Enhancing the Production of New Neurons to Promote Neurological
advertisement

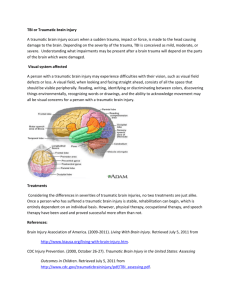
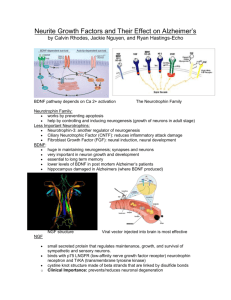
Enhancing the Production of New Neurons to Promote Neurological Recovery Following Traumatic Brain Injury Enhancing the Production of New Neurons to Promote Neurological Recovery Following Traumatic Brain Injury Chief Investigator: Associate Professor Cristina Morganti-Kossmann, Dr Nicole Bye Associate Investigators: Professor Perry Bartlett, Professor Esther Shohami, Associate Professor Ann Turnley Lead Organisation: Monash University TAC Neurotrauma Funding: $506,996 Project Start Date: 1 March 2011 Project Summary: Traumatic brain injury (TBI) is a devastating condition leading to progressive neuronal loss and consequent neurological deficit. Numerous clinical trials targeting neurotoxic cascades in TBI patients have failed pharmacological translation. Recently, a novel opportunity for therapeutic intervention has been identified with the discovery of resident neuronal stem cells in the adult brain that have the capacity to undergo neurogenesis to generate new neurons. Our preliminary studies have shown that immature neurons are indeed generated in response to TBI and, for the first time, that these cells migrate into damaged regions of the brain. Other investigations, including our own, have shown that neurogenesis can be stimulated by administering factors that selectively promote specific regulatory stages. We recently demonstrated that the neuroprotective cytokine erythropoietin (EPO) is a potent stimulator of proliferation of precursor cells, while brain-derived neurotrophic factor (BDNF) is essential for their differentiation into neurons. Moreover, we have shown that treatment with EPO leads to improved motor and memory function after TBI, however, neurogenesis was not investigated. Based on this work, the proposed studies will explore whether treatment with EPO and BDNF, individually or combined, enhances the neurogenic response occurring after TBI, and whether enhanced neurogenesis contributes to improved neurological recovery in TBI mice. In rodent models of TBI, robust evidence supports an undeniable association between increased neurogenesis and improved neurological function, however, the actual contribution of new neurons to recovery post-TBI has never been definitively shown. In order to address this issue, we have designed experiments aimed at elucidating the role of neurogenesis in post-TBI recovery by using our recently developed genetically modified mouse strain, in which neurogenesis can be specifically ablated. This new strain will allow us to directly determine for the first time whether new neurons are necessary for neurological recovery following TBI with and without growth factor treatment. This study will have a significant impact in the neurogenesis and TBI fields and may set the ground for new therapies with potential translation into the clinic. The overall hypotheses of this project are: 1. Endogenous neurogenesis induced after TBI directly contributes to functional recovery. 2. Administration of EPO and BDNF improves motor and cognitive recovery following TBI by enhancing neurogenesis. This study will be the first to establish the contribution of neurogenesis to neurological recovery after TBI. If neurogenesis is a mechanism leading to improved cognition and motor function following trauma, we will have identified this process as an important new target for the development of regenerative therapies. Furthermore, the neurotrophin administration as proposed here has an imminent relevance to human TBI, as it explores a novel pharmacological paradigm to direct and augment brain repair and neurological recovery, which could potentially be implemented in the clinic. Publications: (as at Sept 2012) ZIEBELL JM, BYE N, SEMPLE BD, KOSSMANN T, MORGANTI-KOSSMANN MC. Attenuated neurological deficit, cell death and lesion volume in Fas-mutant mice is associated with altered neuroinflammation following traumatic brain injury. Brain Res 1414: 94-105. BYE N, CARRON S, HAN X, AGYAPOMAA D, NG SY, YAN E, ROSENFELD JV, MORGANTIKOSSMANN MC. Neurogenesis and glial proliferation are stimulated following diffuse traumatic brain injury in adult rats. J Neurosci Res 89:986–1000. BYE N, Turnley AM, Morganti-Kossmann M.C. Inflammatory regulators of redirected neural migration in the injured brain. NeuroSignals 20(3):132-146. HELEWELL SC. MORGANTI-KOSSMANN MC. Guilty Molecules, Guilty Minds? The Conflicting Roles of the Innate immune responses to traumatic brain injury? Mediators of Inflammation: Volume 2012:356494. BYE N, TURNLEY AM, MORGANTI-KOSSMANN MC. Inflammatory regulators of redirected neural migration in the injured brain. NeuroSignals, 2012;20(3):132-46. NG SY, SEMPLE BD, MORGANTI-KOSSMAN MC, BYE N. Attenuation of microglial activation with minocycline is not associated with changes in neurogenesis after focal traumatic brain injury in adult mice. J Neurotrauma 29:1410-1425. Presentations: (as at Sept 2012) BYE N, ROSENFELD JV, MORGANTI-KOSSMANN MC. Traumatic brain injury stimulates production of new immature neurons, but their survival may require exogenous neurotrophic support. Trauma, 2011 November, Melbourne Australia. HELLEWELL S, YAN E, MORGANTI-KOSSMANN MC. EPO ameliorates axonal damage, attenuates macrophage infiltration and restores motor function in a combined model of TAI and hypoxia. 9th World Congress on Brain Injury 2012 March 21-25, Edinburgh, UK. YAN E, SATGUNASEELAN L, BYE N, ROSENFELD J, MORGANTI-KOSSMANN MC. A hypoxic insult in patients with traumatic brain injury enhances cerebral inflammatory cytokines, serum biomarkers and blood-brain barrier dysfunction associated with unfavourable outcome. 9th World Congress on Brain Injury 2012 March 21-25, Edinburgh, UK. BYE N, ROSENFELD JV, MORGANTI-KOSSMANN MC. Traumatic brain injury stimulates production of new immature neurons, but their survival may require exogenous neurotrophic support. 9th World Congress on Brain Injury 2012 March 21-25, Edinburgh, UK. BYE N, CONQUEST A, ROSENFELD JV, MORGANTI-KOSSMAN MC. Traumatic brain injury stimulates production of new immature neurons, but their survival may require exogenous neurotrophic support. Alfred Medical Research and Education Precinct (AMREP). BYE N. Stimulating neurogenesis to enhance brain repair and neurological function after brain injury. World Federation for NeuroRehabilitation’s 7th World Congress for Neurorehabilitation, 2012 May 17-19, Melbourne Convention Centre, Melbourne, Australia.