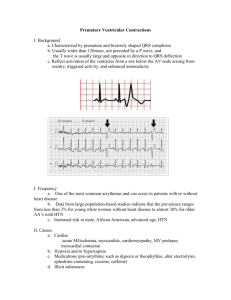
Cardiology
advertisement

2. Acute coronary syndromes Acute coronary syndrome Plaque rupture Partially occlusive No ST elevation ST depressions, T-wave inversions () markers Unstable angina Occlusive ST elevation () markers NSTEMI () markers STEMI Non-Q wave MI Q-wave MI Modified from ACC/AHA Practice Guidelines History Typical angina defined (N Engl J Med 1979;300:1350) substernal chest pain exertional relieved by rest or NTG Exam Assess for hemodynamic stability, check bilateral blood pressures to assess for aortic dissection Check for new S4, new MR murmur secondary to ischemic papillary muscle dysfunction, evidence of CHF (elevated JVP, rales, new S3), Kussmaul’s sign for RV MI EKG STE localize better than ST depressions/T wave inversions Anterior (V1-V4); apical (V5,V6); lateral (I, aVL) Inferior (II, III, aVF), III>II elevation suggests RCA as culprit rather than circumflex Posterior (V7-9, “inverse” of V1-V3) RV (1 mm STE in V4R most predictive of RV infarct). Wellens’s syndrome: pre-anterior wall infarction. Two types of Wellens’s T waves (V1-V3) indicative of critical proximal LAD stenosis: Deeply inverted T wave Biphasic terminal T wave inversion Consider catheterization rather than non-invasive investigation MGH Medical Housestaff Manual 14 2. Acute coronary syndromes Cardiac biomarkers Cardiac troponins more sensitive marker of myocardial damage than CK-MB CK-MB more useful for assessing reinfarction and infarct size Elevated troponin, negative CK-MB Serum marker Time to initial increase Time to peak value Time to return to normal Sens of cardiac enzymes in detecting NSTEMI on arrival 53% 51% 66% >6h after pain onset 91% 94% 100% Recent MI ~2-10 d prior CK-MB 3-12h 24h 48-72h “Microinfarct.” In absence of recent MI, Tn T 3-12h 12-48h 5-14d increased long term risk of death, MI, Tn I 3-12h 24h 5-10d need for urgent revascularization even in modified from N Engl J Med 1997;337:1648 pts with renal failure; however, unclear how to use this information; no clear increase in risk for imminent arrhythmia, no clear need for SDU/CCU monitoring End stage renal disease; estimated that 29% of patients on dialysis have elevated troponin T without evidence of myocardial injury; portends poor prognosis Early risk stratification Among patients with U A/NSTEMI, there is progressive benefit with increased risk from newer, advanced therapies including LMWH, platelet GP IIb/IIIa inhibition, and early invasive strategy/PCI. Rates of all-cause mortality, MI, need for urgent revascularization through 14 days TIMI risk score. 7 independent predictors of outcome (JAMA 2000;284:835) 1. 2. 3. 4. 5. 6. 7. Age >65 >3 risk factors for CAD Prior coronary stenosis > 50 percent ST deviation on admission ECG >2 anginal episodes within 24 hours Elevated cardiac biomarkers ASA use in last 7 days Treatment of ST elevation MI Hemodynamic support: if evidence of cardiogenic shock, institute pressors/inotropes, optimize volume status, consider urgent IABP placement Electrical stability: MGH Medical Housestaff Manual 15 2. Acute coronary syndromes Sustained VT (more than 30 s), consider lidocaine, amiodarone Symptomatic bradycardia, heart block, consider pharmacologic therapy, avoid beta-blockade, institute transcutaneous pacing Reperfusion therapy a. Thrombolytic therapy Indications 1. Angina for at least 30 min and less than 12 hrs AND 2a. ST elevations 1mm in 2 anatomically contiguous leads or 2b. LBBB not known to be old Contraindications Absolute Prior hemorrhagic CVA at any time, nonhemorrhagic CVA within 1 year Known intracranial neoplasm Active internal bleeding (not including menses) Suspected aortic dissection Relative SBP >180 on presentation or chronic HTN Prior non-hemorrhagic CVA at anytime INR >2.0 or known bleeding diatheses Trauma or surgery within 2-4 wks Prolonged CPR (> 10min) Noncompressible vascular punctures (e.g. subclavian lines) Recent internal bleeding (2-4 wks), active PUD If considering SK, prior SK exposure (esp within 2 yrs) Pregnancy Complications—“early hazards” Bleeding Intracranial hemorrhage: increased risk in females, African Americans, the elderly, pts with prior CVA, HTN, wt <70 kg, short stature, supratherapeutic lytic dose Overall incidence 0.75%: 0 RF 0.26%, 1 RF 0.96%, 2 RF 1.32%, 3 RF 2.17% 70% bleeding complication at puncture sites Other Myocardial rupture, reperfusion injury, splenic rupture, aortic dissection, cholesterol emboli Benefit Overall 18% decrease in 35d mortality. No clear mortality benefit with lysis 12-24h after onset of symptoms (LATE, EMRAS trials). Failed lysis Evolving chest pain and ST elevations that persist (at >50% baseline) for 90 minutes after onset of administration of thrombolytics rescue angioplasty MGH Medical Housestaff Manual 16 2. Acute coronary syndromes Thrombolytic agent Dose Alteplase (tPA) “accelerated dosing” Streptokinase (SK)* Reteplase (Retavase) Tenecteplase (TNK-tPA) 15 mg IV bolus 0.75 mg/kg (50 mg) over 30 min 0.5 mg/kg (up to 35 mg) over 60 min (100 mg total in average pt) 1.5 MU IV over 30-60 min *(only lytic agent not routinely dosed with IV heparin) 10 MU IV bolus q 30 min x 2 0.5 mg/kg IV bolus x 1 (max 50 mg) b. Catheter-based reperfusion Primary angioplasty: Consider if contraindications to lysis, anterior MI, pt is s/p CABG, diabetic, or in cardiogenic shock. PTCA with 20% lower rate of combined cardiac endpoint, 65% lower rate of CVA v. lysis when performed at high volume centers (JAMA 1997; 278:2093). Of note: door to balloon time in most trials 1-2h. Opting for angioplasty should not significantly increase time to revascularization. Primary stenting. Lower rate of need for revascularization but equivalent mortality when compared with primary angioplasty alone. Rescue angioplasty. Performed in setting of failed lysis (see above). c. Antiplatelet and antithrombin therapy Drug Dose Aspirin (ASA) Heparin IV (used in conjunction with tPA, rPA, TNK-tPA and PTCA) 162-325 mg po x 1 chewed (trans-buccal mucosal absorption) Bolus 60 U/kg IV (max 4000 U); initial infusion 12 U/kg/hr (max 1000 U/h); note lower dosing compared to previous recommendations PTT goal 50-70 (lower than DVT/PE goal of 65-80) Use “cardiac” heparin sliding scale on order entry The only data in STEMI supports abciximab (ADMIRAL, RAPPORT) 0.25 mg/kg IVB 0.125 mcg/kg/min x 12-24h 180 mcg/kg IVB 2 mcg/kg/min x 72h 0.4 mcg/kg/min x 30min 0.1 mcg/kg/min x 48-96h GP IIb/IIIa inhibitors Abciximab (Reopro) Eptifibatide (Integrilin) Tirofiban (Aggrastat) 4. Adjunctive therapy Drug Dose Oxygen Morphine Keep oxygen sat >93% 2-4 mg IV boluses MGH Medical Housestaff Manual Cautions Unusual to see somnolence/ decreased respiratory drive in STEMI 17 2. Acute