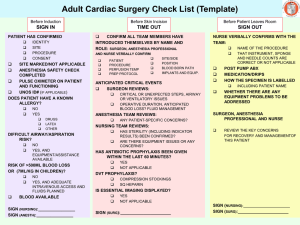
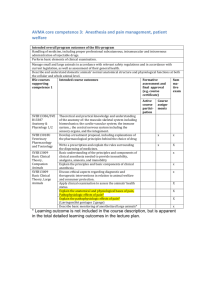
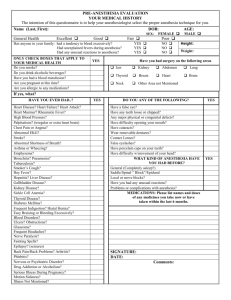
Table 2 Bibliography of the Articles Utilizing the Concept of Accuracy
advertisement

Table 2 Bibliography of the Articles Utilizing the Concept of Accuracy Author/Year Design/Purpose Sample Method Theory 12,287 electronic anesthesia records from a 17 OR* hospital in Jerusalem were collected over a one year time period. The AIMS built-in query function transferred endotracheal tube size, laryngoscope blade size, and intravenous access device size/location from all anesthesia records into an Access database. Data concordance is the proxy for accuracy, defined as the use of age appropriate equipment (i.e, appropriate size). Anesthesia specific literature Avidan & Weisman (2012) This study was a retrospective chart review to determine data concordance between specific documentation data and the patient's age. Data completeness is the presence of mandatory data fields. Benson et al. (2001) This study was a retrospective chart review for comparison of manually entered and automatically entered vital signs in terms of variability of data and improbable results. 22,531 electronic anesthesia records from a hospital in Germany were collected over a two year time period. Only the physiological data (vital signs) were analyzed. The AIMS** database was queried using SQL. Variability was calculated for automatically documented vital signs and for manually entered vital signs. Data validity is defined as a plausible result (i.e., physically possible and highly likely). Data variability was calculated using estimation of variance statistics. Table 2- (Continued) Author/Year Design/Purpose Sample Method Theory Anesthesia specific literature (continued) Bloomfield & Feinglass (2008) This is a review article [editorial] providing an overview of the reasons associated with the slow adoption of AIMS** and the benefits of implementing them. The literature review focused on potential uses of AIMS with a focus on advantages, disadvantages, patient safety, and error reduction. A very basic literature review presenting some research studies, but no critiquing. This article presents information that supports the significance of accuracy in documentation. Quality in AIMS documentations defined in terms of preciseness; clear & concise documentation; and accuracy. Devitt et al. (1999) This study was a onegroup, prospective, single-blinded, observational design that was used for the purpose of determining if the level of training and years of anesthesia experience have an effect on the accuracy of anesthesia documentation. A convenience sample of 124 total participants that included medical students, anesthesia residents, and anesthesiologists from an academic institution were included in the study. No time frame of data collection was given. Participants were observed during a simulated anesthesia case in a controlled laboratory environment. They managed the care of a simulated patient that experience 3 adverse events each. The generated anesthesia record was then compared to the electronic simulator for comparison of key vital signs. Completeness is defined as having data present for every physiological variable for each adverse event. Discrepancy, the proxy for accuracy in physiological variables, is the difference between observed and recorded values divided by the actual value. [observed – actual]/actual Table 2- (Continued) Author/Year Design/Purpose Sample Method Theory Driscoll, Columbia, This study was a & Peterfreund (2007) retrospective chart review to determine sources of incomplete data in electronic anesthesia records. All electronic anesthesia records for a one month period at a large academic hospital in the United States (N = 2838) were included in the analysis. The anesthesia records were reviewed for the presence of patient allergies, intravenous access type, electrocardiogram rhythm, ease of mask ventilation, grade of laryngoscopic view, and depth of endotracheal tube placement. Completeness is defined as the presence of predetermined mandatory data fields (six in this study). Edsall et al. (1993) 3 nurse anesthetists (volunteer convenience sample) were observed for a total of 10 anesthesia cases that were performed consecutively. Two reviewers (identified as authors of the manuscript) reviewed the anesthesia records and videotape. The video was assessed for the amount of time the nurse anesthetist spent on documentation generation. The documentation was reviewed for completeness with a 46-item questionnaire. Accuracy of documentation was reflected by quantity of data collected. Accuracy is defined in terms of the quantity of physiological data (vital signs) that can be recorded. Anesthesia specific literature (continued) This study was a prospective observational design using video recording of simulated anesthesia cases for the purpose of comparing manual anesthesia documentation with an automated anesthesia