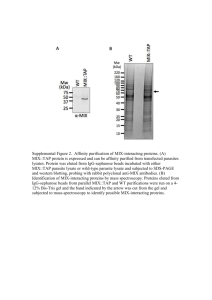
1 - GUL
advertisement

Individual assignment Drug development 7.5 ECTS, KMG045 (Chalmers)/ BIO523 (GU) fall term 2013 Submit your answers (as attached Word, pdf, or text files) by E-mail to Per.Sunnerhagen.gu@analys.urkund.se no later than Friday Dec. 20 Treat one of the following topics, using no more than 2000 words (excluding literature references). All literature is allowed as an aid. The essay is then graded on a scale from 0 to 10. The points are added to the points of the regular final exam. There is not one, but several possible “correct” answers for each topic. The more appropriate and realistic answer (still respecting the upper limit of 2000 words), the higher the grade. Each essay must be an original document produced by the student, and each document will be automatically scanned by “Urkund” in order to reveal cases of plagiarism. It is allowed to co-operate between you in discussions about a literature topic, but each essay still has to be original and unique. 1. Proteins are generally the functional units of cells and are therefore the most logical drug targets. A majority of proteins (some claim that almost all) work in complex with other proteins as heteromeric complexes where both components are equally important for function, i.e. inhibition of either one of the proteins in the complex results in drastic effects on the activity of the whole complex. Thus, in cases where one protein has been identified as an important target to treat a disease, there is great interest to find other proteins that physically link to the first protein. There are various techniques to screen for proteins that physically interact with an important disease protein thereby identifying potentially new drug target. Discuss various means to screen for these new targets, in particular to human GPCRs. GPCRs are the most common class of drug target and therefore of great medical importance. In particular, consider using other organisms/systems as test-bed for the screens and compare those procedures to methods where one can screen for physical interactions directly in the human systems. What are the pros and cons in time, money and outcome? 2. Small molecules (fragments) binding to a protein, are considered attractive starting points for findings leads in a drug hunting campaign. In order to develop the fragments hits into useful leads, several methods have been described (fragment linking, merging, growing, etc). Describe two such methods, using an example from literature for each one. Discuss the reasons the authors had for selecting the particular strategy for each case, and discuss, and possibly explain, the changes in chemical structure that caused the improvement, in affinity or selectivity, obtained in the process. 3. Designing drugs with activity against eukaryotic parasites (Trypanosoma, Leishmania, Plasmodium etc.) presents a formidable challenge. The main reason is that the gene products of these organisms is so similar to our own homologs that it is difficult to avoid side activity against our own cells; in other words there is a great problem of selectivity. Assuming you have access to small molecules with some selectivity against a parasite (i.e. they kill off the parasites more efficiently than our own cells at a given concentration), but the selectivity needs to be improved. Furthermore, the molecular target of these molecules is not known. Discuss how you can use yeast as a tool to identify the target protein (or at least the targeted pathway) of such a compound. Assuming this first step was successful, discuss how you can use yeast in a second step to also improve the selectivity (its inhibitory effect vs. the parasite protein homolog over that vs. the human protein homolog) of the compound by screening among related molecules.