coronary syndromes Beta blockers Metoprolol (Lopressor) 5 mg IV q5 min x 3, then 25 mg PO q6h, titrate to HR 55-60 Nitrates IV nitroglycerin ACE-inhibitors Captopril 10-1000 mg/min, titrate to symptom relief/SBP ~100 6.25 mg PO tid, titrate as tolerated to SBP ~100 HR <60, SBP <100, severe CHF, 1 (PR>0.24 s), 2, 3 AVB, severe bronchospastic disease Idiosyncratic profound hypotension/bradycardia. SBP <100, renal failure Treatment of unstable angina (UA)/Non-ST elevation MI (NSTEMI) Drug Dose Aspirin (ASA) Clopidogrel (Plavix) Heparin IV (unfractionated) or LMWH Enoxaparin (Lovenox) Dalteparin (Fragmin) 160-325 mg PO x 1 chewed 300 mg PO loading, then 75 mg PO QD Bolus: 60 U/kg IV (max 4000 U), initial infusion 12 U/kg/hr (max 1000 U/h). PTT goal 50-70 (lower than DVT/PE goal of 65-80). Use “cardiac” heparin sliding scale on computer order entry. Nitrates IV nitroglycerin Nitropaste Beta blockers Metoprolol (Lopressor) Propranolol (Inderal) GP IIb/IIIa inhibitors Abciximab (Reopro) Eptifibatide (Integrilin) Tirofiban (Aggrastat) 1 mg/kg SC bid x 2-8d 120 U/kg SC bid x 5-6d discuss with cardiology before using LMWH given lack of easy reversibility Titrate to symptom relief, SBP ~100 10-1000 mcg/min (<300 mcg/min SDU, >300mcg/min CCU) Sliding scale on computer order entry Titrate as tolerated to HR 55-60, contraindicated in decompensated CHF 5 mg IV q5 min x 3, then 25 mg PO q6h 1 mg IV q5 min x 4, then 20 mg PO q6h The best data is in favor or eptifibatide and tirofiban (PURSUIT, PRISM-PLUS), not abciximab. 0.25 mg/kg IVB 0.125 mcg/kg/min x 18-24h (max 10 mcg/min) 180 mcg/kg IVG 2 mcg/kg/min x 72h 0.4 mcg/kg/min x 30 min 0.1mcg/kg/min x 48-108h Post-catheterization care Complication Bleeding groin Groin hematoma Retroperitoneal hematoma Pseudoaneurysm AV-fistula Distal embolization Finding Overt bleeding Assess for neurovascular compromise of limb Decreased Hct ± back, abd, LE pain; hypotension Imaging none ± groin US Groin: pulsatile mass, tenderness, bruit Groin: continuous bruit ± thrill Diminished distal pulses, decreased cap refill, LE neurologic compromise Groin US MGH Medical Housestaff Manual I abd CT Groin US LE arterial non invasives; angio Management Call cards fellow ASAP, compress Stable in size observation Rapidly expanding cards ASAP Stabilize hemodynamically, transfuse General surgery consult, to OR if unstable or LE neurovasc compromise Vascular surgery for repair IR for graded compression (by US) Vascular surgery observation with repeat imaging v. OR repair Vascular surgery consult ? anticoagulation v. urgent 18 2. Acute coronary syndromes embolectomy Renal insuff. Contrast ATN Chol. emboli Increased Cr ± decreased urine output ± renal u/s if concern for obstruction Optimize volume status Prophylaxis with N-acetylcysteine 600 mg PO bid x 48h peri-cath if inc Cr Livedo reticularis, rash, urine eosinophils Post-stent care Antithrombotics Aspirin 325 mg PO qd Clopidogrel 300 mg PO x 1 (in cath lab) 75 mg PO qd x 1 year, likely up to 4 yrs Heparin IV: cont 48h post-sheath pull if poor angiographic result, otherwise discontinue GP IIb/IIIa: usually continued 12-24h post-procedure Statins Folate, B6, B12 Lowers target vessel revascularization rate (LIPS) Lower restenosis rates independent of homocysteine levels (Swiss Heart Study) Post-stent complications Timing Abrupt closure Subacute thrombotic occlusion Min-24h 1-30d In-stent restenosis Vessel rupture/perforation Distal embolization Incidence Findings Pathophys 1% Severe CP, Dissection, STE on thrombosis 4% vs. <1% with drug- EKG eluting stent 1-6mo ~15-36% vs. Increasing Endothelial <1% to 8% angina proliferation with DES Minutes-48h, rare complication, pleuritic chest pain cardiac tamponade secondary to hemopericardium. Do not perform pericardiocentesis Minutes-48h, common complication, minor ischemic pain without ECG changes Management Contact cards ASAP, urgent revascularization Semi-elective revascularization Contact cards ASAP, stat echo Follow serial ECGs Contacting cardiology: 6-9292 (cardiology page operator) Cardiology patients covered by either “ward service” or “private attending” For post-cath complication, call 6-9292 and ask for “interventional fellow on call.” For urgent overnight assistance, call 4-5110 (Ellison 11) and ask for “access fellow” For stat or weekend echo, call x 6-9292 and ask for “fellow on call for echo” Prior to instituting thrombolysis at MGH, call appropriate cardiology coverage; they may wish to mobilize cath lab instead, re: private patients, call 6-9292 and ask for attending coverage re: ward patients, call 6-9292 and ask for “ward cardiology fellow on call” Roderick Tung, M.D. MGH Medical Housestaff Manual 19 3. Noninvasive testing General considerations Exercise tolerance test with MIBI imaging (ETT-MIBI) is the most commonly used noninvasive risk stratification for patients who are able to ambulate at MGH Adenosine-MIBI, a pharmacologic stress test, is useful for patients who are unable to sufficiently ambulate for an ETT-MIBI. Nuclear imaging (MIBI) is used in nearly all stress tests performed at MGH, though there may be variation in practice at other institutions. In most cases, testing should be performed within 72 hours of presentation for low-risk patients. Exercise tolerance test (ETT) Of choice when patient can ambulate Can do without imaging if no baseline ECG abnormalities (digoxin effect, LVH, LBBB, paced, WPW, ST abnormalities associated with SVT, atrial fibrillation, mitral valve prolapse, severe anemia). RBBB okay for ECG interpretation Advantages: inexpensive, low morbidity, estimates functional capacity, predictor of mortality Indications: diagnosis of CAD, risk assessment and prognosis among patients with symptoms or known CAD, post-MI In low risk patients, testing can be performed when patients have been free of active ischemic or heart failure symptoms for a minimum of 8-12 hours and have ruled out for myocardial infarction After MI, can perform submaximal stress test at about 4-76 days post event (but not necessary in patients who have undergone cardiac catheterization). Submaximal test is generally exercise to predetermined end point, i.e. peak heart rate 120 or 70% of predicted max heart rate, or 5 METs Contraindications include acute coronary syndrome, MI within past 2 days, uncontrolled arrhythmia, acute CHF, severe AS, recent PE/DVT, acute infection, aortic dissection, uncontrolled hypertension (SBP >200 or DBP >110). Sensitivity/specificity: single VD 68/77%, multi VD 81/66%, 3VD or LM 86/53% Patient preparation: if to diagnose in CAD, hold beta-blockers if possible unless the test is solely for functional purposes, NPO within 3 hours of testing and longer if imaging is used (which is generally