information management system. Completeness is determined by the presence of predefined mandatory data fields Table 2- (Continued) Author/Year Design/Purpose Sample Method Theory Anesthesia specific literature (continued) Thrush (1992) A prospective, blinded, descriptive study of one group was conducted for the purpose of determining the difference in accuracy between paper-based and electronic anesthesia documentation. A convenience sample of 13 anesthesiologists documented using a paper based anesthesia record and an electronic record system during the course of one anesthetic each. The paper based anesthesia record and the electronic anesthesia record both had the frequencies of deviations from pre-defined values (normal physiological ranges) in systolic blood pressure, diastolic blood pressure, heart rate, end-tidal carbon dioxide, and specific hemoglobin calculated. The corresponding electronic & manual records were compared using Fisher's exact test. Accuracy differences between manual & electronic anesthesia records are quantified by computing the frequencies of deviations from predefined limits. This overcomes the obstacle of trying to compare two graphical displays of vital signs that use different time intervals. Wilbanks, Moss, & Berner (2012) A prospective, descriptive observational study conducted for the purpose of identifying sources of inaccuracy in incompleteness in a newly implemented information management system. A convenience sample of 20 anesthesia cases involving nurse anesthetists. Nurse anesthetists were observed during the entire intraoperative period of anesthesia care. Study participants were given a questionnaire to assess perceived accuracy and satisfaction with the anesthesia information management system. Accuracy is defined in terms of correctness of data; using percentagreement statistics as a proxy. Completeness is determined using a pre-defined list of required charting elements. Table 2- (Continued) Author/Year Design/Purpose Sample Method Theory All paper-based anesthesia records (N = 890) from a three year time period were reviewed at a hospital in Thailand. A 44-item checklist was used by anesthesiologists scoring each category on a scale using the groupings “good”, “fair”, “poor”, or “no data”. Each anesthesia record (N = 890) was manually reviewed using this checklist. Data analysis was performed by calculating frequencies and using Chi-square between the complete and incomplete groups. Completeness is determined by the presence of predetermined mandatory data elements. The authors evaluated accuracy in terms of the degree of completeness. Anesthesia specific literature (continued) Yunuswangsa & Nimmaanrat (2008) A retrospective chart review was conducted to quantify anesthesia record completeness. Table 2- (Continued) Author/Year Design/Purpose Sample This study is a one group descriptive study for the purpose of testing the performance of a specific logistic regression model on evaluating the accuracy of machines that determine genomic mapping. (The disciplines of electrical engineering and genetic bioinformatics are represented in this study.) A training data set was used to test the model (a known genomic map). Method Theory Non-nursing literature Ruffalo et al. (2012) A known data set was read by genomic mapper and the logistic regression model was used to measure the accuracy of the mapper. Since a known genetic set was used the results of the genomic mapping could be compared to the already known values. Accuracy is defined as a data set that very closely represents actual values. The determination of accuracy requires comparison of recorded data with previously known values. Precision is the primary aspect of accuracy and is defined as a measured data set that correctly represents "actual" values. Table 2- (Continued) Author/Year Design/Purpose Sample Method Theory The database being evaluated in the study was created using a well-established software program used for mathematical computations in optical science. The generated database were created using linear algebra and advanced calculus. The databases represent the scattering patterns of wave particles when they hit spheroid particles. The practical applications involve remote sensing in the atmosphere of earth (i.e., studying weather and climate changes). Accuracy is the degree of correctly detecting (measuring) phenomenon. The gold standard for determining the accuracy of a measuring device is by measuring an already known phenomenon and comparing the outcome to the already known value. In this article the "known" values are represented by a database. Non-nursing literature (continued) Schmidt et al. (2009) This article is a case study for the purposes of presenting a benchmark database for calculating electromagnetic and light scattering properties of spheroidal particles (i.e., dust size matter). This would allow determination of accuracy in measurements. (This article represents the discipline of meteorology.) Table 