoy, C.J. Chung, M.L.M. Graf, and L. R. Pohl (2003) “Tolerogenic
role of Kupffer cells in allergic reactions”, Chem. Res. Toxicol. 16: 1514-1519.
C. Ju, T. P. Reilly, M. Bourdi, M. F. Radonovich, J.N. Brady, J. W. George and L. R.
Pohl (2002) “Protective role of Kupffer cells in acetaminophen-induced hepatic injury in
mice”, Chem. Res. Toxicol. 15: 1504-1513.
C. Ju and L. R. Pohl (2001) “Immunohistochemical detection of protein adducts of 2,4dinitrochlorobenzene in antigen presenting cells and lymphocytes after oral
administration to mice: lack of a role of Kupffer cells in oral tolerance”, Chem. Res.
Toxicol. 14: 1209-1217.
C. Ju and J. P. Uetrecht (1999) "Oxidation of 2-hydroxyiminostilbene, a metabolite of
carbamazepine, to a reactive iminoquinone intermediate and detection of the glucuronide
conjugate of 2-hydroxyiminostilbene in patient urine", J. Pharm. Exp. Ther. 288: 51-56.
Cynthia Ju, Ph.D.
C. Ju and J. P. Uetrecht (1998) "Oxidation of a metabolite of indomethacin (DMBI) to
reactive intermediates by activated neutrophils, HOCl and the myeloperoxidase system”,
Drug Metab.Dispos. 26: 676-680.
C. Ju and C. Bohne (1996) "Dynamics of probe complexation to bile salt aggregates", J.
Phys. Chem. 100: 3847-3854.
C. Ju and C. Bohne (1996) "Probing bile salt aggregates by fluorescence quenching",
Photochem. Photobiol. 63: 60-67.
Review Articles
C. Ju (2008) “The Role of Hepatic Macrophages in Regulation of Idiosyncratic Drug
Reactions”, Toxicologic Pathology, Accepted.
M.P. Holt and C. Ju (2006) “Mechanisms of Drug-Induced Liver Injury”, AAPS
Journal, 8(1), E48-54.
R.A. Roberts, P.E. Ganey, C. Ju, L.M. Kamendulis, I. Rusyn, and J.E. Klaunig
(2006) “Role of the Kupffer Cells in Mediating Hepatic Toxicity and
Carcinogenesis”, Toxicological Sciences.
C. Ju and Lance R. Pohl (2005) “Tolerogenic role of Kupffer cells in immunemediated adverse drug reactions”, Toxicology, 209: 109-112.
C. Ju (2005) “Immunological mechanisms of drug-induced liver injury”, Current
Opinion in Drug Discovery and Development, 8: 38-43.
T. P. Reilly and C. Ju (2002) “Mechanisms responsible for sulfonamide-induced
cutaneous reactions”, Curr. Opin. Allergy Clin. Immunol. 2: 307-315.
C. Ju and J. P. Uetrecht (2001) “Mechanism of idiosyncratic drug reactions: reactive
metabolite formation, protein binding and the regulation of the immune system”, Curr.
Drug Metab. 3: 367-377.
Book Chapters
M.P. Holt, and C. Ju (2008) “The Adaptive Immune System and Liver Toxicity”. In:
Hepatic Toxicology. Editors: R.A. Roth and P.E. Ganey. Publisher: Elsevier. Accepted.
M.P. Holt, and C. Ju (2008) “Organ Specific Toxicity”. In: Molecular Toxicology –
Prediction of Toxicity Parameters. Editors: B.K. Park, D. Williams, and D. Antoine.
Publisher: Wiley-VCH. Submitted.
M.P. Holt, and C. Ju (2008) “Drug-Induced Liver Injury”. In: Mechanisms of Adverse
Drug Reactions. Editor: J.P. Uetrecht. Publisher: Springer-Verlag. Submitted.
 0
0