the case at MGH) Data ST segment is most sensitive for ischemia ST depression of at least 1 mm that is horizontal or downsloping ST depression of at least 2 mm that is upsloping Criteria that further increase probability of ischemia: number of leads, workload at which changes occur, angle of ST slope, time to ST recovery, ventricular ectopy during recovery Location of ECG changes does not localize area of ischemia Age-predicted maximum heart rate (220 age): if 85% is not reached, test is non-diagnostic SBP monitoring: failure of SBP to rise with exercise indicates CAD and test should be terminated Functional capacity: in metabolic equivalents (METs), with >6 indicating good prognosis; failure to achieve 5 METs is associated with a worse prognosis Symptoms: angina, shortness of breath, etc. MGH Medical Housestaff Manual 20 3. Noninvasive testing Data interpretation: 1. normal, 2. abnormal, 3. normal except for, 4. nondiagnostic (<85% max HR and no abnormal ECG changes or baseline ECG changes are present) Prognosis, can use Duke treadmill score = exercise time (min) – (5 max ST deviation in any lead) – (4 angina index). Angina index is no angina = 0, non-limiting angina = 1, angina reason for stopping = 2. See N Engl J Med 1991;325:849. Risk of death Low Moderate High Score +5 10 to +4 <10 Inpatients % of pts 4 yr surv 34% 98% 57% 92% 9% 71% Outpatients % of pts 4 yr surv 62% 99% 34% 95% 4% 79% Nuclear perfusion imaging Improves sensitivity (75-90%) and specificity (65-90%) over standard ETT Indications: assessment of physiologic importance of known CAD, pts with abnormal baseline ECG, major to intermediate clinical predictors, high risk surgeries, unable to exercise Advantages: higher sensitivity, localizes ischemic area, can use pharmacologic agents for pts unable to exercise (see below). Limitations: lower specificity for single vessel disease, low PPV for post-op events (~20%), attenuation from adjoining tissue Two major radiopharmaceuticals are used: thallium-201 and technetium-99m-sestamibi (MIBI) Thallium Produced in cyclotron, 72 hour half life, actively transported into cells, redistributes with time to other cardiac and non-cardiac cells MIBI (most commonly used at MGH) Produced on site, 6 hour half life, uptake passive and thus proportional to regional myocardial blood flow, no redistribution Tracer is excreted into the GI tract through biliary system, and appearance of tracer in GI tract can interfere with imaging of the inferior wall (this is why patients must be NPO) Produces sufficient count densities to allow ECG gating and thus allows for calculation of EF Defects characterized as fixed (scar), reversible (ischemic), partially reversible (mixture of scar and ischemia), artifact (breast, diaphragm), high-risk (defect in >1 distribution, increased lung thallium uptake, LV dilation) Pharmacologic testing Used in concert with nuclear imaging Dipyridamole (not commonly used at MGH) Indirect vasodilator, flow enhanced in normal vessels and much less so in stenosed vessels (which possess little flow reserve) Side effects: headache, nausea, chest pain, hypotension, dizziness, flushing Avoid in severe COPD, theophylline therapy, and critical carotid stenosis Adenosine (used at MGH) Vasoactive end product of dipyridamole and has much shorter half-life Side effects: chest pain, headache, nausea, flushing, dyspnea, AV block MGH Medical Housestaff Manual 21 3. Noninvasive testing Avoid in settings similar to dipyridamole Dobutamine (used at MGH) Simulates exercise by increasing heart rate Side effects: ectopy, headache, flushing, dyspnea, parasthesias, hypotension Stress echocardiography To screen for CAD and localize coronary lesions, with sens and spec similar to nuclear imaging Can use exercise or dobutamine. At MGH, exercise imaging is done with nuclear techniques, and stress echo is limited to dobutamine studies Advantages: provides EF, rule out valvulopathy, images not attenuated (useful in women) Limitations: severe mitral or aortic regurgitation and dilated cardiomyopathy may make interpretation difficult Dobutamine echo is especially helpful in evaluating pts with LV dysfunction and severe aortic stenosis to quantify the extent of AS Myocardial viability Key is to differentiate dysfunctional yet viable myocardium from necrosed and scarred myocardium in order to determine which patients may benefit from revascularization Stunned myocardium—viable, occurs after acute episode, can last days to weeks, may recover with or without revascularization Hibernating myocardium—viable, caused by repetitive ischemic injury or chronic reduction in blood flow, needs revascularization for recovery PET is the current standard for assessment, but is not typically used at MGH Rest-redistribution thallium imaging—redistribution of tracer can only occur in viable myocardium MIBI is less accurate in detecting viability Daniel Krauser, M.D. MGH Medical Housestaff Manual 22 4. Complications of myocardial infarction Pump failure Rales, S3, cardiogenic shock (occurs in 7% patients with MI) Treatment Na restriction Diuretics Digoxin for symptomatic relief Afterload reduction (ACE-I or hydralazine and nitrates) Hemodynamic stabilization and look for reversible causes Consider PA catheter, aortic balloon pump or ventricular assist device Mechanical complications Accounts for 15% mortality after MI Myocardial free wall rupture Accounts for 10% of post MI deaths Typically occurs in first 5 days of MI (can occur day 1-21) Occurs in small infarcts and commonly anterolateral MI Single vessel MI with poor collaterals Elderly females with first MI or few prior MIs Leads to death or PEA Associated with less pulmonary edema, more hypertension Not associated with murmur Increased risk after thrombolysis May lead to false aneurysm Ventricular septal defect Occurs in 1-3% of post MI patients, 1-10 days post MI Causes 5% peri-infarct deaths If anterior MI, VSD usually in apical septum If inferior MI, defect located on basal inferior septum (worse outcome/more difficult to repair) Associated with new pansystolic murmur and occasionally thrill Can detect shunt with step-up Usually seen in elderly females with few prior MIs and lack of collaterals Highest overall mortality, but not acutely fatal Class I indication for IABP MGH Medical Housestaff Manual Acute disruption of the mitral valve with severe mitral regurgitation Accounts for 5% post MI deaths; occurs 2-7 days post MI; rarest of mechanical complications Associated with inferior MI (due to posteriormedial papillary muscle supplied by PDA) and/or true posterior MI Rarely seen with ALMI Females > male; less likely to have STE; EF> in end LV function becomes hyperdynamic May hear thrill on exam