X-ray Diffraction (XRD)
advertisement

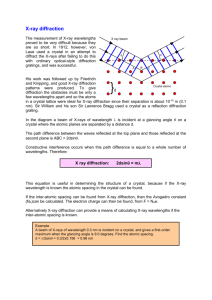
Stanford EMSI Science Teachers Workshop – July 2006: XRD Tutorial and Demonstration X-ray Diffraction (XRD) and Scanning Electron Microscopy (SEM) Background – from Klein and Hurlbut, Manual of Mineralogy, 22nd Edition (1989) and other sources History – the discovery of a mysterious new way to observe Mrs. Roentgen’s hand 1895 – Wilhelm Conrad Roentgen, a professor of physics at the University of Freiberg, Germany, accidentally discovered “X-radiation”. Roentgen was unsuccessful in his efforts to measure the wavelength of X-rays but he was successful in showing that X-rays penetrate matter, including human flesh and show density contrasts. He recorded the x-ray image of his wife’s hand, for example (see below). He was awarded the first Nobel Prize in Physics in 1901 for his discovery of this mysterious new form of highly penetrating radiation. Wilhelm Conrad Roentgen Mrs. Roentgen’s Hand A Simple X-ray Diffraction Experiment 1912 – Max von Laue, a professor of physics at the University of Munich in Germany, suggested to one of his research assistants (Walter Friedrich) and a doctoral student (Paul Knipping) that they use X-rays in the first diffraction experiment. The crystal used for this pioneering experiment was copper sulfate. Von Laue’s reasoning was that X-rays have a wavelength similar to the interatomic distances in crystals, and as a result, the crystal should act as a 3-D diffraction grating. He was able to demonstrate in a later experiment on sphalerite (ZnS) that the pattern recorded by Friedrich and Knipping for copper sulfate on photographic film was due to diffraction of very short wavelength electromagnetic radiation (about 1.5 Å, where 1Å = 10-8 cm) from a regular arrangement of atoms in the ZnS crystal. If the interatomic distances in the crystal are known (as they were in cubic ZnS), then the wavelength of the X-rays can be measured, or alternatively, if the wavelength is known, diffraction experiments can be used to determine the interplanar spacings of a crystal. Von Laue, as well as Friedrich and Knipping were awarded Nobel Prizes in Physics for their discoveries involving X-ray diffraction. 1913 – William H. Bragg and his son William L. Bragg (University of Manchester, England) determined the first mineral structure (NaCl) from XRD, and also determined the atomiclevel structure of many more minerals including those of pyrite (FeS2), fluorite (CaF2), and calcite (CaCO3). They also simplified von Laue’s mathematical generalization and introduced the now famous Bragg equation ( = 2d sin), which describes the condition for 1 Stanford EMSI Science Teachers Workshop – July 2006: XRD Tutorial and Demonstration diffraction to occur in terms of the wavelength of the x-radiation (), the interplanar (“d”) spacings of the crystal, and the angle of incidence of the radiation with respect to the crystal planes (). Father and son Bragg shared the Nobel Prize in Physics for their contributions to this new field, which became known as x-ray crystallography. The Electromagnetic Spectrum, generation of X-rays, and the Braggs’ Equation Nature of X-rays: X-rays can be thought of as waves with wavelengths on the order of 0.1 Å to ~10 Å. The shorter the wavelength, the more energetic the wave. Because of the relatively short wavelengths of electromagnetic radiation in the X-ray region, X-rays are high energy waves and are much more penetrating compared to UV, visible, IR, or radio waves. The conversion between energy, frequency, and wavelength is the well-known de Broglie relationship: E = h = hc/, where is the frequency, h is Planck’s constant (6.62 x 10-34 joule-second), c is the speed of light (2.998 x 108m/sec), and is the wavelength of the radiation (in m). Generation of X-rays: When electrons strike a metal anode with sufficient energy, X-rays are produced. This process is typically accomplished using a sealed x-ray tube, which consists of a metal target (often copper metal) and a tungsten metal filament, which can be heated by passing a current through it (typically 10-15 mA), resulting in the “boiling off” of electrons from the hot tungsten metal surface. These “hot” electrons are accelerated from the tungsten filament (negative bias) to the metal target (positive bias) by an applied voltage (typically 1530 kilovolts). The collision between these energetic electrons and electrons in the target atoms results in electron from target atoms being excited out of their core-level orbitals, placing the atom in a short-lived excited state. The atom returns to its ground state by having electrons from lower binding energy levels (i.e. levels further from the nucleus) make transitions to the empty core levels. The difference in energy between these lower and higher binding energy levels is radiated in the form of X-rays. This process results in the production of characteristic X-rays (i.e. X-rays whose energies are unique to the target metal due to the quantized nature of the electron energy levels of each atom and the unique energies of these energy levels) [Cu K (L3 to K electronic transition: E = 8047.78 eV, = 1.54051 Å), Cu K (L2 to K electronic transition: E = 8027.83 eV, = 1.54433 Å), Cu K1 (M3 to K electronic transition: E = 8905.29 eV, = 1.39217 Å)]. Thus X-rays provide a convenient means of determining what elements are present in a sample because of the unique wavelengths produced by each unique element. A lower energy process that involves the interaction of electrons with the nucleus of an atom in the target metal produces a continuum of lower intensity Xradiation over a broad energy range known as Bremstrahllung. As the voltage on an X-ray tube is increased, the characteristic line spectra of the target element are superimposed upon the continuous spectrum (at right). 