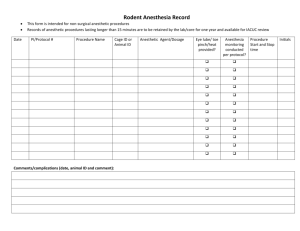
Revised May 2009 Official IACUC Office use only IACUC Number ________________ UNIVERSITY OF PENNSYLVANIA IACUC PROTOCOL REVIEW FORM-Form A-2006 This form is for all new protocols and 3 year de novo reviews. All protocols must be typed. Submit the original signed protocol, and one electronic copy sent to IACUC@pobox.upenn.edu to the Director of Animal Welfare, 3624 Market St., 301 S, Ph.: 215.898.2615. Protocols must be approved by the IACUC prior to the initiation of any investigation or teaching activity involving vertebrate animals. The IACUC Office will forward the protocol to all relevant secondary reviewers (e.g. EHRS, ULAR, etc.). Protocol Title: Grant title (if different from protocol title): a. Estimated dates of protocol: From To b. Funding Source: this protocol). , Grant #: (Not to exceed 3 years) (Please submit a copy of the grant minus appendices with c. Institution # (from Office of Research Services-(ORS)) Required for all sponsored research proposals: (Contact your BA or call ORS at 215-898-7293 to find out your number) d. Is this protocol part of a funded or non-funded RO1, training, center, or program grant? Yes . If Yes, Title: P.I.: , No Answer only those that apply: (check off answer with an “X”) a. b. Is this a new protocol submission? Yes Is this a 3-year resubmission? Yes If yes, Protocol # Principal Investigator (PI): Dept.: IACUC Policy on who may be a Principal Investigator on a Protocol. Responsible Organization Number (Required-Four digit #): Campus Address: Lab Location: , email: Co-Investigator: Campus Address: Lab Location: , email: Intramural Mail code: Lab. Phone w/area code: Office Phone w/area code: Emergency Phone # (not work) w/area code: Dept.: Intramural Mail code: Lab. Phone w/area code: Office Phone w/area code: Emergency Phone # (not work) w/area code: 1 Revised May 2009 Day to Day Contact Person (this person should be routinely available by telephone): Dept.: Phone w/area code: Emergency Phone # (not work) w/area code: email: If the principal investigator is a department chair, the Dean's signature must be obtained. If the study involves two departments, both Chairs’ signatures are required. Dept. Chairperson (Type Name) Dept. Dept. Chairperson (Signature)______________________________________ Date ___________ Dept. Chairperson (Type Name) Dept. Dept. Chairperson (Signature)______________________________________ Date ___________ Dean (Print Name) School Dean (Signature)_________________________________________________ Date ___________ SECTION I: GENERAL INFORMATION Refer to IACUC Guides (“Guidelines”) which can be found on the Regulatory Affairs web site (www.upenn.edu/regulatoryaffairs) for guidance when completing this form. 1. Goals and/or Benefits Of The Proposed Research in Lay Language Provide the goals of the experiment. Include all anticipated benefits. This must be explained in terms that can be understood by a lay person without a scientific background. “Procedures involving animals should be designed and performed with due consideration for relevance to human or animal health, the advancement of knowledge, or the good of society.”(Limit: half page) 2. Protocol Category - See Assigning Pain and Distress Levels To Animal Use Protocols for a more complete description A. Please assign this protocol a pain/distress category using the Penn categories (A, B, C). Category A No pain or distress beyond that involved in the restraint, injections, or collection of samples. An example of this level is tissue harvest (taking tissues from animals which have been euthanized following the 2000 AVMA Guidelines and exsanguination under anesthesia). Category B Potential for pain/distress but relief is provided by anesthetic/analgesic/tranquilizing drugs as appropriate-see below***. Please note that the USDA has defined any surgery as potentially painful (this includes both survival and non-survival surgery). Other examples include: antibody production, tumor production, perfusion, and use of neuromuscular blocking agents (NMBs)*** ). 2 Revised May 2009 Category C Pain or distress not relieved by sedatives or analgesics (see below***). Provide a scientific justification as to why you cannot use sedatives or analgesics. This justification may be included on the University's USDA Annual Report, which is available to the public through the Freedom of Information Act. *** The following protocols will be sent by the IACUC staff to a ULAR veterinarian for their comments: 1) All Category C protocols 2) All Category B protocols that use USDA covered species; 3) All protocols that employ neuromuscular blocking agents. *Protocols requiring veterinary review will not be reviewed by the IACUC until the veterinarian’s comments have been received.