Right ventricular infarct Common sequela of inferior MI Typical triad of hypotension, increased JVD, clear lungs Kussmaul’s sign from pseudoconstriction 1 mm STE in R sided leads, particularly V4R RV wall motion abnormal, RV dilatation Elevated right atrial pressure Increased incidence of high grade AV block Volume load and add vasopressors Infarct expansion Thinning and dilatation of infarct segment without pain or CK leak Infarct extension Recurrent pain and CK-MB leak Left ventricular aneurysm Occurs days to weeks post MI Begins with infarct expansion, necrosis, removal of debris, replace with scar Risk factors include large infarcts, uncontrolled hypertension, receiving steroids or NSAIDs Apical dyskinesis/aneurysm predisposes to thrombus Can compromise pump function and cause VT Left ventricular thrombus Observed in 10-40% of anterior wall infarcts Usually in LV apex Increased risk of embolization during first 3-6 months post MI Prevent with anticoagulation in patients with: Large anterior MI CHF 23 4. Complications of myocardial infarction Large apical aneurysm or dyskinetic segments Acute management of suspected mechanical complication Stat echo O2 sat run (oxygen sat step-up between RA and PA >5 indicates VSD) Cardiac catheterization PCI IABP Vasodilators (and may require pressors) Surgery Pericarditis Frequently occurs with transmural MI Pericardial rub, pleuritic CP, pericardial effusion Dressler’s syndrome: late pericardial inflammation (2 wks-3 months) Treat with salicylates, NSAIDs, colchicine Try to avoid steroids because of high relapse rate Conduction disturbances Result from ischemic injury to conduction system or surrounding myocardium and abnormal reflexes vagally-mediated Blood supply Sinus node: RCA 60% cases, LCx 40% cases A-V node: determines dominance, distal branches RCA 90% cases, distal portions LCx 10% cases His bundle: primarily AV nodal artery with LAD septals Right bundle branch from LAD septals, some collateral from RCA/circumflex Left bundle branch from LAD Left anterior fascicle from LAD septals, 50% have AV nodal collaterals Left posterior fascicle from proximal AV nodal artery, distally dual supply from LAD/PDA septal perforators Sinus bradycardia Occurs with anterior or inferior MI (up to 40%) Atropine if symptomatic First degree AV block More often in inferior than anterior MI due to AV nodal artery ischemia May be intranodal, intra, or infra-Hisian MGH Medical Housestaff Manual Usually transient and benign course Second degree AV block Usually develops within first 24 hrs of MI Mobitz type I Usually with inf/post MI Often responds to atropine Narrow QRS Observe unless symptoms or HR <45 Mobitz type II Uncommon in inferior, mostly anterior MI due to infranodal conduction system injury Wide QRS 1/3 progress to complete heart block Complete heart block 3-7% of patients with acute MI Inferior MI from intranodal lesion; more benign with narrow QRS escape Anterior MI carries high mortality rate (80%); wide unstable escape rhythm Recovery usually within 3-7 days Temporary transvenous pacemaker required MILIS trial. Prediction of complete heart block. 1 point assigned for each: 1. New development of PR prolongation 2. Second degree AV block 3. Left anterior or posterior fascicular block 4. LBBB 5. RBBB The risk of progression to CHB was: 0 points 1.2-6.8% 1 point 7.8-10% 2 points 25-30% 3 points 36% Bundle branch block of any type Identifies patients with extensive infarction LAHB 5% pts with benign prognosis Complete RBBB or LBBB 10-15%, usually RBBB Late post infarction bradyarrhythmias If complete heart block, high grade AVB, persistent second degree type II AVB may need permanent pacemaker 24 4. Complications of myocardial infarction New BBB and transient but resolved CHB during acute MI may need permanent pacemaker If monomorphic implies arrhythmogenic substrate Supraventricular arrhythmias Sinus tachycardia 25% of pts with acute MI Persistent sinus tachycardia marker of LV dysfunction, poor prognostic sign Goal HR < 70; treat with beta blocker Atrial premature beats May represent increased left atrial pressure If occurs in isolation, observe Recommendations for Temporary Transvenous Pacing - ACC/AHA Updated Guidelines (1999) Class I 1. 2. Atrial fibrillation 10-15% with acute MI Presence early signifies atrial ischemia Later may represent atrial stretch 3. 4. A-V nodal reentry Occurs infrequently May respond to adenosine or digitalis Ventricular tachyarrhythmias In STEMI, incidence of VT or VF 10%; about 80-85% occurred in first 48 hours In NSTEMI, overall incidence of VT or VF about 2.1%; median time to arrhythmia was 78 hrs, 25-75th percentile 16 hrs-7 days Accelerated idioventricular arrhythmia (50-110 bpm) Originally seen as reperfusion rhythm May occur in up to 40%; generally considered benign If it accelerates to 110-120, consider atropine to overdrive suppress If symptomatic, treat as if VT Ventricular tachycardia 15% pts during acute MI Rate usually 120-140 May degenerate to VF If non-sustained (<30 s with hemodynamic stability), does not necessarily need antiarrhythmic treatment Ventricular fibrillation 8% pts surviving hospitalization More frequent in large Q wave MI May occur without warning If it occurs late it is usually due to LV dysfunction and portends poor prognosis 5. Asystole. Symptomatic bradycardia (includes sinus bradycardia with hypotension and type I seconddegree AV block with hypotension not responsive to atropine). Bilateral BBB (alternating BBB or RBBB with alternating LAFB/LPFB) (any age). New or indeterminate age bifascicular block (RBBB with LAFB or LPFB, or LBBB) with firstdegree AV block. Mobitz type II second-degree AV block. Class IIa 1. 2. 3. 4. 5. RBBB and LAFB or LPFB (new or indeterminate). RBBB with first-degree AV block. LBBB, new or indeterminate. Incessant VT, for atrial or ventricular overdrive pacing. Recurrent sinus pauses (greater than 3 seconds) not responsive to atropine. Class IIb 1. 2. Bifascicular block of indeterminate age. New or age-indeterminate isolated RBBB. Class III 1. 2. 3. 