2- (Continued) Author/Year Design/Purpose Sample Method Theory A reviewer (who was not clearly identified) evaluated each death certificate for all legally required data elements and then evaluated accuracy by comparing the data in the death certificate to available medical records. Accuracy is correct data fields that meet pre-defined requirements; determined by data concordance with other records containing the same types of data. Having correct data is not enough, the data must reflect meeting legal requirements. Non-nursing literature (continued) Sellinger, Ellis, & Harrington (2007) This study is a crosssectional, retrospective chart review for the purpose of determining the accuracy of death certificates and verifying all legally required elements are present. (This article represents the disciplines of criminal justice and medicine). Every available death certificate (N = 140) from a single hospital for a 4-month period, starting in February 2004, was evaluated. Completeness is determined by the presence of mandatory data fields. Table 2- (Continued) Author/Year Design/Purpose Sample Method Theory Callen, Alderton, & McIntosh (2008) This study is a retrospective chart review to determine accuracy in discharge summaries by comparing discharge medications with the inpatient medication record. 245 inpatient discharge summaries (62% electronic and 38% handwritten) were collected over a three month period in 2005 at an Australian hospital. Discharge summaries where analyzed in regards to discharge medications by compiling a list of all medications the patient was prescribed and then directly verifying the medications with the patient's inpatient medication administration record. Discrepancy is a difference between observed and recorded data (proxy for accuracy and completeness). Callen & McIntosh (2010) This study was a retrospective chart review to determine accuracy in patient medication lists created by health care providers. Discharge medication lists (966 handwritten and 842 electronically generated) where collected over a twoyear period in an Australian 78-bed hospital. Patient's self-reported medication lists (documented by nurses) within the patient charts where analyzed for drug details (drug name, dosage, and schedule of administration) and then confirmed directly with the patient for accuracy. Correctness is the presence of data that reflects actual events (confirmed by going directly to the source of the data). Nursing literature Completeness is a component of correctness and is represented by the frequency of missing events. Table 2- (Continued) Author/Year Design/Purpose Sample Method Theory 413 inpatient records from the same year (2002) where included in the study. The setting was a Swedish hospital that had surgical, orthopedic, and medical patients. Two data collections instruments (a pressure ulcer data collection tool and comprehensiveness in nursing documentation tool) were used to collect data from patient records. Two auditors (undergraduate nursing students) scored each record and then interrater reliability was calculated. The same three nursing students took turns performing the audit. The results were compared with physical exams by researchers that were performed on the same patients from a previous study. Completeness is defined with a 5-point Likert scale that measures comprehensiveness of nursing documentation. Nursing literature (continued) Gunningberg & Ehrenberg (2004) This study was a retrospective chart review to determine the accuracy of nursing documentation of pressure ulcers. Table 2- (Continued) Author/Year Design/Purpose Sample Method Theory 10 hospitals in the Netherlands (out of a total of 94) were randomly selected to participate. A convenience sample of 341 patient records was obtained from these hospitals. Data collection occurred between 2007 and 2008. Two reviewers at a time, who each had 20-hours of training on the use of the D-catch instrument, evaluated each patient record. This resulted in total scores for accuracy (evaluating the categories of record structure, admission data, nursing diagnosis, nursing interventions, outcome evaluations, and legibility). Inter-rater reliability was calculated to determine agreement in evaluation. Exploratory factor analysis with principal components analysis was conducted to evaluate the results. Accuracy is evaluated using quantitative and qualitative components. The quantity of data is an aspect of accuracy (i.e., if more data was collected then the record was more accurate). Nursing literature (continued) Paans et al. (2010) This study was a cross-sectional retrospective chart review for the purpose of describing the accuracy in nursing documentation at a specific hospital. Completeness is combined and measured with accuracy; and is represented partially by the quantity of data. Table 2- (Continued) Author/Year Design/Purpose Sample Method Theory 38 articles published between 2001 and 2011 were identified for inclusion in the