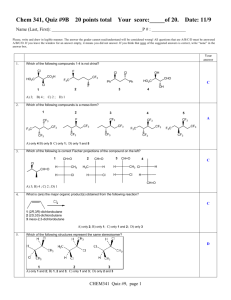
Microsoft Word
advertisement

Abstract Indazole nucleus is present in naturally occurring alkaloids and biologically active molecules. Nigellidine is a natural product containing an indazole nucleus, was isolated from nigella sativa, which is used for the treatment of various diseases. Commonly believed to have carminative, stimulatory and diaphoretic properties. Indazole based heterocycles like indazolo pyrimidines and their derivatives are found to have wide range of activities. Earlier findings on indazole derivatives specifically known to be active as protein kinase inhibitors, cancer cell proliferative disorders, alzheimers disease, viral infections, auto immune and neuro degenerative disorders. In recent years, some of the indazole ring systems are being evaluated as potential drugs for a variety of physiological activities with at least few compounds approved for clinical use. Further, pyrimidine ring in an organic molecule also shows prominent activity against several diseases. Therefore reactions concerning the synthesis of indazole ring and their derivatives mainly pyrimidine fused is a part of scaffold are of current interest. In addition, fluorine or trifluoromethyl group at an appropriate position of molecule promotes lipophilicity to the molecule as a result increased solubility and transport mechanism of the drug enhances in lipid system. Therefore the synthesis of new analogues of indazole based compounds with fluorine or trifluoromethyl group at a specified position have gained significance to find more potent organic molecules, which ultimately become drugs. Thus in this research programme, synthesized a number of novel pyrimido[1,2-b]indazol derivatives and developed novel synthetic methodologies for important organic compounds. The synthesized compounds were subjected to antibacterial, anti-fungal and anti-cancer activity thereby promising compounds have been identified. The interesting aspect of this research work is starting from single compound to a variety of new ring systems. Number of methods adopted in each set of reactions is discussed in various chapters. The total research contribution will be formatted into thesis with five chapters, which includes introduction. The research work covered under each chapter has been briefly outlined below. Chapter 1 Chapter-I covers overview on the importance of indazoles, pyrimidine fused indazoles, oxazines, quinolines and their synthetic methods reported in literature so far upto June 2007. It also covers present work in brief which includes number of methods adopted in synthesis of indazole derivatives, oxazines and quinolines and development of novel methodologies for important organic compounds and their activity studies. Chapter 2 This chapter involves preparation of 3-trifluoromethyl-5- phenyl- 2, 6-dicyano aniline 2 and 3-Iodo-5-trifluoromethyl-biphenyl-2, 4-dicarbonitrile 4, 3-amino - 6- phenyl-4-trifluoro methyl-1H-indazole-7-carbonitrile 5a, 3-Amino-4-phenyl-6trifluoromethyl-1H-indazole-7-carbonitrile 5b. Compounds 5a and 5b were independently reacted with various active methylene compounds and obtained pyrimidine fused indazoles 6, 7, 8, 9 and 10. a. Preparation of 3-trifluoromethyl-5-phenyl-2,6-dicyanoaniline (2) 1-phenyl-4,4,4-trifluoro-1,3-butanedione was reacted with malononitrile in methanol and piperidine and obtained 3-trifluoromethyl-5-phenyl-2,6-dicyanoaniline 2 and oxo-6-phenyl-4-(trifluoromethyl)-1,2-dihydropyridine-3-carbonitrile 3 in 2- different proportions. The compound 2 is an interesting trifunctional intermediate is diazotised followed by reaction with potassium iodide as a result 3-Iodo-5-trifluoromethylbiphenyl-2, 4-dicarbonitrile 4 is formed. CF3 CF3 CN CN piperidine CF3COCH2COPh + CH2(CN)2 MeOH rt 1 + Ph NH2 