4. First-degree heart block. Type I second-degree AV block with normal hemodynamics. Accelerated idioventricular rhythm. Bundle branch block or fascicular block known to exist before acute MI. Class I. Conditions for which there is evidence and/or general agreement that a given procedure or treatment is useful and effective. Class IIa. Weight of evidence/opinion is in favor of usefulness/efficacy. Class IIb. Usefulness/efficacy is less well established by evidence/opinion. Class III. Conditions for which there is evidence and/or general agreement that the procedure/treatment is not useful/effective and in some cases may be harmful. Roderick Tung, M.D. MGH Medical Housestaff Manual 25 5. Intra-aortic balloon pump General considerations Primary purpose is to decrease afterload/oxygen demand (deflation) and increase coronary blood flow/oxygen supply (inflation). Systole Diastole Non-augmented systolic pressure Augmented diastolic pressure Reduced systolic pressure Reduced aortic end-diastolic pressure Unassisted aortic end-diastolic pressure Dicrotic notch Indications Recommendations for IABP in acute MI (ACC/AHA Updated Guidelines (1999)). Class I Cardiogenic shock not quickly reversed with pharmacological therapy as a stabilizing measure for angiography and prompt revascularization. Acute mitral regurgitation or VSD complicating MI as a stabilizing therapy for angiography and repair/revascularization. Recurrent intractable ventricular arrhythmias with hemodynamic instability. Refractory post-MI angina as a bridge to angiography and revascularization. Class IIa Signs of hemodynamic instability, poor LV function, or persistent ischemia in patients with large areas of myocardium at risk. Class IIb In patients with successful PTCA after failed thrombolysis or those with three-vessel coronary disease to prevent reocclusion. In patients known to have large areas of myocardium at risk with or without active ischemia. Contraindications Aortic insufficiency. Aortic dissection or aneurysm. Peripheral vascular disease, severe aorto-iliac or femoral artery disease, bilateral fem-pop bypass. Uncontrolled sepsis. MGH Medical Housestaff Manual 26 5. Intra-aortic balloon pump Uncontrolled bleeding diathesis. Balloon position Inserted in cardiac cath lab, tip 2 cm from aortic knob. If balloon is too high (proximal to L subclavian), increased risk of CVA/limb ischemia. If balloon is too low, decreased renal perfusion, oliguric ARF. Balloon occludes 70-80% of aorta. Timing Key to efficacy Inflation starts at dicrotic notch (AV closure), deflation starts at end-diastole Early inflation Increased afterload Late inflation Submaximal augmentation Early deflation Submaximal augmentation Late deflation Increased afterload Complications Bleeding: thrombocytopenia/hemolysis from balloon itself, HIT Infection: cefazolin or vancomycin prophylaxis while IABP in place Embolic phenomena leading to visceral and limb ischemia; atheroemboli Vascular laceration/dissection: check distal pulses qd (can also use pulse volume recorder) Shift in balloon position: daily CXR to verify position Balloon rupture: rare Weaning Heparin is usually off about 4h prior to balloon pull. Cardiology fellow usually pulls IABP. IABP must be at 1:1 while off heparin to prevent clot formation Check EKG and hemodynamics (if PA line in place) with each change Fast wean (UAP, MI, and/or s/p PTCA and EF > 40%) 1:2 x 1h 1:4 x 1h 1:8 x 1h 1:1 x 4h IABP out Slow wean (cardiogenic shock) 1:2 x 4h 1:4 x 4h 1:8 x 4h 1:1 overnight In AM 1:2 x 1h 1:4 x 1h 1:8 x 1h 1:1 x 4h IABP out Roderick Tung, M.D. MGH Medical Housestaff Manual 27 6. PA catheterization Indications Diagnostic Shock (septic, cardiogenic, etc) Pulmonary edema (cardiogenic, increased permeability) Evaluation of ventricular function Pericardial tamponade Constrictive pericarditis Valvular lesions Pulmonary hypertension Assessment of oxygen transport Management When to send patient to cath lab for fluoroscopically-guided PA line placement Temporary pacemaker or recently (within 3 months) placed permanent pacemaker, ICD, or ASD/PFO/VSD closure device Inability to achieve PA or PCW position on previous attempt without fluoroscopy Placement from femoral vein Left bundle branch block Suspected or known PA systolic pressure >70 mm Hg Severe tricuspid valve disease ARDS Shock Heart failure Response to therapy (diuretics, vasodilators, pressors, inotropes) Perioperative monitoring Contraindications Right-sided endocarditis Prosthetic right heart valve (mechanical incurs more risk than bioprosthetic) Coagulopathy Thrombocytopenia Pacemaker Ventricular arrhythmias Severe pulmonary hypertension Left bundle branch block (there is a 3% incidence of inducing RBBB with PA catheters and thus, with underlying LBBB, can lead to complete heart block), this should be performed only with transcutaneous pacemakers applied or in cath lab. Theoretical considerations Pulmonary capillary wedge pressure (PCWP) roughly reflects the left ventricular preload: PCWP approximates left atrial (LA) pressure. LA pressure approximates left ventricular end diastolic pressure (LVEDP). LVEDP is proportional to left ventricular end diastolic volume (LVEDV). Valvular disease and changes in left ventricular compliance alter these underlying assumptions. Basic assumptions fail under certain circumstances: PA diastolic pressure > PCWP (e.g. pulmonary hypertension) PCWP > LA pressure (e.g. mediastinal fibrosis, veno-occlusive disease) LA pressure > LVEDP (e.g. mitral stenosis) Alteration of the normal LVEDV/LVEDP relationship (e.g. decreased LV compliance) The catheter tip not located in West lung zone 3 such that PCWP approximates alveolar pressure and not LA pressure. MGH Medical Housestaff Manual 28 6. PA catheterization Technical considerations The PA catheter used at MGH, the Edwards VIP, has five ports: the distal PA port (yellow bulb), the thermistor (connector on yellow bulb), the balloon (red bulb), the proximal injection used for thermodilution (blue bulb), and the proximal infusion (white bulb) Placed preferably in either the right internal jugular vein or the left subclavian vein. Rule of 10’s. Hemodynamic changes occur at approximately 10 cm intervals, with RA at 20 cm, RV at 30 cm, PA at 40 cm, and pulmonary capillary wedge pressure (PCWP) at 50 cm. The balloon is inflated as the catheter is advanced or when “wedging” the catheter. The balloon is deflated when withdrawing the catheter and at all other times. Obtain chest radiograph after placement to confirm proper position and daily to assess placement. Waveforms of normal hemodynamics RA waveform a A wave is due to atrial contraction; the peak follows the peak c v y of the electrical P wave by about 80 ms. x C wave is due to sudden motion of tricuspid valve ring towards RA at the onset of ventricular contraction; the C wave follows A wave by a time period similar to PR interval. V wave is due to atrial filling during ventricular systole when tricuspid valve is closed; peak of V wave occurs near end of T wave. X descent reflects atrial relaxation and the sudden downward movement of the atrioventricular junction. Y descent corresponds to rapid atrial emptying following opening of the tricuspid valve. During inspiration the mean right atrial (and wedge) pressure decreases due to decreased intrathoracic pressure. The A and V waves and X descent typically