review. The keywords 'record keeping', 'documentation', 'documenting', 'records', 'paperwork', and 'care plans' were used to identify articles in Ovid, CINAHL, Internurse, Eduserve, Intute, and Google Scholar. A summary of key concepts in the literature was presented. Accuracy is viewed in terms of truthfully representing patient care by providing a comprehensive presentation of the patient's experiences using clear terminology. Nursing literature (continued) Prideau (2011) This manuscript presented a basic literature review on nursing documentation for the purpose of defining quality documentation and factors associated with decreasing the quality of documentation. Completeness is defined as comprehensive documentation that covers all aspects of the nursing process. Table 2- (Continued) Author/Year Design/Purpose Sample Method Theory Two hospitals (one using electronic documentation and one using paper-based documentation) were used to collect data over four consecutive days. A total of 1312 records were included for analysis. Two reviewers (trained coders) analyzed the chart for completeness of patient history, physical examination, and documentation that guides medical decision making (laboratory values, radiology reports, vital signs, etc.). Interrater reliability was calculated to determine agreement on the evaluation. Completeness and discrepancy rates were then calculated. Completeness is defined as the presence of predetermined mandatory data elements. Nursing literature (continued) Silfen (2006) This study was a cross-sectional, retrospective chart review for the purpose of comparing the accuracy of procedural coding between a paperbased and electronic documentation system. Discrepancy is the difference in coding between what was actually coded and what an "expert" determines should have been coded. Accuracy is defined as concordance between discrepancy rates between different types of documentation. Table 2- (Continued) Author/Year Design/Purpose Sample Method Theory Nursing literature (continued) Stengal et al. (2004) Tse & You (2011) This study was a randomized controlled trial conducted for the purpose of determining if use of a portable documentation system could improve documentation quality. In 2001, 80 orthopedic surgical patients were consecutively randomized to either electronic or paperbased documentation. This pilot study was a retrospective chart review conducted for the purpose of determining the degree of accuracy in electronic documentation at a primary care physician’s office. 33 patients ranging from infants to elderly individuals, exact ages not stated, were recruited at an Australian general physician practice setting. Two "expert" evaluators (orthopedic physicians) reviewed patient records and determined if all possible diagnosis were documented with no redundancy; based on physical assessments and laboratory values in the record. Completeness rates were determined based on the physician’s evaluations of the possible and actual diagnosis present. Completeness is the inclusion of all possible diagnosis based on documented patient assessment. The researcher confirmed demographics, health history, and current medications in the patient's record directly with the patient. Frequencies of discrepancies were recorded. Accuracy is defined in terms of data reflecting reality and is determined by direct confirmation with the patient. Quality in documentation is represented by completeness of data without redundancy. Completeness is not a separate concept but is incorporated into the determination of accuracy. Table 2- (Continued) Author/Year Design/Purpose Sample Method Theory 77 articles published from 2000 to 2010 were identified in peerreviewed publications written in English. The keywords 'quality', 'evaluation', 'audit', 'nursing records', and 'nursing documentation' were used to search CINAHL, the Cochrane Library, Health Reference Center, ProQuest, InterScience, Medline, and Nursing Resource Centre databases. Articles dealing with auditing nursing documentation were included in the final analysis. All data was abstracted into Endnote for processing and synthesis. Completeness is evaluated using audit instruments specific to the type of documentation being evaluated (mandatory data fields). Nursing literature (continued) Wang, Hailey, & Yu (2011) This manuscript is a mixed-method systematic literature review for the purposes of summarizing how the quality of nursing documentation is evaluated (i.e., audited). Accuracy is defined as concordance between patient assessment and documentation. Determination of accuracy requires going directly to the source of the data for confirmation. Note. This is a summary of all of the articles included in the integrative literature review. None of the studies identified a specific theoretical framework. The theoretical and conceptual definitions of the concepts related to accuracy are included. *operating room (OR) **anesthesia information management system (AIMS)