Ph CN 2 (64%) CF3 O 3 (22%) CF3 CF3 CN NaNO2 / HCl CN CN KI CH3CN / 0OC Ph N H + N2Cl Ph NH2 Ph I CN CN 2 CN 4 Scheme 1 Compound 4 was reacted with hydrazine hydrate in refluxing ethanol resulted two regioisomers such as 3-amino-6-phenyl-4-trifluoromethyl-1H-indazole-7- carbonitrile 5a, 3-amino-4-phenyl-6-trifluoromethyl-1H-indazole-7-carbonitrile 5b in definite proportions. Each regioisomer is separated and identified based on spectral data followed by single crystal X-ray data. CF3 CF3 Ph NH2 NH2 CN N2H4.8H2O / EtOH Ph I CN 4 reflux, 2hrs N N H Ph N + N H F3C CN CN 5a (55%) 5b (45%) Scheme 2 b. Synthesis of Indazolo pyrimidines The indazole regioisomers 5a and 5b were independently reacted with 1,3-diketones (symmetrical and unsymmetrical) in a sealed tube under microwave irradiation conditions obtained pyrimido[1,2-b]indazole derivatives in single step. The role of substituents in 1,3-diketones and compounds 5a, 5b on formation of products 6 is studied in detail. In case of reaction of unsymmetrical 1,3-diketones with compounds 5a/5b resulted exclusively one regioisomer 6a / 6b respectively. R CF3 NH2 R O N CH3COOH / 90 sec O N R' N Ph R' CN N sealed tube / MW / 450w + N Ph CF3 CN 6a 5a R Ph NH 2 N N F3C CN 5b Ph R O sealed tube / MW / 450w O CH3COOH / 90 sec + R' N N N F3C R' CN Scheme 3 6b Compound 5a, 5b were also independently reacted with EMMN/EMCA in sealed tube under microwave irradiation conditions at 450MW gave pyrimido[1,2-b]indazole 7. The 1H NMR of compounds 7a and 7b is quite interesting in a way that the NH2 protons in each of compounds appeared as two broad singlets with different chemical shifts. This is due to extended conjugation with ester carbonyl as a result proton became nonequivalent. CF3 CF3 NH2 EtO N N CH3COOH / 90 sec R' H CN NH2 N Ph CN R' = CN, CO2Et 5a 7a Ph NH2 Ph EtO N R' sealed tube / MW / 450W R' H R' = CN, CO2Et CN N CN + N H F3C R' sealed tube / MW / 450W + N H Ph CN N N CH3COOH / 90 sec 5b N F3C NH2 CN 7b Scheme 4 The compound 5a and 5b were similarly reacted with -ketoesters/ DMAD in sealed tube under microwave irradiation conditions at 450MW gave pyrimido[1,2-b]indazole 8a and 8b respectively. In case of -ketoester the reaction is initiated by nucleophilic attack on carbonyl carbon followed by cyclisation whereas with DMAD, the reaction is addition f ollowed by cyclisation. CF3 CF3 NH2 MeO2 C CO2 Me N N N H Ph OEt H3 C CN sealed tube / MW / 450w 5a O N Ph O O CN CO2CH3/CH3 H N 8a 150 sec Ph Ph NH2 MeO2 C H N CO2 Me N N N H F3C CN 5b . CO2CH3 / CH3 OEt H3 C O O N F3C CN sealed tube / MW / 450w 150 sec Scheme 5 8b O Compound 5a and 5b were further reacted with ethoxymethylene diethylmalonate (EMME) or dimethylamino diethyl malonate (DAMM) in sealed tube under microwave irradiation conditions and obtained exclusively an uncyclised intermediate 9a/9b. They are cyclised in sealed tube under microwave irradiation conditions resulted pyrimido [1,2-b] indazole derivatives 10a, 10b respectively. The mode of reaction is initially the substitution of the ethoxy or dimethylamino group in EMME or DAMM followed by cyclisation on to nitrile or ester carbonyl group. The cyclisation depends on the nucleophilic nature of nitrile carbon (CN) verses ester carbonyl (CO2Et) and ester carbonyl is found to be sluggish. R NH2 N R COOEt sealed tube MW / 450W H COOEt CH3COOH + N H R' R" CN R" = OEt, N(CH3)2 60 sec N COOEt COOEt N H R' CN 9 5 a) R = CF3, R' = Ph b) R = Ph, R' = CF3 R H N a) R = CF3, R' = Ph b) R = Ph, R' = CF3 N COOEt CH3COOH sealed tube MW / 500W 120 sec N N R' OH CN 10 a) R = CF3, R' = Ph b) R = Ph, R' = CF3 Scheme 6 Chapter 3 Synthesis of trifluoromethyl tetrahydro-pyrimido [1,2-b] indazole carbonitrile derivatives. Compound 5a and 5b were treated with formaldehyde followed by various electron rich alkenes in