become more prominent. RV waveform. Systolic pressure 15-30 mm Hg. Diastolic pressure 0-4 mm Hg. PA waveform. Systolic pressure 15-30 mm Hg. Diastolic pressure 6-12 mm Hg. Mean PA pressure usually <20 mm Hg. PCWP waveform. This is similar to the RA waveform but dampened and delayed (the peak of the A wave follows the peak of the electrical P wave by about 240 msec and the peak of the V wave occurs after the electrical T wave). Measurements The intrathoracic pressure variation during the respiratory cycle is transmitted to the heart and vasculature. ALL MEASUREMENTS ARE TAKEN AT ENDEXPIRATION. Intrathoracic pressure closest to Cardiac index 2.7-4.3 Normal range 2.2-2.7 Subclinical depression 1.8-2.2 Onset of clinical hypoperfusion <1.8 Cardiogenic shock Cardiac index = cardiac output/BSA N Engl J Med 1976;295:1356 atmospheric pressure at end-expiration. End-expiration corresponds to the “high point” in the spontaneously breathing patient and the “low point” in patients on positive pressure ventilation. Note that PEEP >10 cm H2O may artificially raise measurements of intracardiac pressures MGH Medical Housestaff Manual 29 6. PA catheterization Cardiac output Thermodilution. A known volume of cold indicator fluid is injected into the proximal (RA) port and the temperature is measured by the thermistor located near the distal tip of the catheter (PA). Thermodilution curve is generated by plotting decline in pulmonary artery temperature versus time. Area under the curve is used to calculate cardiac output. TR produces significant distortions in this curve Fick method. oxygen consumption cardiac output arterial oxygen content - mixed venous content In conditions of distributive shock (e.g. sepsis), this method does not correlate well with cardiac output. Green dye. Cardiac output estimated by injecting a known amount of indocyanine green dye into a central line and then measuring its concentration in arterial samples taken over time. Valvular regurgitation and intracardiac shunts may make this method inaccurate. Page the balloon pump techs during regular hours to perform the study. Requires arterial line. Systemic vascular resistance Based on Ohm’s equation in electrical circuits (V = IR), vascular resistance can be estimated in hydraulic fluid mechanics (P = QR), multiplied by 80 to convert the units into dynes-sec/cm5: SVR (dynes sec cm 5 ) MAP (mmHg) CVP (mmHg ) 80 CO (L/min) Specific conditions and arrhythmias Acute mitral regurgitation Tricuspid regurgitation RV infarction Pulmonary embolism Pericardial tamponade VSD Atrial fibrillation Giant V waves on the wedge tracing (can be mistaken for PA waveform) Accentuation of RA V wave with a steep Y decent and elevation of the mean right atrial pressure. RA pressure > wedge pressure. RA waveform prominent X and Y descents (Y descent may exceed X descent) Kussmaul's sign (increase in RA pressure with inspiration) Narrow PA pulse pressure (from decreased RV stroke volume in severe RV infarction) Mean PA pressure rarely exceeds 40 mm Hg (mean PA pressures above this suggest chronic component) PA diastolic pressure > PCWP. A and V waves frequently disappear from the wedge tracing because transmission of LA pressure disrupted Elevation and equalization of the RA, PA diastolic and wedge pressures coupled with pulsus paradoxus. RA waveform, dominant X descent. Y descent is attenuated or absent. RA pressure often decreases with inspiration which may help to distinguish tamponade from other conditions resulting in equalization of pressures, such as RV infarction of constrictive pericarditis RA, mean PA, and wedge pressures are all elevated. In acute left-to-right shunt, see increase (7% or more) in O2 saturation from SVC to PA Atrial systole is lost and the A wave disappears MGH Medical Housestaff Manual 30 6. PA catheterization Atrial flutter Atrioventricular reentry tachycardia Ventricular tachycardia Mechanical flutter waves at approximately 300/minute Regular cannon A waves in RA tracing (atrial contraction when the atrioventicular valves are closed. Diastole shortened resulting in summation of A and V waves. Cannon A waves frequently encountered with ventricular arrhythmias (from AV dissociation). Complications From central venous cannulation. Pneumothorax or hemothorax (1-3%), arterial puncture, air embolism, or thrombosis. From advancement of catheter. Atrial or ventricular arrhythmias, RBBB, complete heart block (about 3% in patients with pre-existing LBBB), catheter knotting, cardiac perforation and tamponade, and pulmonary artery rupture. From maintenance of catheter. Infection (especially if left in place for greater than 3 days), thrombus, pulmonary infarction (<1.4%), balloon rupture, and pulmonary artery rupture. Normal PA line tracing Jonathan Passeri, M.D. Andrew Yee, M.D. MGH Medical Housestaff Manual 31 7. ICU drips Generic Trade Mechanism Usage Dosing Side effects Vasopressors/Inotropes Norepinephrine Levophed Hypotension, especially 1, 1 agonist, causes vasoconstriction and increases “cold” septic shock. cardiac output. 0-100 mcg/min. Peripheral or renal hypoperfusion, also potentially arrhythmogenic. Phenylephrine NeoSynephrine -1 agonist, raises BP by causing peripheral vasoconstriction. Hypotension with high CO, low SVR (“warm” or early sepsis, neurogenic shock). 10-1000 mcg/min. Peripheral or renal hypoperfusion due to vasoconstriction. Dopamine Intropin Low dose (1-3 mcg/kg/min) acts on D1 receptors, causes visceral vasodilation (“renal dose”). Medium dose (3-10 mcg/kg/min), adds 1 agonism, increasing CO. High dose (>10 mcg/kg/min), adds 1 agonism, causing vasoconstriction. At “renal doses” augments 10-1000 mcg/min. diuresis but has no clear renal-protective benefit. At higher doses, treats hypotension due to sepsis or cardiac failure. Prominent natriuretic effect, tachycardia and arrhythmias at higher doses. Vasopressin Pitressin Renal ADH effects as well as generalized vasoconstriction. Refractory septic shock, also used in VF/pulseless VT arrests. Peripheral or visceral hypoperfusion due to vasoconstriction (though not observed at lower doses used for hypotension) Dobutamine Dobutrex 1, 2 agonism increases inotropy/chronotropy with peripheral vasodilation. Increasing CO in 2-40 mcg/kg/min, Hypotension due to vasodilation, arrhythmias. cardiogenic shock, not to titrate to desired be used as a pressor due to hemodynamic profile. vasodilation. Amrinone Inocor Inhibits phosphodiesterase-III, Similar to dobutamine, but increasing CO and peripheral with less arrhythmogenic vasodilation. potential. Milrinone Primacor Similar to amrinone. Epinephrine Adrenalin Hypotension due to vasodilation, arrhythmias. Similar to amrinone. 