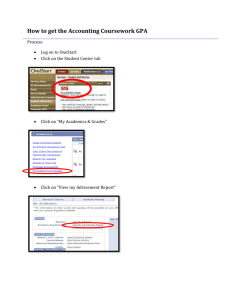
MSE227 Homework AnswersChapters 15_16
advertisement

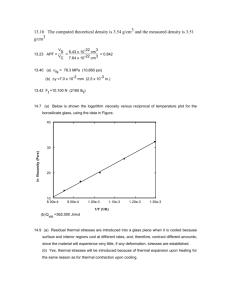
MSE227 Homework Answers Chapters 15 & 16 Chapter 15 15.1 You should have used the rule of mixtures approach to generate the curves below: 15.4 cl* = 1640 MPa Ecl = 60.3 GPa 15.7 (a) (b) (c) (d) 15.11 (a) Ff = 44.7 Fm Fm – 1168 N f = 242 MPa m = 4.4 MPa m = 1.84 x 10-3 Specific Strength: Glass/Fiber: 486 MPa Carbon Fiber: 775 MPa Aramid Fiber (Kevlar): 986 MPa Cold rolled 7-7PH: 180 MPa 1040 plain carbon : 75 MPa 7075 T6 Al: 204 MPa C2600 brass: 62 MPa AZ31B Mg: 148 MPa Ti-5Al-2.5Sn: 176 MPa (b) Specific Modulus: glass-fiber reinforced epoxy: 21.4 GPa carbon-fiber reinforced epoxy: 90.6 GPa aramid-fiber reinforced epoxy: 54.3 GPa cold rolled 17-7PH stainless steel: 26.7 GPa 1040 plain-carbon steel: 26.4 GPa 7075-T6 Al: 25.4 GPa C26000 brass: 12.9 GPa AZ31B Mg: 25.4 GPa Ti-5Al-2.5Sn: 24.6 GPa 15D2 Such a composite is possible if 0.353 < Vf < 0.391. Chapter 16 16.1 (a) The possible oxidation and reduction half-reactions for magnesium in various solutions are as follows:. (i) In HCl, possible reactions are Mg Mg 2+ + 2e- (oxidation) 2H+ + 2e- H2 (reduction) (ii) In an HCl solution containing dissolved oxygen, possible reactions are Mg Mg 2+ + 2e- (oxidation) 4H+ + O2 + 4e- 2H2O (reduction) (iii) In an HCl solution containing dissolved oxygen and Fe2+ ions, possible reactions are Mg Mg 2+ + 2e- (oxidation) 4H+ + O2 + 4e- 2H2O (reduction) Fe 2+ + 2e- Fe (reduction) (b) The magnesium would probably oxidize most rapidly in the HCl solution containing dissolved oxygen and Fe2+ ions because there are two reduction reactions that will consume electrons from the oxidation of magnesium. 16.2 (a) (b) V = -0.011 V Since the V is negative, the spontaneous cell direction is just the reverse of that in (a), or Sn 2+ + Pb Sn + Pb 2+ 16.12 For each of the forms of corrosion, the conditions under which it occurs, and measures that may be taken to prevent or control it are outlined in Section 16.7. 16.13 For a small anode-to-cathode area ratio, the corrosion rate will be higher than for a large ratio. The reason for this is that for some given current flow associated with the corrosion reaction, for a small area ratio the current density at the anode will be greater than for a large ratio. The corrosion rate is proportional to the current density (i) according to Equation 16.24. 16.D1 (a) Laboratory bottles to contain relatively dilute solutions of nitric acid. Probably the best material for this application would be polytetrafluoroethylene (PTFE). The reasons for this are: (1) it is flexible and will not easily break if dropped; and (2) PTFE is resistant to this type of acid, as noted in Table 16.4. (b) Barrels to contain benzene. Poly(ethylene terephthalate) (PET) would be suited for this application, since it is resistant to degradation by benzene (C6H6) (Table 16.4), and is less expensive than the other three materials listed as satisfactory (S) in Table 16.4 (see Appendix C for cost data). (c) Pipe to transport hot alkaline (basic) solutions. The best material for this application would probably be a nickel alloy (Section 13.3). Polymeric materials listed in Table 16.4 would not be suitable inasmuch as the solutions are hot. (d) Underground tanks to store large quantities of high-purity water. The outside of the tanks should probably be some type of low-carbon steel that is cathodically protected (Sections 16.8 and 16.9). Inside the steel shell should be coated with an inert polymeric material; polytetrafluoroethylene or some other fluorocarbon would probably be the material of choice (Table 16.4). (e) Architectural trim for high-rise buildings. The most likely candidate for this application would probably be an aluminum alloy. Aluminum and its alloys are relatively corrosion resistant in normal atmospheres (Section 16.8), retain their lustrous appearance, and are relatively inexpensive (Appendix C).