acetonitrile in presence of one equivalent of GdCl3 at room temperature to give the corresponding adducts 11. The reaction proceeds probably by a concerted mechanism is evidenced by the fact that there were no intermediates observed in study. The synthesized adducts were characterized and structures were established based on 1H-NMR data. The low coupling constant (J) of the ring junction protons in the adducts suggests cis ring fusion. The formation of single cis ring fused product indicates that the cyclisation is regioselective. CF3 CF3 NH2 N CN N CN 11a rt N H CF3 CN Ph NH2 N H N Ph 5a Ph H HCHO/CH3 CN/GdCl3 N H Ph H N H N HCHO/CH3CN/GdCl3 N N CF3 rt H H CN 11b 5b Scheme 7 Compound 5a and 5b were similarly treated with benzaldehyde followed by various electron rich alkenes in acetonitrile in presence of one equivalent of GdCl 3 at room temperature to give the corresponding adducts 12. The synthesized adducts were characterized and structures were established based on 1H-NMR and X-ray crystallographic data. The low coupling constant (J) of the ring junction protons in the adducts suggests cis ring fusion. The formation of single cis ring fused product indicates that the cyclisation is regioselective. CF3 NH2 N PhCHO/CH3 CN/GdCl3 N N H Ph N Ph CN 5a CN 12a Ph CF3 NH2 N N N Ph CN 5b Ph H N PhCHO/CH3 CN/GdCl3 N H CF3 Ph H N CF3 CN 12b Scheme 8 Chapter 4 Section A. Part I: Synthesis of trifluoromethyl substituted Benzoxazines. Compound 2 on reaction with phenylmagnesium bromide in ether at room temperature resulted 2-imino derivative 13. Compound 13 is further refluxed in HCl using para toluene sulphonic acid for 6hrs. to undergo hydrolysis and obtained ketone 14 quantitatively. This ketone on reaction with zinc borohydride in ether resulted optically pure alcohol 15. This on reaction with pivalaldehyde in acetic acid gave product 16. CF3 CF3 CF3 Ph CN PhMgBr Ph NH 2 NH Ph NH 2 CN Zn(BH4)2 ether / 0OC / 2hrs Ph Ph NH 2 13 14 CF3 AcOH / 0OC Ph CHO Ph CN Scheme 9 H O / 3hrs CN 15 NH 2 CN OH H Ph CN 2 CF3 O HCl / PTS reflux / 6hrs ether Ph 16 N H Part II: Synthesis of trifluoromethyl aminoquinolines/acridines. Trifluoromethyl substituted Phenyl amino nitrile 2 on reaction with cyclic ketones under microwave irradiation conditions at 450W gave product 17. CF3 CF3 O CN + Ph ( )n ZnCl 2 / 450W MW NH2 NH2 Ph CN N ( )n CN Scheme 10 2 17 Section B: New methodologies for organic synthesis. Part I: Synthesis of benzylidene derivatives. Knoevenagel condensation reaction of aromatic aldehydes and active methylene compounds using ZrOCl2.8H2O in dichloro methane under microwave irradiation conditions gave substituted benzylidene compounds 18. R' R CHO R' ZrOCl2. 8H2O + R CH 600W R" R" 18 Scheme 11 Part II. Synthesi of β-acetamido ketones. Aromatic aldehydes are also treated with substituted benzophenones in acetonitrile and acetylchloride in presence of silica sulphuric acid on heating at 80 OC gave β- acetamido ketones 19. O X CHO + H3C CH3CN./ CH3COCl Y X Y silica-sulphuric acid. 80 oC Scheme 12 Chapter 5 NHAc O 19 Physiological activity and molecular modeling studies. The number of compounds synthesized in each chapter is subjected to screening for anti-bacterial, anti-fungal and anti-cancer activity. Some of the compounds showed promising activity. The data is also co-related with QSAR, molecular modeling and docking studies.The details will be discussed in this chapter. A number of novel compounds such as indazolo pyrimidines, Oxazines and iminotetrahydro-1H-quinolines have been synthesized. Mechanistic paths for each reaction is formulated in order to have a clear understanding of synthetic sequence. All the compounds were screened for various activities in order to identify a promising candidate, which can become a lead molecule in search of potential drug.