* B. There is is not an alternative or alternative method (tissue culture, computer models, bacterial cultures etc.) available for this proposed work that does not require the use of living animals. If there is, why must you use animals? C. Does this research duplicate previous work? YES , NO If yes, why is this duplication necessary? D) If this protocol is categorized as level "B" or "C", by law you must consider alternatives to potentially painful/distressful procedures. See Guideline 18. In the following section, check which of the following databases and other sources have been searched to assure that the proposed experiments do not unnecessarily duplicate previous experiments, do not cause undue distress or pain, and that alternative species or alternative methods have been considered and are not available. The search should include “refining” by using less stressful procedures, “replacing” the species selected, with one lower on the phylogenetic scale, and “reducing” the number of animals you are requesting. The search should have been conducted within the last 3 months. Literature Search Check Database Date Search Performed (X) Medline Agricola Current Research Information System(CRIS) Biosis/Life Science CAB Abstracts Embase Altweb (http://altweb.jhsph.edu/) Years Covered 3 Revised May 2009 Association of Veterinarians for Animal Rights http://www.avar.org (For Teaching Protocols) Literature Review Other(s) You must provide a written narrative of the methods and sources used to consider alternatives to procedures for research and teaching that may cause pain or distress to the animals. Please include your search strategy along with the key words and linked key words. The following key words are provided as suggestions to use when looking for alternatives: “animal model, animal testing alternative, animal alternative, and invitro techniques”. Whether one or more of these terms are used or whether more suitable words of your choice are used, the search must reflect a sincere effort to consider alternatives. Alternatives to both the animal model and the procedures being used must be addressed in the literature search. Therefore, include in your search words EACH PROCEDURE that has the potential for causing pain/distress to the animal. Search Results: For help with your literature search or additional databases contact the Animal Welfare Information Center at www.nal.usda.gov/awic/databases/database.htm or (301) 504-6212 or awic@nal.usda.gov. 3. Description Of The Proposed Research Describe in general terms what procedures will be performed. The following items should be included; the experimental design (including treatment groups and appropriate controls); the endpoint of the experiment; and any unusual procedures not covered in any of the IACUC Guidelines. 4. Species, Species Justification, and Total Number Of Animals A. Provide the total number of animals anticipated to be used for the approval term of this protocol (not to exceed 3 years). Please include those animals that will be 1) “procured” i.e., purchased and/or obtained from outside the University (approved animal breeders or other institutions), 2) “transferred” from other protocols (list protocols, if applicable), or 3) “produced” within this protocol (breeding). All animals, regardless of their source, must be ordered or transferred through the ULAR Procurement Office: 215898-2564. Species and strain Number Procured Number Transferred (include protocol #) Number Produced Total #= Total #= Total #= 4 Revised May 2009 Grand total/species= B. Species Justification Provide separate justification for each species listed on the protocol. Why must you use the species you have requested? (A separate justification must be provided below for each species.) C. Animal Numbers What is the scientific justification for the number of animals to be used (i.e. statistical validity, previous experience, etc.)? Please be aware of the following when planning your experiments: Limit animal involvement by using the minimum number required to obtain reliable results. Use non-animal methods, such as mathematical models, computer simulation, or in vitro biological systems if possible. Avoid or minimize animal discomfort, distress, and pain as in consistent with sound scientific practices. Use appropriate sedation, analgesia, or anesthesia when your procedures will cause more than momentary pain or distress. Do not perform surgical, or other painful procedures on non-anesthetized animals. In addition, include information such as the number of control and experimental groups, number of animals per group or other reasoning. A table/flow chart describing experimental groups and controls MUST be provided. 5. *UPDATED* Protection of Humans – Use of Substances Potentially Hazardous to Humans Animal genus and species used in this protocol: Please indicate if you will be using the following in animals: A. Microbiological Agents: (i.e. bacteria, viruses, parasites, fungi, etc) Yes No Please list: 1. 2. 3. 4. 5. B. Human Source Material: (human blood, tissue, cells, or body fluids) Yes No If yes, please describe: C. Tumor cell lines: Yes No If yes, please indicate cell line source and description: Human Non-human primate Other Description 5 Revised May 2009 D. Recombinant DNA (rDNA): 1. Does this protocol involve the generation of rDNA or the use of rDNA in animals? Yes No If yes, please list the rDNA construct(s) and IBC number(s) in table below. 2. Are you using rDNA for the DEVELOPMENT of genetically modified animals (includes transgenics, knock-outs or -ins, etc.)? Yes No If yes, please list the rDNA construct(s) and IBC number(s) in table below. 