The role of Mage proteins in tamoxifen resistance
advertisement

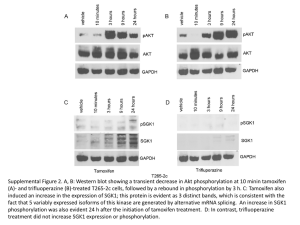
Institute of Cancer School PhD Project Proposal First Supervisor Helen Hurst Second supervisor Louise Jones Centre Tumour Biology Project Title The role of Mage proteins in tamoxifen resistance Project outline including background, aims and methodology (1 page only minimum font size 10) Background: The majority of breast tumours (70%) are oestrogen receptor positive (ER+) and can be treated with tamoxifen, however, a significant proportion of patients will relapse with resistant disease. Newer endocrine therapies have been developed, but meta-analysis of comparative clinical trials suggests that switching protocols, whereby patients receive tamoxifen for 2-3 years before moving to an aromatase inhibitor, offer the best long-term outcomes. Thus, research into the mechanisms behind tamoxifen resistance (TR) remains highly relevant. Expression profiling analysis of wt and TR lines identified MAGEA2 as a significantly upregulated gene. This has been functionaly validated: silencing MAGEA2 in a TR line restores sensitivity to tamoxifen; overexpression of MAGEA2 in ER+ lines confers resistance in vitro and as xenografts in nude mice; MageA expression in clinical samples correlated with a poorer outcome on tamoxifen. Furthermore, MageA2 can form complexes with p53 and inhibit its activation in tamoxifen media. MAGEA2, a member of the class 1 MAGE gene family of cancer-testis antigens, defined by their lack of expression in somatic adult tissues but frequent aberrant expression in tumours. The proteins are characterised by a ~200 amino acid c-terminal MAGE-homology domain (MHD) whose precise function is uncertain although it appears to act as a scaffold for protein-protein interactions. Tumour expression of Mage proteins can elicit spontaneous immunogenicity in patients and immunotherapy strategies to boost these responses, particularly for MAGEA3, are currently undergoing promising clinical trials. Aims: Our data suggests strongly that MAGEA2 overexpression is a determinant, and not merely a correlate, of tamoxifen resistance and acts both in vitro and in vivo to inhibit growth arrest pathways normally induced by tamoxifen. Due to the similarity between class 1 MAGE genes, we hypothesise that most MageA and possibly B and C proteins may act in a similar manner to confer resistance. Project aims are to determine to what extent other class 1 Mage proteins also contribute to resistance and to generate a molecular understanding of the pathways altered by MAGE expression in ER+ breast epithelia. The findings will be used to inform further translational/pre-clinical studies to target MAGE, or the pathways they activate, in conjunction with tamoxifen treatment to improve patient outcomes and establish the utility of Mage expression as a marker of de novo or acquired tamoxifen resistance in breast cancer. Methodology: 1. Additional MAGE cDNA clones (IMAGE) will be stably overexpressed in ER+ breast tumour lines and pools of clones will be tested for growth in tamoxifen as for MAGEA2. Tagged versions of MAGEA2 will be checked for functional equivalence with wt protein, including co-immunoprecipitation (CoIP) with p53 for aim 3. As a surrogate readout of p53 activity, we will use luciferase reporter assays; dose escalation studies with MAGEA2 show progressive repression of p53-dependent reporter genes. 2. Further coIP assays will be used to explore interaction with additional pathways implicated in TR including ER and its co-regulators. MageA2 expression confers resistance to a range of tamoxifen concentrations but not to faslodex, suggesting that Mage proteins also act on the ER. We have also found that the addition of tamoxifen stabilises and re-localises MageA2 to the nuclear compartment. We will create a series of MAGEA2 deletion mutants to investigate which domain is required for its nuclear localisation and its tamoxifen resistant function and determine if this correlates with the ability to downregulate the p53 pathway. 3. Candidate gene approaches, however, do not always lead to new biological insights. We aim to perform a screen to identify additional MageA2-interacting proteins using a functionally verified tagged protein. Complexes will be separated on coomassie 1D gels and identified by collecting LC-MS/MS data on the peptide mixtures generated by proteolytic digestion of the samples using the cross-charged service offered by the Mass Spectrometry Laboratory. Samples will be purified from at least two distinct breast tumour lines, running three replicates (and controls). MageA2-interacting proteins common to both lines will be prioritised for functional validation using further CoIP and siRNA silencing assays. 4. If MageB or C proteins can also contribute to tamoxifen resistance in the functional studies above, then the associations between Mage expression and TR in tumour samples we have calculated to date may be an underestimate. Therefore the tumour cohorts we have will be reassessed for expression of additional Mage (B and C) proteins. We have additional cases of patients who relapsed on tamoxifen treatment from Valerie Spears in Leeds which will also be assessed for Mage expression. Page 1 Institute of Cancer School PhD Project Proposal A short (200 word) summary of the project to go with the studentship advert. The majority of breast cancer patients can be treated with tamoxifen, however, a significant proportion will relapse with resistant disease. Newer endocrine therapies have been developed, but current clinical data suggests that patients respond better if they receive tamoxifen for 2-3 years before moving to another treatment. Therefore, research into the mechanisms behind tamoxifen resistance (TR) remains highly relevant. We have observed upregulation of the cancer/testis antigen MAGEA2 in TR cell lines. Expression of MAGEA2 in sensitive cells allowed continued proliferation in tamoxifen media, while silencing MAGEA2 expression in TR lines restored sensitivity. Furthermore, MageA2 formed complexes with p53 and inhibited its activation in tamoxifen media. Analysis of MageA expression in clinical samples showed a correlation with poorer outcome and MAGEA2-expressing xenografts also grew in the presence of tamoxifen confirming a functional role for MAGEA2 in resistance. The project will use a variety of molecular biology and proteomic techniques to test the wider significance of these data by determining if other MAGE genes can confer resistance. The cellular pathways acted on by Mage proteins will be further explored using co-immunoprecipitation assays and testing for interaction with candidate genes from known TR pathways, including ER itself and to use mass spectromotery to identify novel interactors. The in vitro findings will be validated by reference to clinical samples. Page 2