2 Stanford EMSI Science Teachers Workshop – July 2006: XRD Tutorial and Demonstration X-ray diffraction: Crystals are ordered, three-dimensional arrangements of atoms with characteristic periodicities. As the spacing between atoms is on the same order as X-ray wavelengths (1-3 Å), crystals can diffract the radiation when the diffracted beams are in-phase. The Bragg equation is given as n = 2dsin. For a given wavelength (), diffraction can only occur at a certain angle () for a given d-spacing. (Figures from; http://hyperphysics.phy-astr.gsu.edu/hbase/hframe.html) Diffraction Single Crystal (Laue) Diffraction – a beam of X-rays of all wavelengths is directed at a single crystal, which sits stationary in front of a photographic plate. A series of diffraction spots surround the central point of the beam, corresponding to diffraction from a given series of atomic planes (at right). Powder Diffraction – a powder is used to ensure completely random crystal orientation to get diffraction from all possible planes. The diffraction pattern can be recorded on a flat photographic film or on a CRT (cathode ray tube). When the incident beam satisfies the Bragg condition, a set of planes forms a cone of diffracted radiation at an angle to the sample. Since the cone of X-rays intersects the flat photographic filmstrip in two arcs equally spaced from the direct X-ray beam, two curved lines will be recorded on the photographic film. The distance of the lines from the center can be used to determine the angle , which can then be used to determine the interplanar d spacing. X-ray powder diffractometers record all reflections using a scintillation detector (in counts per second of X-rays). The pattern of diffracted X-rays is unique for a particular structure type and can be used as a “fingerprint” to identify the structure type. Different minerals have different structure types, thus X-ray diffraction is an ideal tool for identifying different minerals. Above at left; powder film of (a) spodumene (LiAlSi2O6), (b) aragonite (CaCO3), (c) feldspar (KAlSi3O8) and (d) alpha-quartz (SiO2). At right, a diffraction pattern from a mixture of magnetite and hematite using a XRD powder diffractometer. (from; http://spade.ncl.ac.uk/materials/materials/services/xrd.html) 3 Stanford EMSI Science Teachers Workshop – July 2006: XRD Tutorial and Demonstration Primary Uses Limitations X-rays have made it possible to measure the distance between successive atomic planes and XRD can only work with crystalline materials, positions of atoms or ions within a crystal, hence glasses and partially crystalline materials allowing for the determination of crystal cannot be identified using this method. structure. Powder XRD can be used to fingerprint Phases that comprise less than about 3-5 wt.% minerals without any prior knowledge of (depending on crystal symmetry) of a sample crystal structure or symmetry. will not be detected using a bench-top XRD. Relative proportions of mineral mixtures can Mixtures of phases with low symmetry will be be obtained by comparing diffraction line difficult to differentiate due to the larger intensity. number of diffraction peaks. Example I: Name that biomineral This sample is from an equine enterolith (an intestinal stone that was almost fatal for Gordon’s wife’s horse Ripley). This condition has been found to be particularly common in California, due to the type of alfalfa the horses eat (Hintz, 2001). Chemical breakdown of the grain causes the usually acidic environment in the intestinal track to become more alkaline. This causes salts of magnesium, ammonium, and phosphate to precipitate, typically nucleating on a foreign object (such as a piece of plastic, wood, gravel, etc.) (at right). As precipitation proceeds, the stone can become large enough to block the intestinal track (up to 20 cm in diameter) and become fatal. However, enteroliths can be easily removed with surgery if detected soon enough. We will attempt to identify the major phase(s) that comprise this enterolith. Some questions to consider How many phases are present? Does the line pattern for struvite account for all pattern features? If you think there is more than one phase, which one dominates the sample? Given the composition of the enterolith, how might the enterolith sequester other metals or molecules? Struvite can also be found clogging wastewater treatment pipes. Think about what ions could substitute into the mineral structures in the horse’s gut, or in the water pipe. 4 Stanford EMSI Science Teachers Workshop – July 2006: XRD Tutorial and Demonstration Example II: Beach Sand – an aquifer analog Setting: An environmental consulting firm has asked you to characterize the mineralogy of a groundwater aquifer. There has been a recent industrial spill, and they want to know if metals like Pb and Cd will be retarded due to mineral surface interactions. XRD pattern – what do you see? This pattern looks pretty clean, and the sample may only be comprised of one or two phases. There is no large amorphous background, so the phases are probably all crystalline. Fitting – how many phases are present? When we fit the XRD pattern, the characteristic quartz lines account for all peaks in the pattern. There are no extra peaks to fit. From the XRD pattern, we might deduce that the sample is 100% quartz. Visual Observation – what can you see with your eyes? Just by looking at the sample, we can see that there is more than quartz in the sand. Other mineral phases include Fe-oxides, feldspar, mica, and calcite. These phases are likely less than ~2 wt.