3. Does this protocol involve the BREEDING of genetically modified animals to produce a new strain of genetically modified animals? Yes No Please describe: 4. Please provide all information below from a current IBC approval letter. Note: All recombinant DNA (including purchased or gifted rDNA) must be registered with the IBC prior to generation and/or use, and must be renewed every 3 years. Registration forms and NIH guidelines can be found at the EHRS website (http://www.ehrs.upenn.edu/protocols/rDNA.html): Construct Name IBC # BSL ABSL E. Biosafety Levels: Please indicate the biosafety level and animal biosafety level at which you will be working. Biosafety Level: 1 2 3 Animal Biosafety Level: 1 2 3 N/A F. Transport of Animals: 1. Will animals be taken out of the animal housing facility? Yes 2. Will animals be moved through patient areas? Yes If yes to questions 1 and/or 2: Indicate the rooms/buildings involved: Indicate type of containment to be used for transport: G. Hazardous Materials: 1. If this protocol involves the administration or treatment of live animals with hazardous chemicals, toxins, anesthetics, and/or drugs, please list in the table below. 6 Revised May 2009 Substance Dose Route of Administration Poses hazard to humans through direct or indirect contact with the animal and/or its bedding (Yes or No) 2. Describe how the hazard will be controlled for all substances in the chart above that poses a hazard to humans: 3. If this protocol involves the use of inhalation anesthetic, please indicate a waste anesthetic gas scavenging method: Active scavenging with a vacuum system Filtering with charcoal canister Used in a fume hood or a Class II type B biosafety cabinet Other (describe): 4. If this protocol involves the perfusion of animals larger than rodents, indicate where the perfusion will take place: H. Radioisotopes Yes If radioisotopes are used, a radiation Protocol Summary Form (http://www.ehrs.upenn.edu/programs/radiation/licensing.html) must also be submitted to EHRS. Isotope(s): Licensee Name: Activity administered per animal: Where will radioisotope administration occur? Bldg: Rm.#: Will animals be returned to their original facility? Yes No If no, please provide the Bldg. Rm.# and contact ULAR Rodent Quality Assurance prior to irradiation and complete the appropriate transfer forms and bleeding procedures (215.898.4008). I. Irradiation Yes (x-ray irradiators fill in “X-Rays/CT section below) Irradiator Licensee: Location of Irradiator: Name of Users: Will animals be returned to their original facility? Yes No If no, please provide the building room # and contact ULAR Rodent Quality Assurance (215-898-4008) prior to irradiation and complete the appropriate transfer forms and bleeding procedures. 7 Revised May 2009 J. X-Rays/CT Yes Please indicate the type of equipment: Radiographic X-Ray Computed Tomography X-Ray Irradiator Other (please specify): Location of machine: Fluoroscopy Cabinet X-Ray K. Lasers Yes 1. Laser manufacturer and model #: 2. Maximum power output: 3. Operational power: 4. Mode of operation (continuous wave or pulsed): 5. If pulsed, pulse width: Pulse repetition frequency: 6. Exit beam diameter: 7. Delivery method (i.e. fiber): 8. Building and room number where laser will be used: 9. Names of personnel who will operate laser: 6. Protection of Animals—Use of Substances Potentially Infectious to Animals. [ULAR Rodent Quality Assurance required] All protocols using any of the materials below must be reviewed and approved by the University Laboratory Animal Resources (ULAR) Associate Director for Diagnostic Services and Rodent Quality Assurance prior to final approval by IACUC. Submit one copy of the protocol to the ULAR, Attention Rodent Quality Assurance, 177E Old Vet Quadrangle, 3800 Spruce St., mail code 6009 or by FAX: 215-746-4146. For questions, call 215-898-4008. Answer all questions below that apply: a. Surveillance testing of cells, plasma, tissues of animal (including human) origin to be injected into rodents. To prevent the possible spread of pathogens into our rodent colonies, all cells, plasma, tissues of animal or human origin and passed through a rodent, and that are to be injected/inoculated into any rodent must be tested for possible contamination by certain infectious agents. Please refer to the University of Missouri PCR Profile Impact II for Mice and Impact VI for Rats at www.radil.missouri.edu for a description of infectious agents. Test results must be submitted to the ULAR Associate Director for Rodent Quality Assurance or his/her designee for review and approval. 1). What procedure (PCR, MAP, RAP Testing etc.) was done to evaluate this tissue for possible contamination? Origin of Material Test(s) Performed Date Laboratory performing test Test result 8 Revised May 2009 2). How is this tissue being maintained? Frozen and/or in tissue culture? Note: If the tissue is being maintained in rodents in a facility other than that in which the recipients are housed, permission MUST be obtained from the Associate Director or his designee for housing these recipient animals. Note: Tissues that tested positive may still be used but must be used in containment with arrangements made with the Associate Director or his designee. b. Use of agents that are potentially infectious to other animals. All agents that are potentially infectious to other animals in the colony must be listed in this section and approved by the Associate Director for Diagnostic Services and Rodent Quality Assurance. Agent Natural host(s) List any species the agent has the potential to infect including people 7. Removal Of Animals From Housing Facility A. In which animal facility will the animals be housed (if known)? B. Will animals be taken out of the animal housing facility? YES , NO If “YES”, please include all buildings and room numbers in Question 10 “Procedure Checklist”. If the procedure is not included, please use the “Other” box to provide the information. C. Transport of Animals 1) What type of containment will be used for transport? 