0.375-0.75 mcg/kg/min Similar to amrinone. Generally used in ACLS, anaphylaxis. 1-10 mcg/min Variable BP effects limit its utility Bradycardia. 2-10 mcg/min. High 2 effect limits pressor utility 1 g iv load is 150 mg Hypotension, multiple bolus, 1 mg/min for 6 systemic side effects with long-term use. hrs, 0.5 mg/min for 18 hrs. Use 150 mg boluses in ACLS. Similar to epinephrine, used for post-anesthesiainduced hypotension. Ephedrine Isoproterenol 0.04-0.067 U/min (in refractory VF, 40 U bolus) (Circulation 2003;107:2313) Isuprel Anti-arrhythmics and anti-hypertensives Amiodarone Cordarone Class III anti-arrhythmic, blocks K+ channels, delaying repolarization. Treatment and prevention of atrial and ventricular tachyarrhythmias. Esmolol Brevibloc -blocker, primarily of 1 receptor. Extremely short Useful as a trial drug, to see if a patient will MGH Medical Housestaff Manual As with other -blockers 32 7. ICU drips half-life (9 minutes). tolerate -blockade (e.g., in RV infarct). Propranolol Inderal Non selective -blocker. Useful for heart rate control, especially in aortic dissection (used with nitroprusside). 1-40 mg/min. Similar to other blockers, but also causes prominent change in mental status. Fenoldopam Corlopam Selective postsynaptic dopamine agonist at D1 receptors. Rapidly lowers BP, as in hypertensive urgency/emergency, but theoretically maintains renal perfusion (not well studied yet). 1-150 mcg/min. May decrease SVR, making it difficult to use in some patients. Labetalol Normodyne Combined and -blocker ( Rapid lowering of BP, as blocking effect 7x greater than in hypertensive urgency/emergency. effect when iv). 1-6 mg/min. Bradycardia, bronchospasm, hypotension. Nitroglycerin Direct nitric oxide-mediated vasodilation, venous > arterial. Hypertension, angina in 1-1000 mcg/min. acute coronary syndromes. Headache, tachycardia, hypotension, tachyphylaxis. Nitroprusside Nipride Direct nitric oxide-mediated arterial and venodilation. Rapid lowering of BP, first-line in hypertensive emergency. 10-800 mcg/min. Hypotension, tachyphylaxis, rare cyanide toxicity. Caution in renal failure as cyanide metabolites are renallycleared. Lidocaine Xylocaine Class IB anti-arrhythmic Used in sustained VT and VF 1-1.5 mg/kg bolus; infuse 1-4 mg/min Mental status change. Procainamide Pronestyl Class IA anti-arrhythmic Used in VT, atrial fibrillation Load 20 mg/min until Hypotension with iv 17 mg/kg reached, infusion. Drug-induced lupus, agranulocytosis. side effects, or arrhythmia controlled; infuse 1-4 mg/min. 8-20 mg bolus, 1-10 mg/hr. Sedatives/Paralytics Cisatracurium Nimbex Neuromuscular blocking agent, blocks ACh-mediated transmission at NMJ. Along with others of its class, paralysis of patients to assist in intubation/ventilation. Fentanyl Sublimaze Binding of opiate receptors. Sedation and analgesia in 50-300 mcg/hr. hemodynamically unstable or morphine sensitive patients. Propofol Diprivan Phenolic derivative with IV infusion for ICU general anaesthetic properties. sedation. 25-300 mg/hr. Hypotension, neuroexcitatory effects, bradycardia. Midazolam Versed Benzodiazepene. 1-7 mg/hr. Cumulative effects, respiratory depression, tolerance. IV infusion for ICU sedation, preferred over other benzos because of short half life, lack of active metabolites. Prolonged paralysis, myopathy, blockade of autonomic activity. Respiratory depression, dependence, tolerance, constipation. Ednan Bajwa, M.D MGH Medical Housestaff Manual 33 8. Anticoagulation for atrial fibrillation, prosthetic valves Atrial fibrillation Recommendations are from Sixth American College of Chest Physicians Consensus Conference on Antithrombotic Therapy in Chest 2001;119:1S. One or more high risk factors (see below), treat with warfarin (target INR 2.5, range 2.0-3.0) Two or more moderate risk factors, treat with warfarin (target INR 2.5, range 2.0-3.0) One moderate risk factor, treat with warfarin or aspirin 325 mg qd Low risk patients, treat with aspirin 325 mg qd only High risk factors Age >75 Previous TIA or stroke Hypertension Poor LV systolic function Rheumatic mitral valve disease Prosthetic heart valve Moderate risk factors Age 65-75 Diabetes CAD with preserved LV function Low risk Patients less than 65 with no clinical or echocardiographic evidence of cardiovascular disease Annual stroke rate Placebo Warfarin Risk categories Age <65 no risk factors 1.0% 1.0% 4.9% 1.7% 1 risk factors Age 65-75 No risk factors 4.3% 1.1% 5.7% 1.7% 1 risk factors Age >75 No risk factors 3.5% 1.7% 8.1% 1.2% 1 risk factors Risk factors in this study are: hypertension, diabetes, prior stroke or TIA Arch Intern Med 1994;154:1449 Valvular heart disease Rheumatic mitral disease with left atrial diameter >5.5 cm or history of systemic embolism, treat with warfarin long-term, target INR 2.5 (2.0-3.0) Mitral regurgitation or annular calcification, with systemic embolism, treat with warfarin long-term, target INR 2.5 (2.0-3.0) Prosthetic heart valves by target INR Sinus rhythm, normal left atrium size with an aortic St. Jude Medical bileaflet, Carbomedics bileaflet, or Medtronic-Hall tilting disk 2.5 (2.0-3.0) Mitral tilting disk and bileaflet valves 3.0 (2.5-3.5) or 2.5 (2.0-3.0) and aspirin 81 mg qd Bileaflet mechanical aortic valves with AF 3.0 (2.5-3.5) or 2.5 (2.0-3.0) and aspirin 81 mg qd Caged ball or caged disk in any position 3.0 (2.5-3.5) and aspirin 81 mg qd Mechanical valve plus other risk factors or systemic embolism 3.0 (2.5-3.5) and aspirin 81 mg qd Bioprosthetic (e.g. Carpentier-Edwards) mitral or aortic valve 2.5 (2.0-3.0) for 3 months after insertion, then aspirin 81 mg qd Ethan Korngold, M.D. MGH Medical Housestaff Manual 34 9. Wide complex tachycardia v. SVT General considerations Wide complex tachycardia is ventricular tachycardia until proven otherwise. Patients with prior MI or low EF, pre-test probability that WCT will be VT >98%. Consider artifact (patient motion can simulate WCT). When in doubt, treat WCT like VT. Avoid verapamil. Differentiation based on atrial activity AV dissociation, atrial activity independent of ventricular activity. P P P Diagnostic aids Presenting symptom History CAD and previous MI First arrhythmia after MI Physical exam AV dissociation Blood pressure Heart rate EKG Rate QRS >0.16 s QRS <0.16 s Significant axis shift Marked left axis Right or normal axis QRS morphology Positive QRS concordance Negative QRS concordance AV dissociation No AV dissociation Usefulness Unhelpful VT VT VT Unhelpful Unhelpful Unhelpful VT Unhelpful VT Suggests VT Unhelpful Usually unhelpful Usually VT (may be antidromic AVRT) Unhelpful VT Unhelpful Fusion beat, simultaneous activation of the ventricular myocardium and ventricular focus. Dressler beat, QRS complex identical to the sinus QRS complex; from