Lab on dissolved Oxygen
advertisement

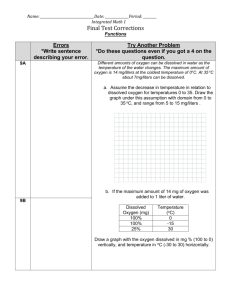
ENGINEERING SCIENCE DEPARTMENT Water and Environmental Technology Program Name _________________ SPRING TERM Aquatic Chemistry II, WQT-134 Laboratory Dissolved Oxygen Purpose: In this lab you will use a Hach dissolved oxygen probe and a Luminescent Dissolved Oxygen (LDO) probe, to perform an assessment of the dissolved oxygen content of a water/waste water sample. Dissolved oxygen analysis measures the amount of gaseous oxygen (O2) dissolved in an aqueous solution. Oxygen gets into water by diffusion from the surrounding air, by aeration (rapid movement), and as a waste product of photosynthesis. Oxygen is one of the most important tests that can be used to determine the quality of water. Dissolved oxygen content can be used as an indication of the effects of waste on a body of water, purity of water, and suitability of the water for fish and other organisms. For example, dissolved oxygen levels of 4 to 5 mg/L are considered borderline for extended survival of aquatic plants and animals. Ideally, most game fish, require levels of 8 to 15 mg/L. Conditions which affect the dissolved oxygen level include: flow rate, photosynthesis, aerobic respiration, elevation, clarity, temperature, sludge deposits, and water depth. Because plants are not producing oxygen during the night, but oxygen is still being used for respiration at that time, dissolved oxygen concentrations will be the lowest within a waterbody just before sunrise. The colder the water, the greater capacity it has to hold oxygen. Atmospheric pressure effects the amount of oxygen dissolved in water. At higher elevations, less oxygen can be dissolved in water. Oftentimes, warm turbid water will have lower DO levels. In addition, nutrient rich water has a greater opportunity for plant growth. With more plants, there will be more photosynthesis, but also more respiration. This means that dissolved oxygen concentrations may be very low in the morning, and very high in the afternoon. In this investigation, you will measure and analyze the dissolved oxygen (DO) concentration in your unknown water samples. Factoids/Conversions Dissolved oxygen content for aquatic life = ~10 mg/L Dissolved oxygen content to stress aquatic life = ~<5 mg/L 21% DO level at sea level An atmosphere is considered oxygen deficient when the oxygen level drops below 19.5% DO As temperature goes down, dissolved oxygen goes up. Solubility of dissolved oxygen also decreases as salinity increases. Solubility of dissolved oxygen into water increases with pressure Advantages No pretreatment necessary for well or potable water samples Ease of use accuracy at 1.5%, 0-20 mg/L LDO probe (0-60 mg/L) no membranes and superior accuracy +/- 0.1 mg/L at <8 mg/L +/- 0.2 mg/L at >8 mg/L, +/- 10% reading >20 mg/L, resolution 0.01 mg/L Disadvantages/Interferences Probe can short circuit and drifts Frequent, recalibration Sample collection: Collect 1 liter sample with no headspace. SUPPLIES/SAFTEY Lab Materials: 1. Hach Ion Sension 6 Dissolved Oxygen Meter 2. Hach LDO probe and sensor 3. Beakers 125-, 250 mL SUMMARY PROCEDURE Dissolved Oxygen LDO Probe HQ 10 MEASUREMENT 1. Place the LDO probe into the sample 2. Press the Blue I button to turn the probe on. 3. Press the Blue Check Mark to take a reading the value will fluctuate and the display will show “Stabilizing…mg/L…” The lock ion will appear after the measurement is complete. This may take awhile. Record the value as __________mg/L and hit the O2 % button to record the O2 percentage. 4. Repeat this procedure Step-by-step Procedure Dissolved Oxygen Multi Sensor Probe HQ40d18 MEASUREMENT 5. Place the LDO probe into the sample 6. Press the GREEN/RIGHT key under Read 7. The display will show “Stabilizing…” and a progress bar will fill from 0 to 100% as the probe stabilizes in the sample. The lock ion will appear after the measurement is complete. 8. Repeat this procedure Dissolved Oxygen Hach Ion Sension 6 Dissolved Oxygen Meter Step-by-step Procedure Dissolved Oxygen Calibration Using the Hach Sension6 Series Dissolved Oxygen 1. Please follow along with the instruction book. 2. Fill the module cap 2/3 full with dissolved oxygen electrolyte filling solution 3. Hold the probe in a vertical position with the tip down. 4. Measure 150 ml of sample in a 250 ml beaker with a magnetic stir bar 5. Turn on the meter by pressing I/O button. 6. Press the CAL key Cal? Numerically enter the barometric pressure and elevation. Press the Enter key. 7. The salinity value of 0/00 will be shown. Press the Enter key. The display will show 100% 8. Press the Enter key, stabilization will occur. The meter will return to the read mode. Press the EXIT key to leave calibration and take a measurement. Press the CONC % button to get from % to mg/L. Department of Engineering Science Water & Environmental Technology Environmental Chemistry II, WQT 134 Dissolved Oxygen Analyzed by Date Facility Name Sample Temperature___________________ Sample pH __________________________ 1. Reported sample dissolved oxygen (DO ) concentration : sample location DO concentration mg/L sample location DO concentration mg/L sample location DO concentration mg/L sample location DO concentration mg/L