2) Will animals be transported outdoors? YES , NO If ”YES”, please refer to Guideline 16. 3) Will animals be transported through Human Patient Areas**? YES If “YES” please complete the chart below. Species , NO Patient Area **Before receiving IACUC approval, the Principal Investigator must provide written permission from the HUP Infection Control Office, 9 West Gates/4283, 215.622.6995 or the Presbyterian Infection Control Office at 215.662.9260, describing the route to the location where the animals will be used. 9 Revised May 2009 D. Will live animals be returned to the original housing facility? YES If “NO”, where will animals be returned to? Building Room , NO and Why? If the animals need to go to a different facility, transfer forms must be submitted to the ULAR procurement office and approved by the Diagnostic services office (www.ular.upenn.edu). For guidance on the removal of infected animals from containment or their transport through human patient areas, consult Guideline 16, available at our web site, IACUC Guidelines and Policies 8. Occupational Health Program- [Form E Required] The Occupational Health Forms (IACUC Form E and IACUC Form E-NBC) are available at IACUC Forms and MUST be completed and submitted with the protocol. Provide information for each individual with animal contact. “Animal contact” is defined as contact with the animal itself and unpreserved animal products (e.g., tissues, body fluids, discharges, urine, blood or feces) or items contaminated by these products. Return ONLY one copy of the Occupational Health Form to the Office of Regulatory Affairs with the original protocol. This form can be obtained at our web site: IACUC Forms If this protocol is classified as an Animal Biosafety Level 2 or higher risk category, you and your staff are encouraged to participate in the Animal Handler’s Occupational Health Program. If you are using non-human primates, it is mandatory that you and your staff participate in the Animal Handler’s Occupational Health Program and send the IACUC confirmation on a yearly basis with the protocol renewal form. To arrange for a consult, contact Occupational Medicine and Health Services at HUP at 215.662.2354. 9. Personnel, Qualifications and Training- It is the responsibility of the Principal Investigator to assure that all personnel performing any procedures on animals receive the proper training (items a-d below). A) List all personnel that will be working with animals and/or animal by-products. In addition, the Principal Investigator and all co-investigators should be listed and their roles described even if they are not handling the animals or animal by-products. Name Position Penn ID number* IACUC on-line orientation ** Yes or No ULAR “Hands on training” Yes or No (see 9c) *The Penn Card ID # is the eight digit number sequence on your Penn ID card. **Required for ALL personnel every three years: Please visit our web site (www.upenn.edu/regulatoryaffairs) for the information on the on-line IACUC Orientation course and the ULAR “Hands on Training” information. B). Personnel Qualifications: List the qualifications of each of the individuals above (9a). Be sure to include: 10 Revised May 2009 degree(s), years of experience with the species, and skills and specific procedures described that they will participate in (i.e. surgery, blood draw, injections, euthanasia, tissue harvest, imaging, etc.). If an individual is not working with animals and/or animal by-products please describe their role in the project. If individuals do not possess the appropriate qualifications and/or skills, please indicate how they will be trained. C). Animal Handling: If you have never worked with the species in this proposal at the University of Pennsylvania and/or have never taken the ULAR “Hands On Training” course for the species listed in this protocol you will need to be certified in animal handling/procedures. If the individual is not working directly with the animals or animal by-products, hands on training is not required. For information and to register for the next available hands on training class please see the ULAR website, www.ular.upenn.edu. Space is limited so you MUST REGISTER. Special technique training will be provided by qualified individuals listed on this protocol OR arranged through the ULAR Training Division (www.ular.upenn.edu). All training must be documented and faxed to the ULAR Training Division at 215-573-9998 . d). All personnel who work in laboratories are required to attend annual health and safety training. EHRS will notify the principal investigator if all participants are not current in health and safety training. For information about training requirements and schedules, consult the EHRS web site, http://www.ehrs.upenn.edu/. 