normal impulse conduction. Fusion Dressler beat beat P wave Morphology Determine if RBBB-type or LBBB-type. In V1 positive or RBBB-type WCT, morphologies suggestive of VT in lead V1 See Wellens, Am J Med 1978;64:27, Brugada, Circulation 1991;83:1649 (for sens and spec for VT). V1 RBBB-type R sens 0.60, spec 0.84 MGH Medical Housestaff Manual PQR or RS sens 0.30, spec. 0.98 “Rabbit ear,” left > right, triphasic sens 0.82, spec 0.91 35 9. Wide complex tachycardia v. SVT RBBB-type WCT, morphologies suggestive of VT in lead V6 See Wellens Am J Med 1978;64:27; see also Brugada, Circulation 1991;83:1649 (for sens and spec for VT). V6 RBBB-type R:S <1 QS (or QR) Monophasic R Triphasic R:S >1 sens 0.41, spec 0.94 sens 0.29, spec 1.0 sens 0.01, spec 1.0 sens 0.64, spec 0.95 sens 0.30, spec 0.76 Kindwall LBBB-type WCT, morphologies suggestive of VT Am J Cardiol 1988;61:1279; see also Brugada, Circulation 1991;83:1649 (for sens and spec for VT). LBBB-type 1. R in V1 or V2 >30 ms 2. Notched or slurred downstroke S wave V6 V1 or V2 4. Any Q in V6 Sens 0.17, spec 1.0 3. >60 ms from QRS onset to S nadir Above V1 or V2 criteria, sens 1.0, spec 0.89 MGH Medical Housestaff Manual 36 9. Wide complex tachycardia v. SVT Brugada algorithm See Circulation 1991;83:1649. Based on prospective analysis of ventricular tachycardia v. SVT with aberrant conduction by EP study Stepwise approach Wide complex tachycardia 1. Absence of an RS complex in all precordial leads Yes No VT sens 0.21, spec 1.0 2. R to S interval >100 ms in one precordial lead Yes No VT sens 0.66, spec 0.98 3. Atrio-ventricular dissociation Yes No VT sens 0.82, spec 0.98 4. Morphology criteria for VT present in both precordial leads V1, V2, and V6? (see above) Yes No VT sens 0.987, spec 0.965 SVT sens 0.965, spec 0.987 Gregory Bashian, M.D. Jonathan Passeri, M.D. Andrew Yee, M.D. MGH Medical Housestaff Manual 37 10. MGH triage guidelines for acute coronary syndromes Ell 9 - CCU / Bl 7 - MICU Floor Patient/Clinical Characteristics for Admission a. b. c. d. e. f. g. h. Persistent ST (>20 minutes) Refractory HTN Severe CHF Cardiogenic shock IABP Invasive hemodynamic monitoring Ventilatory support VF or sustained VT with hemodynamic compromise Interventions/Meds Supported on Unit/Floor Triggers for moving to more acute unit a. IABP b. Sustained high dose IV pressors and inotropes c. Ventilatory support d. IIb/IIIa inhibitors NA Ell 10 - SDU A positive troponin in a hemodynamically stable patient is not necessarily a direct indication for admission to Ellison 10. a. Persistent ST or deep (>3 mm) T wave inversions (>20 minutes) b. Uncomplicated MI post-angioplasty c. Stable acute MI patients post percutaneous coronary intervention with or without thrombolysis d. Unstable angina if hemodynamically stable e. Typical angina >20 minutes or requiring morphine f. SVT g. Unstable bradyarrhythmias h. Other malignant arrhythmias (NSVT or VT without hemodynamic compromise) i. Syncope j. Moderate heart failure k. Comorbidities that compromise cardiac status, e.g., GI bleed Symptomatic bronchospasm (COPD or asthma) Infection TIA/CVA Renal Failure k. Transient hypotension MGH Medical Housestaff Manual a. IV TNG b. Temporary and external pacemakers c. IV antiarrhythmics (e.g. lidocaine, procainamide, bretylium, and amiodarone) d. Low dose dopamine, dobutamine (200600 mcg/min), or milrinone once a stable dose is established in the ICU e. IIb/IIIa inhibitors f. Epoprostenol (Flolan) a. IV meds requiring invasive monitoring e.g., nitroprusside, high dose dopamine and dobutamine, and initial titration of milrinone b. Refractory ischemia despite IV TNG, heparin, beta blocker, and ASA c. Other needs for invasive monitoring 38 10. MGH triage guidelines for acute coronary syndromes Ell 8 - Cardiac Surgery (criteria for medical patients) Ell 11 - Access Unit (cardiac attending) Floor Patient/Clinical Characteristics for Admission a. Priority for beds goes to referral patients b. Ischemic or congestive syndromes in need of a catheterization and/or coronary intervention. c. Admit to observe patients (post-cath patients or RPPR patients) d. Stable acute MI patients post percutaneous coronary intervention with or without thrombolysis e. Exclusions: Life threatening arrhythmia Bradycardia or heart block requiring temporary or external pacemaker SVT in need of cardioversion Acute renal failure possibly requiring dialysis Hemodynamic instability Anticipated long lengths of stay due to comorbidities a. Post-cath patients from Cardiac Access Unit (Team 4) b. Pre-cath referral patients (Team 4) c. Post-AICD (EP Fellow/Team 4 back-up) d. Post-cath pre-op cardiac surgery (Team 4) MGH Medical Housestaff Manual Interventions/Meds Supported on Unit/Floor Triggers for moving to more acute unit a. IV TNG b. IIb/IIIa inhibitors a. Life-threatening arrhythmia b. Bradycardia or heart block requiring temporary or external pacemaker c. SVT in need of cardioversion d. Hemodynamic instability e. Refractory ischemia despite IV TNG, heparin, beta blocker, and ASA f. Respiratory compromise requiring ventilatory support and/or frequent blood gas monitoring a. IV TNG may be started, but this generally triggers move for medical patients on Ellison 8 b. Temporary or external pacemaker 39 10. MGH triage guidelines for acute coronary syndromes Patient/Clinical Characteristics for Admission Interventions/Meds Supported on Unit/Floor Triggers for moving to more acute unit a. Typical angina with or without ischemic ECG with resolution in <20 minutes b. Positive CK-MB/troponin and hemodynamically and electrically stable after discussion with attending MD. a. Defibrillation a. Continuous IV TNG b. Recurrent or refractory ischemia c. Hemodynamically unstable d. Requiring intubation e. Positive CKMB/troponin in patient who is hemodynamically unstable (stable patients may stay on the unit at the discretion of the medical team and nursing staff.) f. Life-threatening arrhythmia g. Bradycardia or heart block requiring temporary or external pacemaker g. SVT in need of cardioversion a. Short-stay patients at low or moderate risk for MI b. Normal ECGs or abnormal ECGs with no new ischemic changes c. Chest pain resolved prior to or in ED d. Hemodynamically and electrically stable e. History of CAD accepted f. Exclusions: LBBB Paced rhythms Significant arrhythmia Suspected UAP requiring heparin or IV nitrates Comorbidities requiring >24 hours hospitalization a. IV TNG Ell 11 – Four CPOU beds (cardiac attending) General Medical Floor Floor (Patients will be supported on Ellison 11 as Cardiac Access patients if their clinical course evolves.) (Same as above for Ellison 11 Cardiac Access Unit) Triage nursing Cardiology MGH Medical Housestaff Manual 40