% of the bulk sample. Some questions to consider Why do you think the XRD pattern only showed the quartz peaks? Are these other phases significant? What is the difference in reporting that the aquifer is 100% versus 99% quartz? Do the other phases behave differently versus quartz with respect to surface sorption of metals? What other techniques might you use to better characterize this sample? If you were interested in a different problem, like flow dynamics, would XRD have been sufficient enough to determine the mineralogy? Conclusions XRD is a very powerful determinative method in the mineral and materials sciences. There are some limitations, but X-ray diffraction can also be a very straightforward and easy to use method to determine the identity of minerals. Some finals questions to ponder: How well does XRD characterize the chemical nature of a sample? Did we learn enough from the XRD patterns alone to understand the mineralogy that dominates reactivity with the enterolith, or the sand? XRD is nearly 100 years old, and still not “out of style”. What would you do to try and improve the technique? 5 Stanford EMSI Science Teachers Workshop – July 2006: XRD Tutorial and Demonstration XRD patterns Example 1 – equine enterolith Example 2 – beach sand 6 Stanford EMSI Science Teachers Workshop – July 2006: XRD Tutorial and Demonstration Scanning Electron Microscopy (SEM) Background Good Reference: J.I. Goldstein, D.E. Newbury, D.C. Joy, C.E. Lyman, P. Echlin, E. Lifshin, L.C. Sawyer, and J.R. Michael (2003) Scanning Electron Microscopy and X-ray Microanalysis, 3rd Ed., Springer: New York, 689 p. The scanning electron microscope (SEM) “sees” low kinetic energy secondary electrons from solid surfaces. When an electron beam interacts with a solid, various types of elastic and inelastic processes occur, including electron scattering and excitation, which produces (1) secondary electrons, (2) backscattered electrons, (3) Auger electrons, (4) characteristic x-rays, (5) bremsstrahlung or continuous x-rays, and (6) photons of various energies, including those in the infrared, visible, and ultraviolet. The fraction of energy deposited by an electron beam in a sample associated with these different processes is dependent on the sample. Secondary and Auger electrons can only be observed when they come from the near-surface region of a solid (typically < 500 Å for insulators, such as silicate minerals, and < 100 Å for metals such as gold). Thus, measurements involving these types of electrons are “surface sensitive”. The reason for the greater escape depth of secondary electron from insulators relative to metals is that inelastic scattering of secondary electron takes place mainly through interactions with conduction-band electrons, which are abundant in metals and significantly less abundant in insulators. Secondary electrons are generated by the primary electron beam as it enters a sample as well as by backscattered electrons as they exit a sample. Secondary electrons, which typically have kinetic energies < 50 eV, are sensitive enough to differences in surface topology that they can be readily observed from the surface of a sample. Such electrons form the basis of scanning electron microscopy. In order to enhance the number of secondary electrons from an insulating sample, the sample is often coated with a thin layer of gold-palladium alloy or another electron-rich conducting material that produces abundant secondary electrons when struck by a focused electron beam. A thin metal coating will not mask surface features or the overall topology of the underlying sample. The gold-palladium coating also conducts electrons away, so that the sample does not develop a significant charge when it loses secondary electrons and other types of electrons. This type of coating is essential for insulator samples, which don’t conduct charged particles. Such samples, if uncoated, would be difficult to image using an SEM because of the fact that they would develop a negative charge (due to build-up of electrons), which would cause the image to become defocused due to deflection of the exciting electron beam. Several figures on the next page show secondary and backscattered electron energy distributions and their paths in solids as well as a schematic of a typical SEM. 7 Stanford EMSI Science Teachers Workshop – July 2006: XRD Tutorial and Demonstration 8 Stanford EMSI Science Teachers Workshop – July 2006: XRD Tutorial and Demonstration SEM Images of meteoritic orthopyroxene surface which has been populated by a filamentous microorganism. A blow-up of a portion of the surface shows rod-shaped calcite crystals, which were originally incorrectly interpreted as nanobacteria. They are instead nano-crystals of calcite. (from Benzerara et al., 2005). mm m SEM image of Acidothiobacillus ferrooxidans on a pyrite surface, showing ferrihydrite precipitate to mthe right and an oxidation rim, which is shown in close-up on the next page. 9 Stanford EMSI Science Teachers Workshop – July 2006: XRD Tutorial and Demonstration SEM lose-up of oxidation rim around A. ferrooxidans showing goethite crystals (smooth laths) and schwertmannite crystals (clusters). The goethite crystals are about 200 nm long. 2 m 10