11 Revised May 2009 SECTION II: SPECIFIC PROCEDURES 10. Procedure Checklist. Please check the procedures to be used in this protocol and provide detailed information in the appropriate section. Those questions already checked off must be answered. Check (X) x Procedure 11 12 13 14 15 16 17a 17b 17c 18 19 20 21 22 23 24 25 26 27 28 29 30 Other Building Room # Tissue Harvest (removal of tissue from dead animals) Imaging Studies Use, Production of Rederivation of Transgenic Animals Animal Identification Use and/or Production Of Monoclonal Or Polyclonal Antibodies Blood Collection Survival Surgical Procedures Terminal non-Survival Surgical Procedures Multiple Surgical Procedures Pharmaceutical Grade Agents Non-surgical Procedures With the Potential for Pain and Distress Use Of Neuromuscular Blocking Agents Prolonged Physical Restraint & Restriction of Movement Capture/Restraint of Wildlife Tumor Production Food &/Or Water Restriction Catheterization or Instrumentation in Animals Special Housing and Husbandry Procedures Special Diets Footpad Injection Drug license and location information Final Disposition of Animals and/or Euthanasia (mandatory) Briefly Describe Procedure 11. Tissue Harvest –performed on non-living animals (for methods of euthanasia see Question 30) Yes a. Tissues/Organs/Body Fluids being harvested include: b. What method of euthanasia will you be using? (please include dose and route of administration) 12. Imaging Procedures A. Location – If you are using a Small Animal Imaging Facility, please contact Sallie Livingston, SAIF Coordinator, Dept. of Radiology, at Sallie.Livingston@uphs.upenn.edu to apply for all of the imaging procedures below. 12 Revised May 2009 Check (X) Procedure MRI / MRS* PET SPECT CT Optical Ultrasound Other Location Species (list all that apply) Duration of Imaging HUP - Founders Richards 5th Floor Richards 5th Floor Richards 5th Floor CRB 437A Stemmler 342 Building Room * Before receiving IACUC approval for MRI / MRS usage, the Principal Investigator must provide written permission from the HUP Infection Control Office, 9 West Gates/4283, Phone 215.662.6995 to the Office of Regulatory Affairs. For guidance on the removal of infected animals from containment or their transport through human patient areas, consult Guideline 16, available at our Guidelines and Policies page. B. Description of Procedure – a. Please describe the imaging procedure(s) including anesthetics (dose, route), and transport of the animals (route, enclosure, how animal will be monitored, etc.). If multiple imaging procedures are to be done please give details regarding the sequence of events and time course (hours, days) between imaging. In addition for SPECT and PET imaging please go to (http://www.ehrs.upenn.edu/programs/radiation/licensing.html) and submit a Protocol Summary Form to EHRS (may be submitted electronically). b. Will the animals be euthanized at the conclusion of each procedure? YES NO If “NO” and the species is MICE and/or RATS, the animals must remain in the designated Richards Small Animal Imaging Facility (SAIF). Please contact ULAR Rodent Quality Assurance prior to imaging and complete the appropriate transfer forms and bleeding procedures 215.898.4008. If “NO” and species is anything other than mice and rats, arrangements must be made through ULAR procurement (215) 898-2564 as to where the animals can be housed. 13. Use, Production, or Rederivation of Genetically Modified Animals, i.e., any animal in which there has been a deliberate introduction or removal of genetic information by experimental manipulation (includes transgenics, knock-outs or ins, etc.) A. Does this protocol involve the USE of genetically modified animals? YES , NO 1. If this protocol involves the USE of genetically modified animals previously developed, please provide the name of the Principal Investigator and institutional affiliation of the source (include Penn protocol # if 13 Revised May 2009 applicable): or state where they are purchased from: (Vendor name): 2. Describe the phenotype of each strain used and list any conditions that are not normal in healthy animals and what course of action will be taken to relieve any pain/distress: B. Does this protocol involve the DEVELOPMENT or REDERIVATION of genetically modified animals? YES , NO . If animals will be developed, Institutional Biosafety Committee (IBC) and EHRS approval are required. For rederivation, EHRS approval is required. Submit a recombinant DNA registration document (http://www.ehrs.upenn.edu) and a copy of the animal protocol electronically to EHRS (approvals@ehrs.upenn.edu). 1. YES I am producing my own animals. If YES, you must request, in question 4, the necessary number of animals required to develop the founders or to re-derive. 2. YES Transgenic Core Facilities with approved IACUC protocols will produce the foundation breeders/perform the re-derivation. Protocol number of transgenic core: 3. Since these animals may develop behavioral or physical conditions that are not normal in a healthy animal, do you anticipate any such conditions? YES , NO If yes, please describe the following: a. Potential abnormal phenotypic conditions and how frequently there are abnormal phenotypic conditions : b. How often will the animals be observed? : c. Action to be taken to relieve pain/distress (eg. Euthanasia/call ULAR vet/analgesia): C. Method Used for Genotyping: Please indicate what method you will use to sample the DNA from the animals. Please see Rodent Genotyping Methods and Rodent Identification Methods for guidance. 