Final AgendaPeds 11-23-09
advertisement

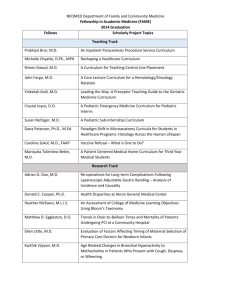
National Pediatric Cancer Registry Work Group December 2-3, 2009 Agenda Goals: To fully explore and understand different data systems used for collection of data about pediatric cancer Explore the needs of pediatric cancer epidemiologists and public health needs Identify methods to address the research needs identified utilizing cancer registries and other databases or other methodologies. Identify opportunities for creating new partnerships between cancer registry community and pediatric cancer researchers to increase the research value of pediatric cancer registries and other research databases Outline the methodology and processes needed to respond to needs of cancer researchers and public health needs. Day One (1:00 PM – 5:30PM) 1-1:30 Welcome and Introductions -Marcus Plescia, MD, MPH, DCPC Director, CDC 1:30-2:00 Background Carolyn Pryce Walker, Conquer Childhood Cancer Act of 2008 -Christie Eheman, PhD., Cancer Surveillance Branch Chief, CDC Data Needs of Epidemiologic Researchers -Debra L Friedman, MD, Vanderbilt-Ingram Cancer Center 2:00-2:15 Questions and Answers 2:15-3:00 Small Group Discussion: What are the primary data needs for epidemiologic research in childhood cancer? What other system issues are important for childhood cancer research (privacy issues, rapid case reporting)? What are the public health needs? What information do central registries collect? Reports Back 3:00-3:15 BREAK 3:15-4:15 Currently Available Data Systems and Data Collection Processes Childhood Cancer Research Network (CCRN) -Julie Ross, PhD. Cancer Registries National Program of Cancer Registries (NPCR-CDC) -Umed Ajani, MD. Surveillance, Epidemiology, and End Results (SEER-NCI) -Dave Stinchcomb, PhD. Children’s Oncology Group Longterm Follow-up Center -Dennis Deapen, DrPH, LA Cancer Surveillance Program Questions and Answers 4:15-5:00 Increasing Rapidity of Case Reporting for Pediatric Cancer Cases – Traditional Methods and New Approaches Traditional and Other Potential Methods for Rapid Case Ascertainment in Cancer Research - Carol Lowenstein, MBA, Harvard School of Public Health Small Group Work: When is rapid case ascertainment important? How Can We Improve/Implement more rapid case reporting of cancer within state cancer registries? Are new approaches like electronic reporting an option? How else can needs be met? Reports Back 5:00-5:30 Ethical and Privacy Issues Human Subjects and Privacy Issues Related to Patient Contact Involving Children - Dennis Deapen, DrPH, LA Cancer Surveillance Program Large Group Discussion: Ethical and Privacy Issues Related to ChildrenMethods of optimizing and methods for balancing those concerns with public health and research needs; state rules and needs; federal rules Day Two (8:30 AM – 12:00 PM) 8:30-9:30 Methods for optimizing and expanding current data systems or building new data systems to meet epidemiologic pediatric cancer research and public health needs Small groups - Explore methods and ideas in an organized way to capture different approaches and concepts 9:30-10:15 Report back from small groups and discussion 10:15-10:30 BREAK 10:30-11 Building Relationships Building relationships among cancer registries, pediatric oncologists and pediatric cancer centers to maximize reporting and research efforts Large Group Discussion: How can we improve our working relationships to maximize efficiency and improve participation in childhood research? 11-11:30 Summary-What Major Themes Arose from Our Discussions 11:30-12 Next Steps and Adjourn