14. Animal Identification – ALL SPECIES *UPDATED* For rodents, see Rodent Identification Methods for guidance. 1. 2. Micro-Tattooing: Requires ULAR “Hands On” training and may require anesthesia. Please indicate what type of anesthesia you will use along with the dose and route. Proof of “hands on training” should be submitted with this protocol. MicroChip Please describe: 14 Revised May 2009 3. Ear-tagging Please describe: 4. 5. 6. Ear-punching: Only to be done on weaned rodents. Please describe: Toe Clipping: A method of identification of small rodents, should be used ONLY when no other individual identification method is feasible and should be performed ONLY on altricial neonates. (i.e. animals considered helpless and whose eyes have not yet opened ; Mice < 12 days old and Rats < 7 days old. Please provide a scientific justification for using this method: Other: Please describe: 15. Use and/or Production of Monoclonal or Polyclonal Antibodies IN LIVE ANIMALS. *UPDATED* I will produce monoclonal or polyclonal antibodies: A. YES , NO If yes, I will follow MonoClonal Antibody Production and Polyclonal Antibody Production as appropriate. If you will not be following the guidelines, please describe the process you will follow: B. YES , NO Complete Freund’s Adjuvant will be administered. Please refer to The Use Of Complete Freund's Adjuvant in Laboratory Animals. Provide scientific justification for use of Complete Freund’s Adjuvant: C. YES , NO Are you producing monoclonal antibodies using the mouse ascites method? If yes, you must scientifically justify your use of the ascites method and explain why in-vitro alternatives cannot be used. Please read MonoClonal Antibody Production prior to answering this question. D. YES , NO I am purchasing monoclonal or polyclonal antibodies to be used in animals from a commercial source. I understand that I must purchase from sources with a PHS Assurance Statement and/or a USDA license as appropriate. 16. Blood Collection See Blood Collection for guidance. Species: Anatomic Site: Volume collected: (ml/kg) Bleeding interval/frequency: Note: Maximum allowed in any 3 week interval is 10 ml/kg. 15 Revised May 2009 Method of restraint used for blood collection: NOTES: Retro-orbital bleeding requires anesthesia. Indicate the dose of injectable or inhalation anesthetic agent to be used. If >5 ml/kg (0.5% of the body weight) is collected at one time it is recommended that the total volume be replaced with sterile fluids given SQ or IV. This is mandatory in USDA covered species (everything except mice, rats, fish and birds). Scientific justification must be given to exceed the above guidelines. 17. Survival Or Non-Survival Surgical Procedures *UPDATED* ALL SURVIVAL SURGERIES must be done in a manner that will maintain a sterile surgical field. Rodent Surgery and Post Anesthetic Monitoring and USDA Species Surgery Guideline describe the techniques that must be followed in rodents and USDA covered species, respectively. ALL SURVIVAL SURGERIES must include a provision of peri- and post-operative analgesia, as determined by the surgical model and in consultation with a ULAR veterinarian. See Rodent Anesthesia and Analgesia Formulary for additional guidance for rodent surgery. All survival surgeries in any species must be done in IACUC approved surgical facilities using aseptic technique. Note: A ULAR veterinarian’s review is required for all surgeries (see Section I). A. Category Check Appropriate Surgery: Species Type of Surgical Procedure Terminal (non-survival) surgery will be performed. Survival surgery will be performed once per animal Multiple survival surgery will be performed Non-survival second surgery will be performed B. Location Check (x) or Provide Appropriate Location of Surgery and Records: 1) All of the locations below require semi-annual inspections by the IACUC SOM-ULAR Surgery Suite-Richards 7th Floor SOM-ULAR Surgery –Stemmler 5th Floor SOM-ULAR BRB 3 Surgery SVM-VHUP 4115 floor SVM-Student Surgery 2107 SVM-ULAR Surgery Vet. 273E-C & 272E-D SVM-NBC Hospital Surgery 16 Revised May 2009 SVM-Vet Rodent Procedure Room 269E Investigators Lab. Bldg./Rm. No. If other, specify 2) Where are the surgical and post-op records maintained? Building: Room: C. Anesthetic/Analgesic Regimen Pre-Anesthetic/Analgesic, Anesthetic Agents & Intra-Operative Analgesic Species Preanesthetic agent mg/kg Preanalgesic agent mg/kg Anesthetic: Induction Dose (mg/kg or % gas) Anesthetic: Maintenance Dose (mg/kg or % gas) IntraOperative Analgesic mg/kg Route 1) Describe below how you will determine depth of anesthesia and how it will be increased if necessary. 2) Description of the surgical procedure MUST be provided. Include description of preparation of the surgical site, anesthesia, peri-operative analgesia (for survival surgery), the actual procedure, and method of closure. Please include if there is an expected mortality rate with this procedure. You may add an additional page if needed. 3) Based on the surgical procedure (e.g. cardiovascular or cardiopulmonary manipulation, cerebral vascular catheterization, etc.) do you anticipate the possibility of sudden death during the post-operative period? Yes , No . If yes, please describe. 4) Do you anticipate the need for postoperative support? Yes , No If Yes, complete “5” below. If NO, how will you observe and monitor the animal(s) during the post operative period? 5) I anticipate that post-operative care will be required. Describe post-surgical care, including (1) 17 Revised May 2009 clinical signs of post-operative pain, (2) frequency and time course of observations, and (3) person(s) who are responsible for post-operative care, observations, and record keeping. Do not include “PRN” or “as needed” without describing pain assessment criteria. Post-Operative Analgesics and Sedatives (Guideline 12, “Post-operative Surgical Care”) Species Agent Dose mg/kg Frequency of Adm. 6) Multiple Survival Surgeries: Please justify the need for multiple survival surgeries. 18. Pharmaceutical or Better Grade Agents will be used Yes No If NO, describe reasons here and explain how the drug will be prepared in a sterile fashion? Non-pharmaceutical grade drugs may only be used if pharmaceutical grade or better agents are not commercially available. All drugs must be used prior to their expiration date. 19. Non-surgical Procedures With the Potential for Pain and Distress: Yes a) Describe the procedure(s). Please include dose and route of anesthetic agents, if applicable. b) Describe the clinical signs of pain/distress associated with the procedure(s). c) Describe the frequency of observation, the endpoint of the experiment and the person(s) who will document these observations: d) Describe all treatment(s) to provide relief. If treatment cannot be provided due to the nature of the study this becomes a Category C. (See Question 1) If the treatment includes drugs, complete the chart below: Analgesics and Sedatives Species Drug Dose (mg/kg) Route of Adm. Frequency 18 Revised May 2009 20. Use Of Neuromuscular Blocking Agents: [ULAR Veterinary Approval required] The use of neuromuscular blocking agents (paralytics/NMBs) in any experimental protocol requires appropriate justification. Such agents will not be approved for use without adequate anesthesia/analgesia AND appropriate detailed procedures that definitively demonstrate that appropriate anesthesia/analgesia is maintained throughout the duration of action both prior to and during the use of the neuromuscular blocking agent. See Use of Neuromuscular Blocking Agents in Animals for guidance and specific requirement for using NMBs. Yes Please answer the following: Paralytic agent: Dosage: Route: Frequency: a) Give scientific justification for its use. b) Describe how the animal will be monitored and precautions you will take to insure the animal is under anesthesia and does not suffer pain or distress. c) Pilot study data is attached to the protocol submission. 21. Prolonged Physical Restraint & Restriction of Movement (Prolonged Restraint): Yes Describe the method of restraint, including duration and frequency, and how the animal will be monitored. Explain why restraint is required. 22. Observations, Capture and/or Restraint of Wildlife: Yes If applicable, describe field studies. Have all applicable governmental permits been obtained? Yes , No . If no, please explain. Otherwise please provide copies with this protocol. 23. Tumor Production: Yes YES , NO : If yes, I will follow guidance in Tumor Load Studies. If the answer is NO, please explain how this protocol differs. How you will evaluate the pain/distress to which the animals are subjected? How you will alleviate those conditions? A. B. As stated in IACUC Guideline 2 Tumor Load Studies, the PI will assure that animals involved in tumor load studies are monitored for tumor growth, pain, and distress a minimum of two times per week during the study by qualified laboratory personnel. Yes 19 Revised May 2009 24. Food &/Or Water Restriction: Yes YES , NO If yes, I will follow Food and Water Restriction. If NO, describe here why the Guideline for Food/Water Deprivation cannot be followed. Indicate the duration, frequency and the possible outcomes. 25. Chronic Catheterization Or Instrumentation In Animals: Yes Describe catheterization or instrumentation and include site preparation (clipping, sterile preparation), maintenance and local anesthetic if required. 26. Special Housing and Husbandry Procedures: Yes Special housing and husbandry includes: - reduced cage sizes - overcrowding - housing for immune-compromised - single housed animals - modified light cycles - modified behavioral enrichment procedures - 12-hour housing outside of the animal facility - Wire-bottom or metabolic cages The facility supervisor needs to be notified, prior to their initiation, if any of these special housing and husbandry procedures are to take place in the animal facility. Provide a complete description and justification. Locations where animals will be housed outside of the animal facility for more than 12 hours, a. MUST be inspected by the IACUC and approved PRIOR to relocating the animals. Please call 215-746-6271 for an appointment. Housing Laboratory Animals in the Study Area. b. You MUST complete ONE COPY of Guideline 25- Requests for Housing of Animals within the Study Area (Laboratory) Application Form Request for Housing of Animals Within a Study Area 27. Special Diets: Yes The Animal Facility Supervisor should be notified prior to initiating the study. a) Detail the composition of the special diet and any special requirements needed. b) Who will prepare the diet and how often will the animals be fed? 20 Revised May 2009 c) What will be the consequences of the diet change imposed? 28. Footpad Injection: Yes If the protocol involves injection of an agent into the FOOTPAD of animals, please provide a scientific justification and describe the steps you will take to reduce pain/distress in the animals and how the animals will be monitored: 29. Drug license and location information: Policy on Expired Drugs and Materials a. Where are all drugs, controlled and non-controlled, kept for in vivo use? Building (s) #(s) . and room b. Are you using controlled substances? Yes , No . If yes, please list the substance(s) here provide the name of the individual who holds the Drug Enforcement Agency (DEA) license apply for a license with the DEA please go to this link http://www.deadiversion.usdoj.gov/drugreg/index.html. and . To 30. Final Disposition Of Animals and/or Euthanasia –(MANDATORY) At the end of the experiment the animal(s) will be: a) Transferred live to another protocol. Protocol # The Animal Transfer Form is available from the animal facility supervisor or from the ULAR web site at http://www.ular.upenn.edu. This form MUST be completed in order to transfer animals from one protocol to another. b) Placed for adoption (contact a ULAR veterinarian and see the IACUC adoption policy) c) Released into the wild. Please describe how this will be done. d) Euthanized (MANDATORY: you must answer all applicable questions below) If the *AVMA Panel on Euthanasia is not being followed, you must provide a scientific justification: *These guidelines are available through links to the ULAR and Regulatory Affairs web sites (http://www.ular.upenn.edu, and www.upenn.edu/regulatoryaffairs) Overdose of a barbiturate or euthanasia solution (# of mg/kg) Agent: Dose (mg/kg): CO2 (Dry Ice as a CO2 source is NOT permitted). 21 Revised May 2009 Other: The methods below require pre-sedation or justification for not administering sedatives: Check if you will use: Decapitation Cervical Dislocation Exsanguination Please describe your method of sedation: The PI acknowledges that he/she is the responsible individual for this protocol, and all workers with animals in his/her laboratory will be trained in examining and recognizing the loss of vital signs (heart beat and respiration) to ensure that the animals is fully euthanized. PI must Initial with pen here ____________. SECTION III: INVESTIGATOR’S CERTIFICATION STATEMENT AND SIGNATURE Principal Investigator Certification and Signature 1. I certify that appropriate pain-relieving drugs have been or will be used throughout the entire study to relieve pain or distress whenever it occurs, including postoperative or post procedural care, unless specifically stated otherwise in this protocol. 2. I further certify that the activities in this protocol do not unnecessarily duplicate previous experiments. 3. I certify that all personnel performing any procedures on animals will receive the proper training and will participate in the training programs available (IACUC, EHRS and ULAR-hands on training). A description of this training and experience is provided in this protocol. Proof of such training for all personnel and myself will be provided to the IACUC upon request. 4. I understand that it is my responsibility as the Principal Investigator to ensure that all individuals listed on the protocol have read and understand the procedures described for each species. 5. I am not using radioactive materials, infectious agents or other biologically or chemically hazardous materials in the animal facility other than those included in this protocol and approved by the EHRS. 6. I agree to abide by the provisions of the PHS policy, USDA-APHIS regulations, and the NRC “Guide for the Care and Use of Laboratory Animals.” 7. If I wish to change any of the procedures or personnel as shown on this protocol, I will request IACUC approval by submitting the details of the change(s) as an amendment to ORA. If necessary, I will submit a new protocol. 8. I understand that any failure to comply with the guidelines and requirements of the IACUC may result in suspension of my studies and notification to the funding agency, the PHS and /or the USDA as mandated by law. 9. I understand that animals on this protocol may be used to train personnel listed on this protocol to fulfill the basic species-specific hands-on training requirements and/or for protocol-related technique training by qualified personnel listed on this protocol or arranged through the ULAR Training Division. 10. I certify that all work described in the grant application or award listed on the face page of this protocol is described fully in this protocol. _________________________________________________________ ______________________________ 22 Revised May 2009 Signature of Principal Investigator Date 23
 0
0
advertisement
Related documents
Download
advertisement
Add this document to collection(s)
You can add this document to your study collection(s)
Sign in Available only to authorized usersAdd this document to saved
You can add this document to your saved list
Sign in Available only to authorized users